Tested Applications
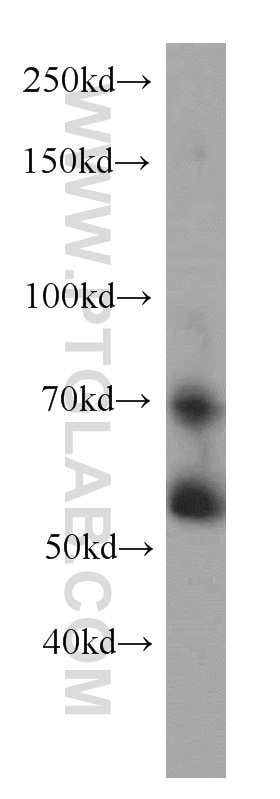
| Positive WB detected in | NIH/3T3 cells, MCF-7 cells |
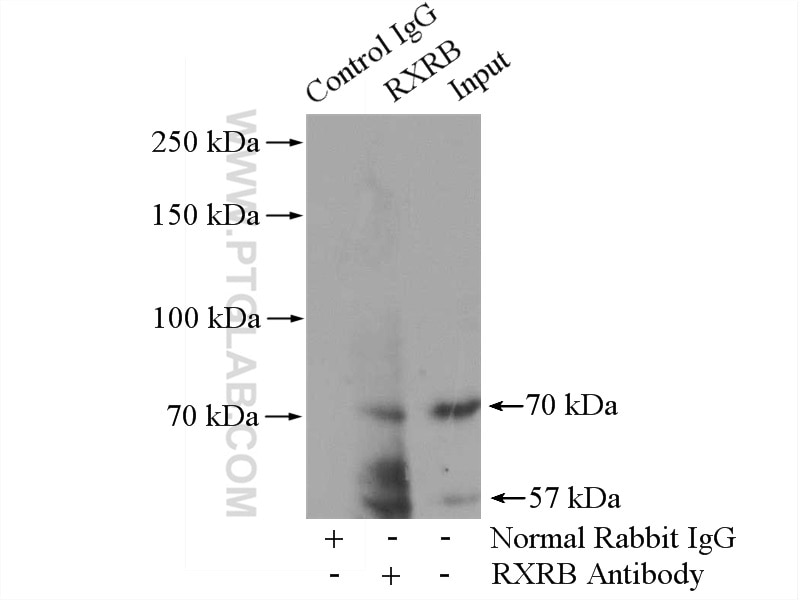
| Positive IP detected in | MCF-7 cells |
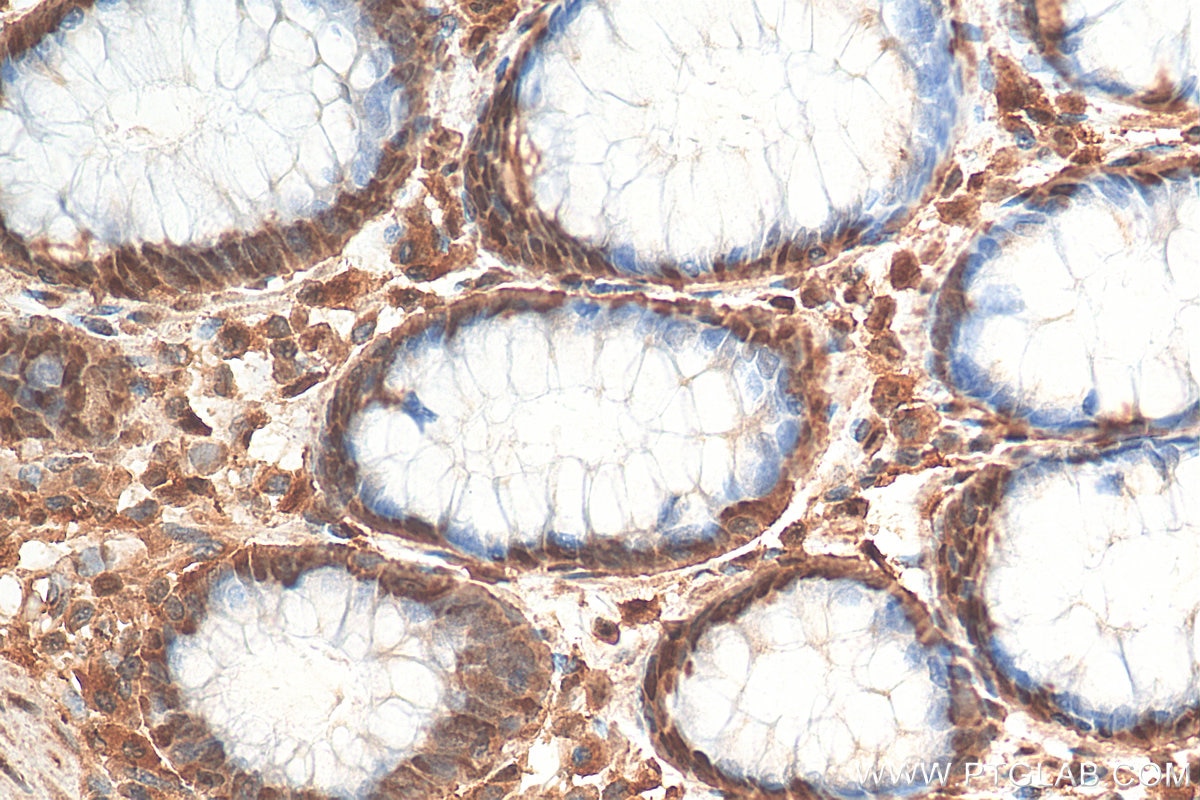
| Positive IHC detected in | human colon cancer tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:1000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunohistochemistry (IHC) | IHC : 1:50-1:500 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| WB | See 3 publications below |
| IF | See 1 publications below |
Product Information
14684-1-AP targets RXRB in WB, IHC, IF, IP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse, rat |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag0263 Product name: Recombinant human RXRB protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 70-238 aa of BC001167 Sequence: RDGMGDSGRDSRSPDSSSPNPLPQGVPPPSPPGPPLPPSTAPSLGGSGAPPPPPMPPPPLGSPFPVISSSMGSPGLPPPAPPGFSGPVSSPQINSTVSLPGGGSGPPEDVKPPVLGVRGLHCPPPPGGPGAGKRLCAICGDRSSGKHYGVYSCEGCKGFFKRTIRKDLT Predict reactive species |
| Full Name | retinoid X receptor, beta |
| Calculated Molecular Weight | 57 kDa |
| Observed Molecular Weight | 57 kDa, 70 kDa |
| GenBank Accession Number | BC001167 |
| Gene Symbol | RXRB |
| Gene ID (NCBI) | 6257 |
