Tested Applications
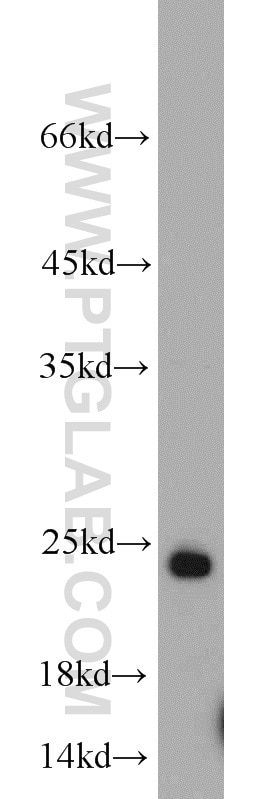
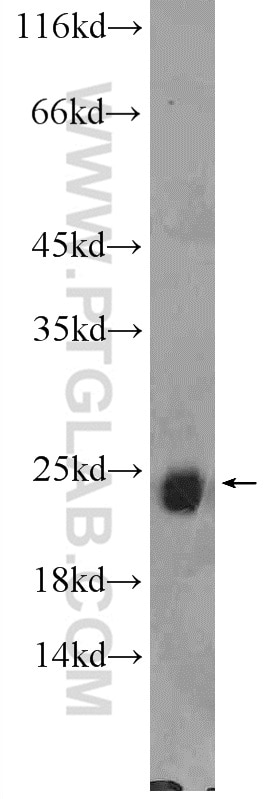
| Positive WB detected in | mouse brain tissue, C6 cells, rat brain tissue |
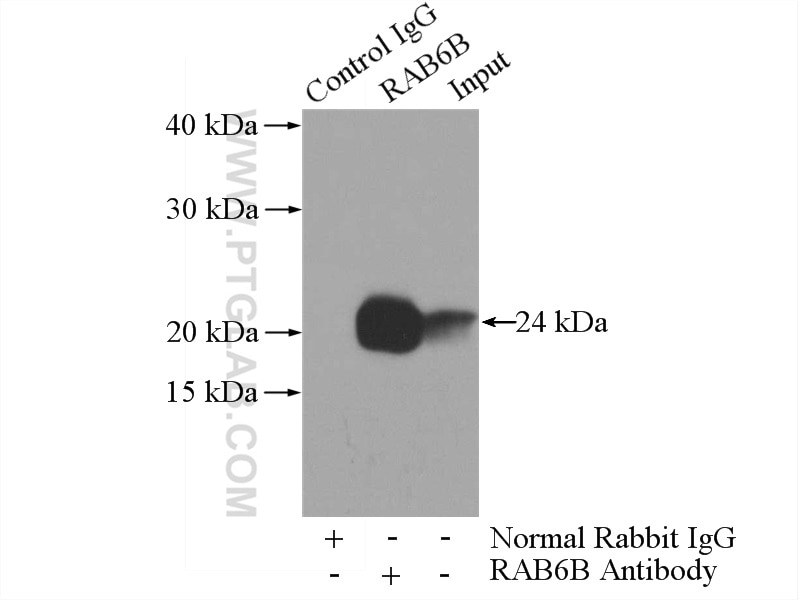
| Positive IP detected in | mouse brain tissue |
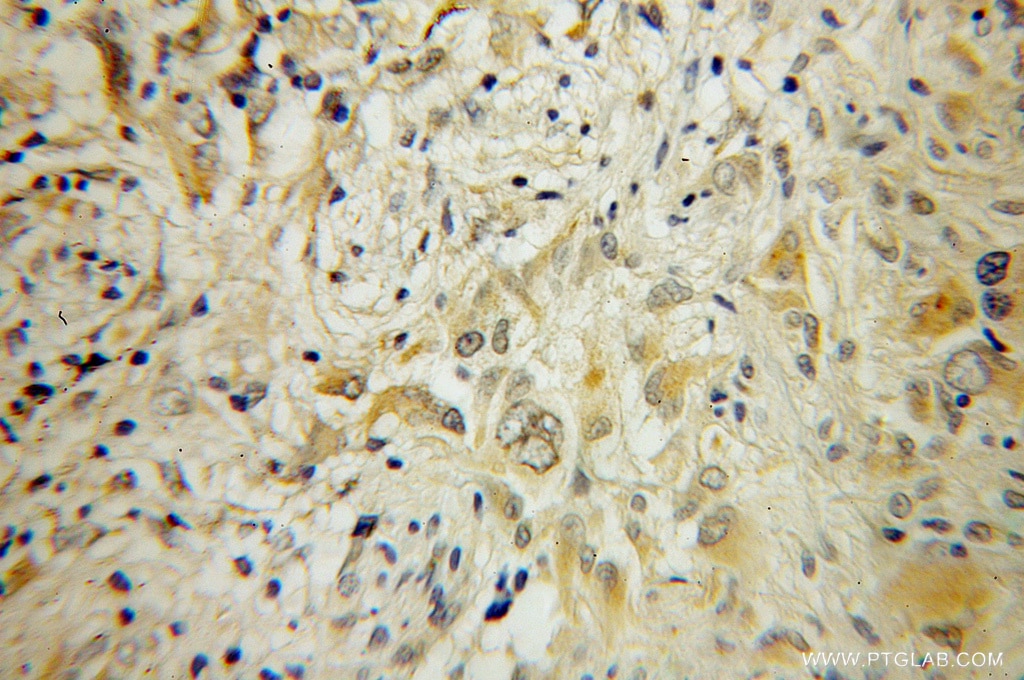
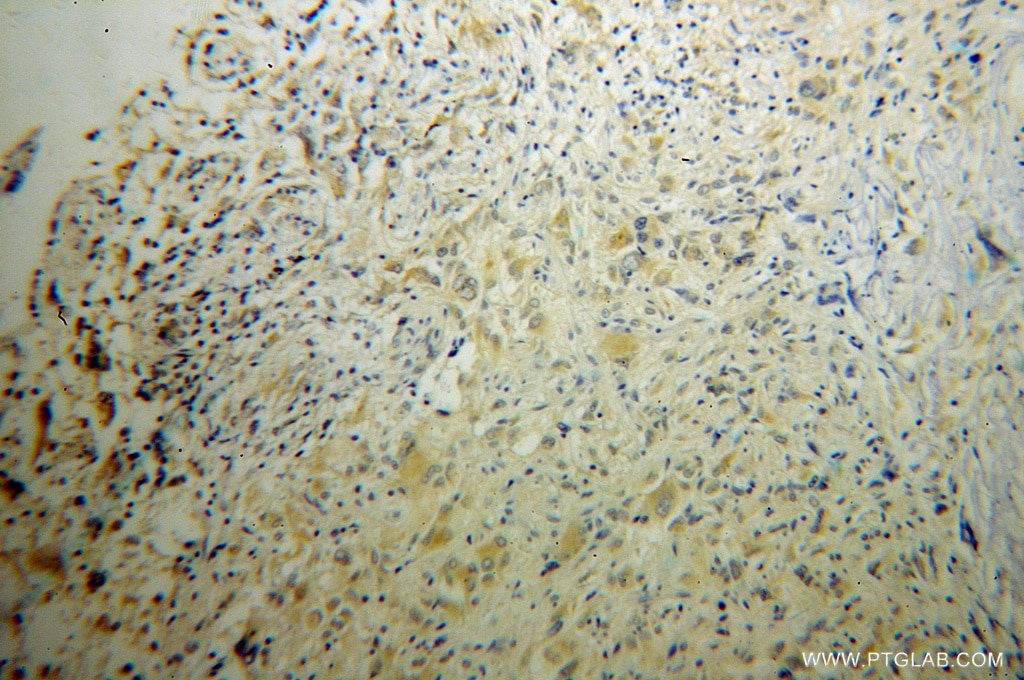
| Positive IHC detected in | human gliomas tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
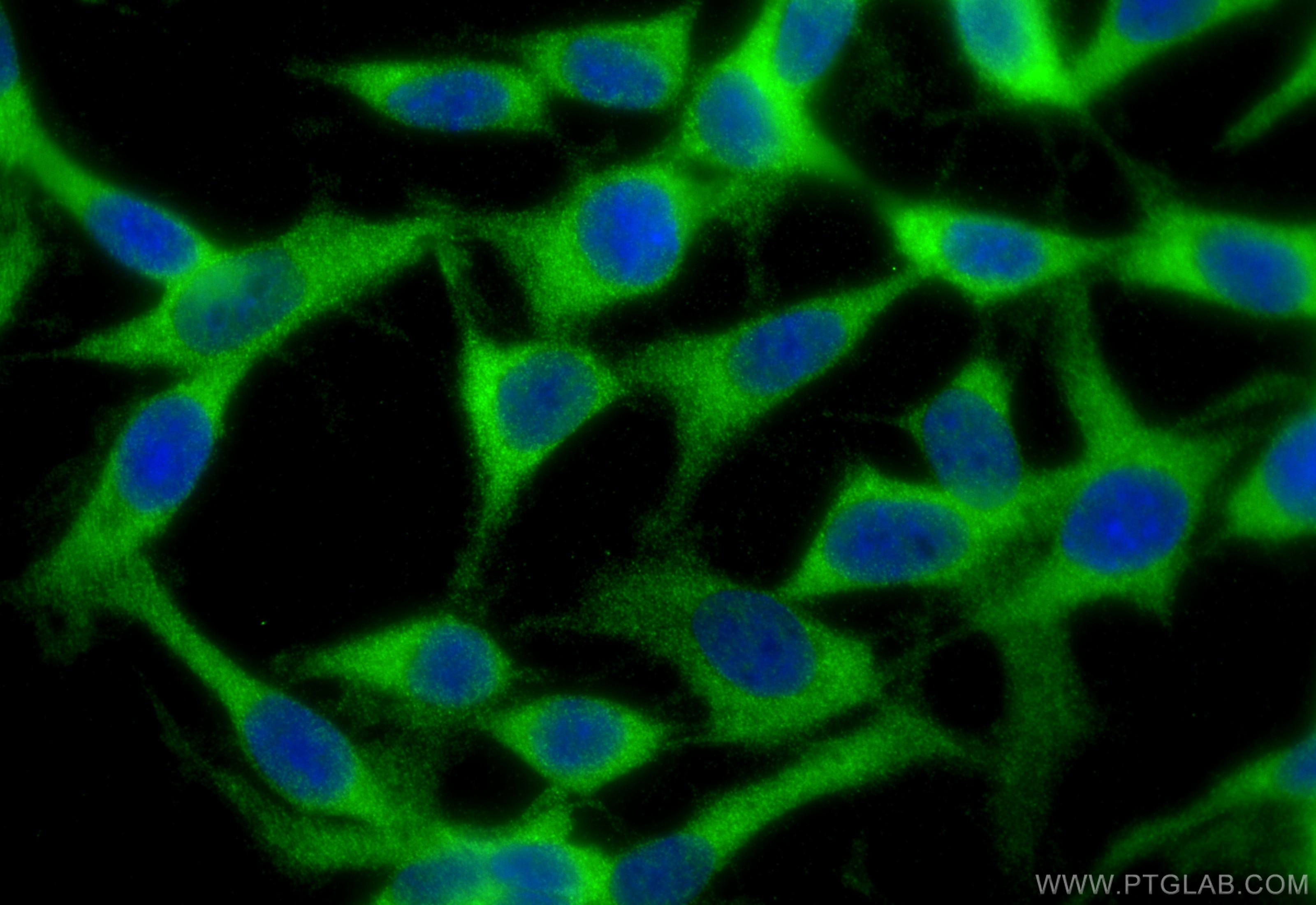
| Positive IF/ICC detected in | C6 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:2000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunohistochemistry (IHC) | IHC : 1:20-1:200 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:200-1:800 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 2 publications below |
| WB | See 4 publications below |
| IF | See 2 publications below |
Product Information
10340-1-AP targets RAB6B in WB, IHC, IF/ICC, IP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse, rat |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag0322 