"DDDDK tag Antibodies" Comparison
View side-by-side comparison of DDDDK tag antibodies from other vendors to find the one that best suits your research needs.
Tested Applications
| Positive WB detected in | Recombinant Protein |
| Positive IP detected in | Recombinant protein protein |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:2000-1:16000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| WB | See 10 publications below |
| IP | See 3 publications below |
Product Information
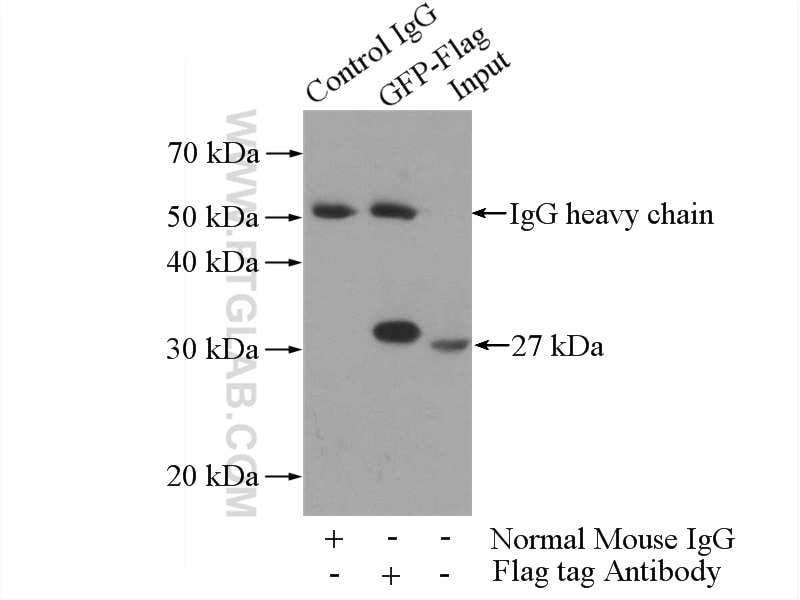
60002-1-Ig targets DDDDK tag in WB, IP, ELISA applications and shows reactivity with recombinant protein samples.
| Tested Reactivity | recombinant protein |
| Host / Isotype | Mouse / IgG2b |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
Peptide Predict reactive species |
| Full Name | DDDDK tag |
| Gene Symbol | |
| Gene ID (NCBI) | |
| RRID | AB_2881349 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | FLAGTAG |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
Protein tags are protein or peptide sequences located either on the C- or N- terminal of the target protein, which facilitates one or several of the following characteristics: solubility, detection, purification, localization and expression. The DYKDDDDK(FLAG) peptide has been used extensively as a general tag in expression vectors. This peptide can be expressed and detected with the protein of interest as an amino-terminal or carboxy-terminal fusion. N-terminal DDDDK vectors provide an Ek cleavage site for removal of the fusion tag. The DDDDK peptide is likely to be located on the surface of a fusion protein because of its hydrophilic nature. As a result, the DDDDK peptide is more likely to be accessible to antibodies. A DDDDK-tag can be used in many different assays that require recognition by an antibody, such as western blotting, immunocytochemistry, immunoprecipitation, flow cytometry, protein purification, and in the study of protein-protein interactions, cell ultrastructure, and protein localization and so on. This antibody is a mouse monoclonal antibody raised against 3xFlag (3xDYKDDDDKT) sequence and recognizes the (3x)DYKDDDDK peptide and detects DDDDK-tagged proteins.
Publications
| Species | Application | Title |
|---|---|---|
Autophagy Tumor-derived lactate promotes resistance to bevacizumab treatment by facilitating autophagy enhancer protein RUBCNL expression through histone H3 lysine 18 lactylation (H3K18la) in colorectal cancer | ||
Oncogene TRIB3 confers radiotherapy resistance in esophageal squamous cell carcinoma by stabilizing TAZ. | ||
Aging (Albany NY) HOXB9 enhances the ability of lung cancer cells to penetrate the blood-brain barrier. | ||
Sci Rep TDP-43 aggregation mirrors TDP-43 knockdown, affecting the expression levels of a common set of proteins. | ||
J Virol Enterovirus D68 Protease 2Apro Targets TRAF3 to Subvert Host Innate Immune Responses. | ||