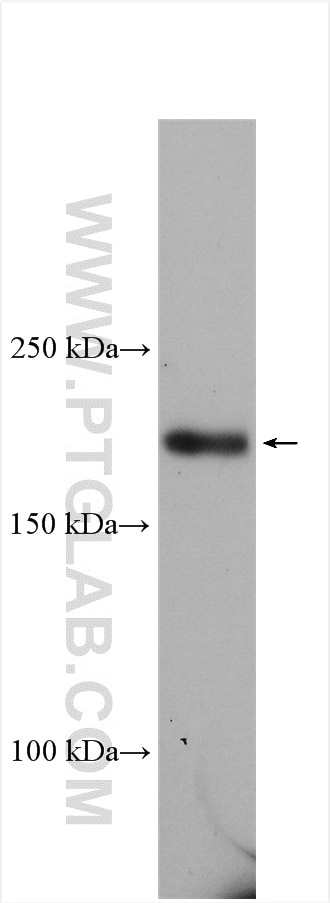
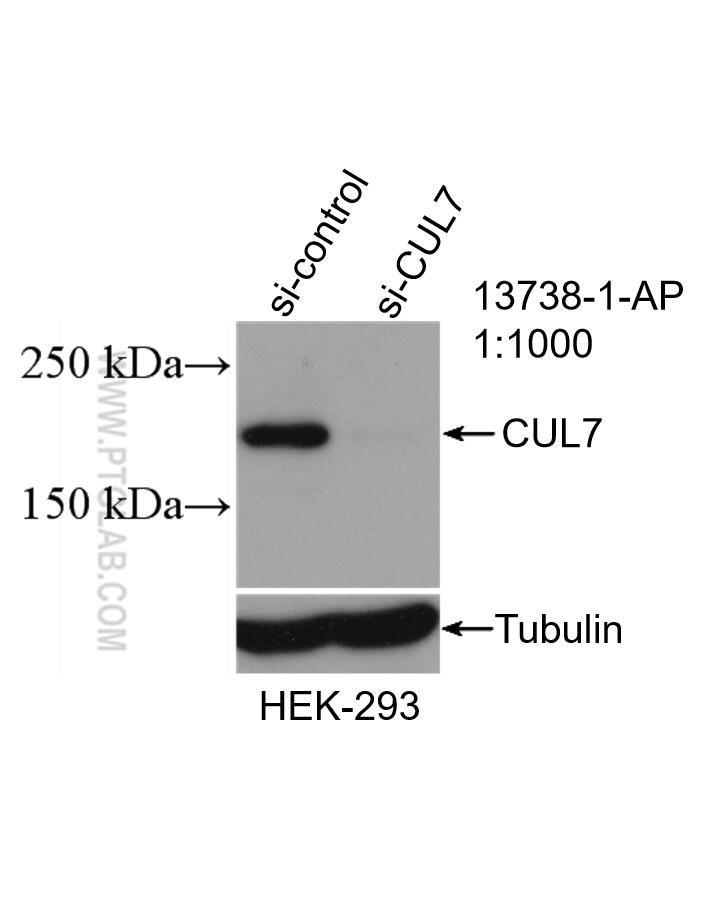
Tested Applications
| Positive WB detected in | HEK-293 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:2000 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 2 publications below |
| WB | See 5 publications below |
Product Information
13738-1-AP targets CUL7 in WB, ELISA applications and shows reactivity with human samples.
| Tested Reactivity | human |
| Cited Reactivity | human, rat |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag4675 Product name: Recombinant human CUL7 protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1348-1698 aa of BC033647 Sequence: GKEHKSEKEEEAGAAAVVDVAEGEEEEEENEDLYYEGAMPEVSVLVLSRHSWPVASICHTLNPRTCLPSYLRGTLNRYSNFYNKSQSHPALERGSQRRLQWTWLGWAELQFGNQTLHVSTVQMWLLLYLNDLKAVSVESLLAFSGLSADMLNQAIGPLTSSRGPLDLHEQKDIPGGVLKIRDGSKEPRSRWDIVRLIPPQTYLQAEGEDGQNLEKRRNLLNCLIVRILKAHGDEGLHIDQLVCLVLEAWQKGPCPPRGLVSSLGKGSACSSTDVLSCILHLLGKGTLRRHDDRPQVLSYAVPVTVMEPHTESLNPGSSGPNPPLTFHTLQIRSRGVPYASCTATQSFSTFR Predict reactive species |
| Full Name | cullin 7 |
| Calculated Molecular Weight | 1698 aa, 191 kDa |
| Observed Molecular Weight | 185 kDa |
| GenBank Accession Number | BC033647 |
| Gene Symbol | CUL7 |
| Gene ID (NCBI) | 9820 |
| RRID | AB_10640531 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q14999 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
The cullin family proteins are scaffold proteins for the Ring finger type E3 ligases, participating in the proteolysis through the ubiquitin-proteasome pathway. Humans express seven cullin proeins: CUL1-3, CUL4A, CUL4B, CUL5, and CUL7. Each cullin protein can form an E3 ligase similar to the prototype Ring-type E3 ligase Skp1-CUL1-F-box complex. The Cullin-RING-finger type E3 ligases are important regulators in early embryonic development, as highlighted by genetic studies demonstrating that knock-out of CUL1, CUL3, or CUL4A in mice results in early embryonic lethality. CUL7 was originally discovered as 185-kDa protein associated with the large T antigen of simian virus 40 (SV40). CUL7-deficient mice exhibit neonatal lethality with reduced size and vascular defects. CUL7 presumably plays a role in the DNA damage response by limiting p53 activity. CUL7 mutations have also been identified in 3-Msyndrome and the Yakuts short stature syndrome, both of which are characterized by pre- and post-natal growth retardation but with relatively normal mental and endocrine functions, suggesting that CUL7 may also be crucial for human placental development.
Protocols
| Product Specific Protocols | |
|---|---|
| WB protocol for CUL7 antibody 13738-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Adv Sci (Weinh) Targeting Methylglyoxal Metabolism to Enhance Ferroptosis Sensitivity in Tumor Therapy
| ||
Diabetes Liver kinase b1 is required for white adipose tissue growth and differentiation. | ||
J Cell Sci Cullin-3-KCTD10-mediated CEP97 degradation promotes primary cilium formation.
| ||
Am J Physiol Renal Physiol Ubiquitination of NKCC2 by the Cullin-RING E3 Ubiquitin Ligase Family in the Rat Thick Ascending Limb of the Loop of Henle | ||
Front Immunol Fbxo16 mediates degradation of NF-κB p65 subunit and inhibits inflammatory response in dendritic cells |