GMP-grade Antibodies for Cell and Gene Therapy
Largest portfolio of recombinant GMP-grade antibodies curated for cell separation, activation, and enrichment steps in cell and gene therapy workflows. Proteintech's GMP-grade antibodies are manufactured in ISO rated clean rooms using animal free components in accordance with USP and WHO standards, ensuring maximum safety and reproducibility which makes it a reliable, scalable, and well-documented solution to accelerate your cell therapy programs.

Recombinant
Curated for greater lot-to-lot consistency and improved scalability and reliable supply
Regulatory-ready
Manufactured in accordance with relevant GMP guidelines as well as USP and Eu.Phr recommendations
Customizable
Customizable pack sizes for easy integration to evolving workflows
GMP Quality

Proteintech is ISO 13485:2016 certified, and our GMP HumanKine products are manufactured and tested in accordance with the relevant sections of the following standards:
- USP Chapter <1043>, Ancillary Materials for Cell, Gene, and Tissue-Engineered Products
- Ph. Eur. General Chapter 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products
- Sterility testing according to USP<71>
- Mycoplasma testing according to USP<63>
- Bacterial endotoxin test according to USP<85>
- Host cell protein and DNA testing according to relevant ICH guidelines
- Purity >95%
GMP manufacturing process
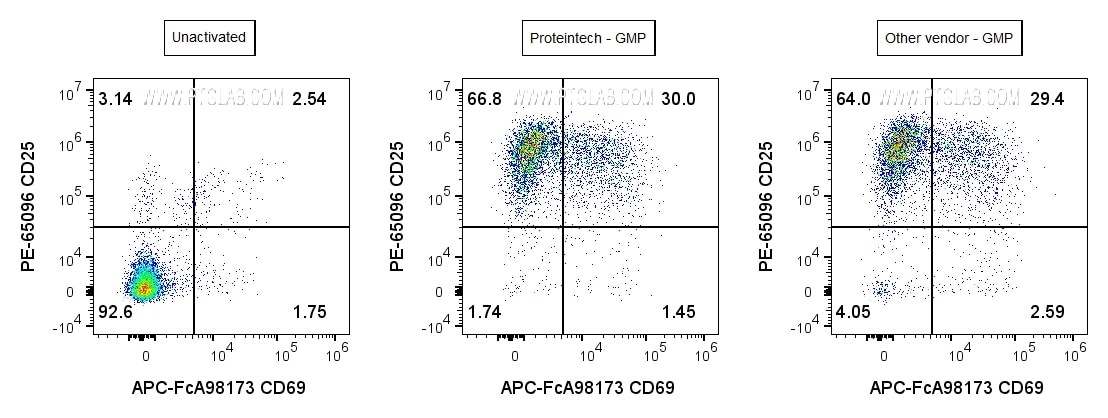
Human PBMCs were incubated for 3 days with soluble CD3 (OKT3) Recombinant Monoclonal Antibody (CT-1000-GMP) at 50ng/mL (center), or GMP-grade Anti-Human CD3 (OKT3) Antibody from another vendor at 50ng/mL (right), or cultured in media alone (left). Cells were then stained with Anti-Human CD3 (AB-65151), Anti-Human CD25 (PE-65096), Anti-Human CD69 (APC-FcA98173). Cells were gated on singlets (FSC-H vs FSC-A), live (Phantom Dye-), and CD3+ lymphocytes.
A New Benchmark in Cell Activation
Proteintech's GMP-grade CD3 antibody delivers equivalent T cell activation performance to leading market alternatives. In side-by-side studies, it induced comparable levels of cell proliferation and activation marker expression (CD25, CD69), confirming its reliability and potency. These results demonstrate that Proteintech's GMP CD3 antibody provides consistent, high-quality performance for T cell activation in cell therapy manufacturing.
CAR-Linker Antibodies for Cell Enrichment
Proteintech's CAR linker-specific GMP antibodies enable reliable enrichment and detection of CAR-engineered cells. Designed to recognize widely used linkers used in CAR scFv constructs such as G4S and Whitlow/218, these antibodies provide consistent performance and high specificity without affecting CAR function. Manufactured under GMP-compliant, animal component-free conditions, they ensure exceptional lot-to-lot consistency and are ideal for both process development and clinical production of CAR-T/CAR-NK cell therapies.
