Immunofluorescence Introduction
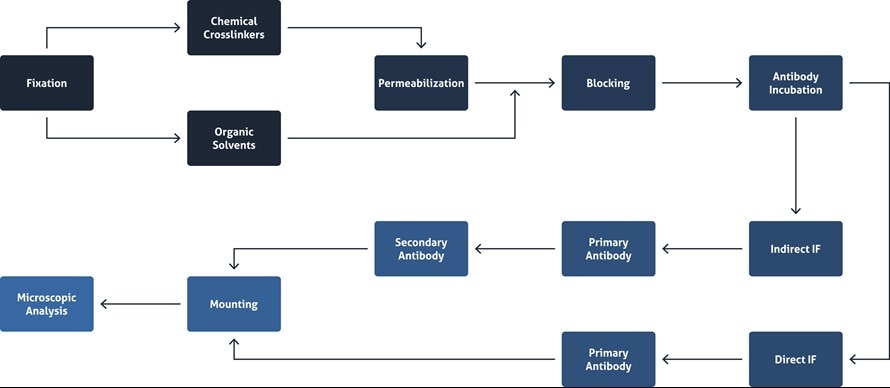
An overview of the immunofluorescence workflow.
What is Immunofluorescence?
Immunofluorescence (IF) staining is a widely used technique in biological research and clinical diagnostics. IF utilizes fluorescent-labeled antibodies in order to detect specific target antigens. Followed by imaging, it is a very direct technique as you can actually see something. Although it is a well-established tool, multiple factors have to be considered and various optimization steps have to be taken to ensure successful staining.
Fundamental Principles
There is no one perfect protocol of how to fix, stain, and analyze cells. Producing beautiful, eye-catching, ready-topublish IF stainings requires optimization of the best conditions for each single step of the protocol in accordance with sample type, target of interest, antibody used, antibody performance, and antigen-antibody combination.
These days, microscope software (e.g., deconvolution software, etc.) is advanced. It helps to enhance the quality of the image and can often be used to overcome the deficiencies of a poorly prepared sample. However, attention has to be paid to these techniques as they might lead to misinterpretation of results. Damaged cell structures or poor labeling of a suboptimal prepared sample can give false data. No microscopy time should be wasted on poorly prepared samples. Therefore, it is essential to appropriately prepare, fix, permeabilize, block, label, and mount your sample. Still, it is common in a lot of laboratories to use the same standardized protocol for all experiments once it has been established.
Therefore, it is important to understand the basics of an IF staining protocol and to know about the variation that could be implemented. Together with an up-front literature research about cell type, antigen, and antibody, you can save time and get closer to generating a beautiful staining for your research.
Immunofluorescence main steps
Overview of the main steps of IF.