| RRID | AB_10858228 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | P28702 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
Retinoid X Receptor beta (RXRB) is a member of the retinoid X receptor (RXR) family of nuclear receptors which are involved in mediating the effects of retinoic acid. RXRB heterodimerizes with other nuclear hormone receptors, such as RAR, the thyroid hormone receptor and the vitamin D3 receptor, to enhance binding to their cognate response elements. (PMID: 8257090)
Protocols
| Product Specific Protocols | |
|---|---|
| IHC protocol for RXRB antibody 14684-1-AP | Download protocol |
| IP protocol for RXRB antibody 14684-1-AP | Download protocol |
| WB protocol for RXRB antibody 14684-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Autophagy Neutrophil-derived serine proteases induce FOXA2-mediated autophagy dysfunction and exacerbate colitis-associated carcinogenesis via F2RL1/protease-activated receptor 2 | ||
Am J Physiol Lung Cell Mol Physiol Inhaled vitamin A is more effective than intramuscular dosing in mitigating hyperoxia-induced lung injury in a neonatal rat model of bronchopulmonary dysplasia. | ||
Mol Neurobiol Verification of Pain-Related Neuromodulation Mechanisms of Calcitonin in Knee Osteoarthritis | ||
Exp Mol Med LncRNA Gm35585 transcriptionally activates the peroxidase EHHADH against diet-induced fatty liver |