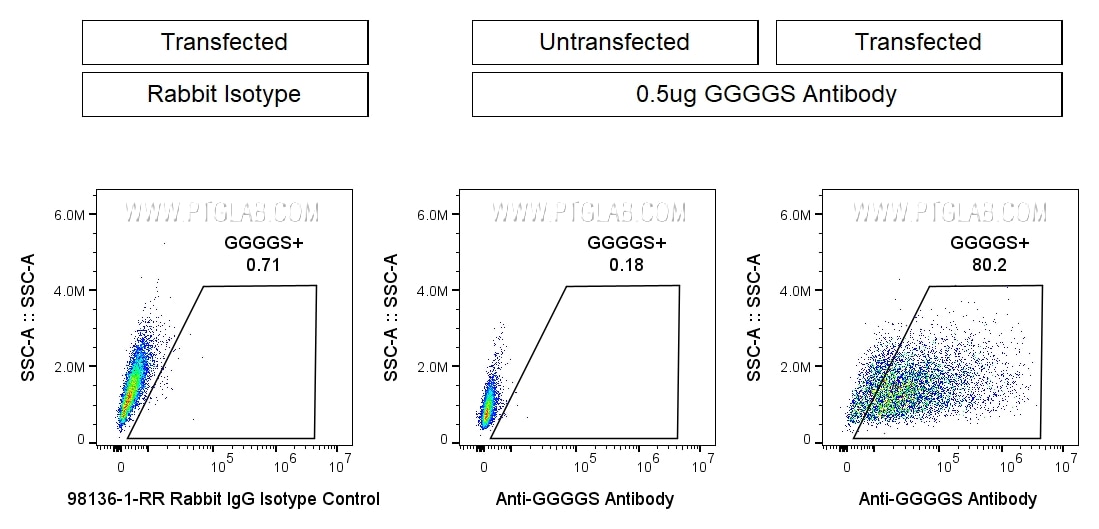
0.5x106 GGGGS-transfected HEK293 cells and untransfected cells (center) were stained with 0.5ug Anti-GGGGS Recombinant Monoclonal Antibody (CT-1002-GMP) (right) or Rabbit IgG Isotype Control (98136-1-RR) (left). All cells were then stained with CL647-conjugated Donkey Anti-Rabbit Secondary (SA00014-7) at 1:500. Propidium Iodide was included in the final resuspension buffer at 1:1000. Cells were gated on Live (PI-), then the cell population in FSC vs. SSC, then single cells, then for positive signal in the APC-A channel.
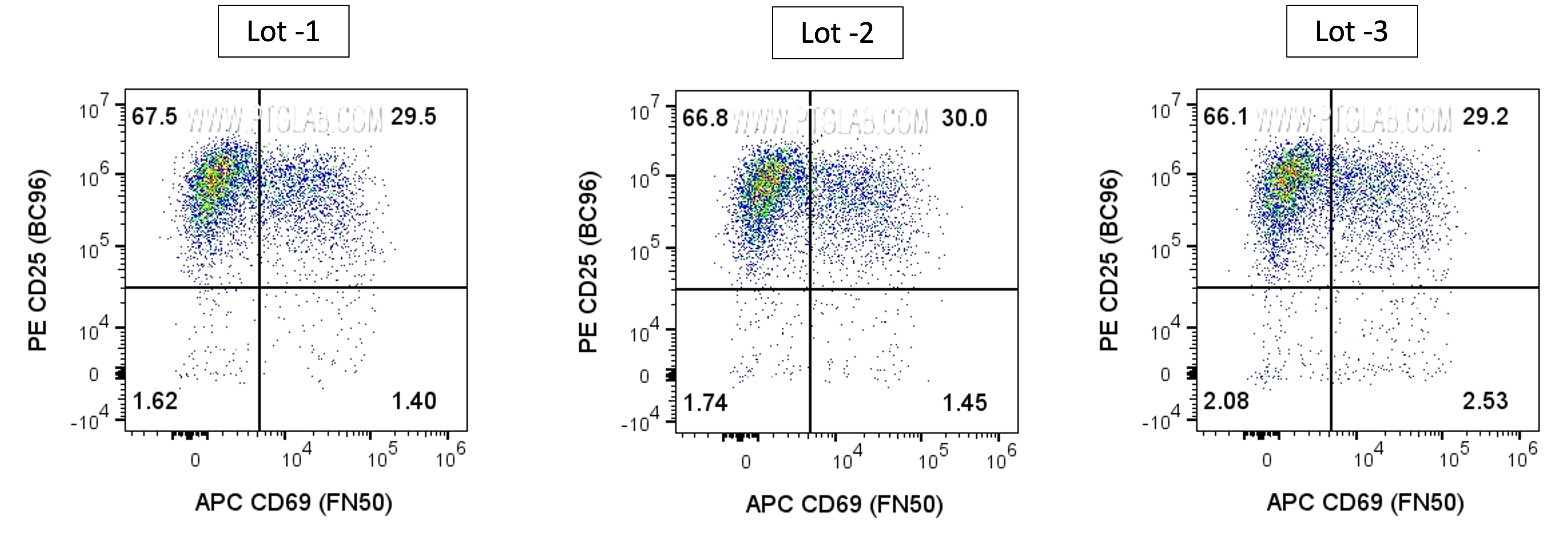
Human PBMCs were incubated for 3 days with soluble CD3 (OKT3) Recombinant Monoclonal Antibody (CT-1000-GMP) at 50ng/mL.Cells were then stained with Anti-Human CD3, Anti-Human CD25, Anti-Human CD69, and Phantom Blue 516 Viability Dye (PD00007); cells were incubated with Fc Receptor Block prior to staining. Cells were gated on singlets (FSC-H vs FSC-A), live (Phantom Dye-), and CD3+ lymphocytes.
Lot-to-Lot Consistency
Proteintech's GMP-grade antibodies deliver exceptional lot-to-lot consistency through a fully recombinant expression system and animal component-free manufacturing process. By eliminating hybridoma variability and serum-derived materials, each production run maintains precise sequence fidelity, purity, and performance. This controlled, chemically defined workflow ensures stable antibody quality, reproducible binding activity, and regulatory confidence across batches.
Pipeline

Supporting Documents
Documents available on request
- Certificate of Analysis (COA)
- Certificate of Origin (COO)
- Certificate of Compliance
- Animal Component free statement
- BSE-TSE free statement
- Viral safety statement
Please provide the following in the email:
- Your company name
- Product name and catalog number
- Document name
Related Products
HumanKine GMP Cytokines
Recombinant proteins for use in translational research, drug discovery, and cell & gene therapy. Smooth, scalable transition from research to the clinic.
CAR Detection Reagents
Reliable and scalable solutions for detection and quantification of Chimeric Antigen Receptor (CAR) engineered cells.
Flow Cytometry Antibodies
Top-cited clones and Proteintech's in-house clones have been conjugated to a variety of dyes for ease of staining and to reduce the length of your protocol. Unconjugated antibodies are also available to give you the most flexibility in building your panel.
ELISA Kits
Explore our range of immunoassay products for highly sensitive detection of secreted proteins, cytokines, growth factors, hormones, chemokines, and more.