Product name: Recombinant human RAB6B protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 25-208 aa of BC002510 Sequence: GKTSLITRFMYDSFDNTYQATIGIDFLSKTMYLEDRTVRLQLWDTAGQERFRSLIPSYIRDSTVAVVVYDITNLNSFQQTSKWIDDVRTERGSDVIIMLVGNKTDLADKRQITIEEGEQRAKELSVMFIETSAKTGYNVKQLFRRVASALPGMENVQEKSKEGMIDIKLDKPQEPPASEGGCSC Predict reactive species |
| Full Name | RAB6B, member RAS oncogene family |
| Calculated Molecular Weight | 23 kDa |
| Observed Molecular Weight | 24 kDa |
| GenBank Accession Number | BC002510 |
| Gene Symbol | RAB6B |
| Gene ID (NCBI) | 51560 |
| RRID | AB_2175461 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q9NRW1 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
The human RAB genes share structural and biochemical properties with the Ras gene superfamily. Accumulating data suggests an important role for RAB proteins either in endocytosis or in biosynthetic protein transport. The transport of newly synthesized proteins from endoplasmic reticulum to the Golgi complex and to secretory vesicles involves the movement of carrier vesicles, a process that appears to involve RAB protein function. Rab6A has been shown to be a regulator of membrane traffic from the Golgi apparatus towards the endoplasmic reticulum (ER). Rab6B is encoded by an independent gene which is located on chromosome 3 region q21-q23. In contrast to Rab6A whose expression is ubiquitous, Rab6B is expressed in a tissue and cell-type specific manner. Rab6B is predominantly expressed in brain and the neuroblastoma cells. In brain, Rab6B was found to be specifically expressed in microglia, pericytes and Purkinje cells. Endogenous Rab6B localises to the Golgi apparatus and to ERGIC-53-positive vesicles. Comparable studies between Rab6A and Rab6B revealed distinct biochemical and cellular properties. Rab6B displays lower GTP-binding activities and is distributed over Golgi and ER membranes, whereas Rab6A is more restricted to the Golgi apparatus. Since the GTP-bound form of Rab6B does interact with all known Rab6A effectors, including Rabkinesin-6, the results suggest a cell-type specific role for Rab6B in retrograde membrane traffic at the level of the Golgi complex.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for RAB6B antibody 10340-1-AP | Download protocol |
| IHC protocol for RAB6B antibody 10340-1-AP | Download protocol |
| IP protocol for RAB6B antibody 10340-1-AP | Download protocol |
| WB protocol for RAB6B antibody 10340-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Life Sci Sex-specific molecular hallmarks point to increased atherogenesis susceptibility in male senescence-accelerated mice | ||
J Neuroimmunol Autoantibody against the Rab6A/Rab6B in primary autoimmune cerebellar ataxia associated with Sjogren's syndrome: A case report. | ||
EMBO Rep RAB6 and dynein drive post-Golgi apical transport to prevent neuronal progenitor delamination
| ||
Mol Biol Cell Movement of the endoplasmic reticulum is driven by multiple classes of vesicles marked by Rab-GTPases
|
Reviews
The reviews below have been submitted by verified Proteintech customers who received an incentive for providing their feedback.
FH Sahil (Verified Customer) (09-02-2020) | Works great as expected.
|