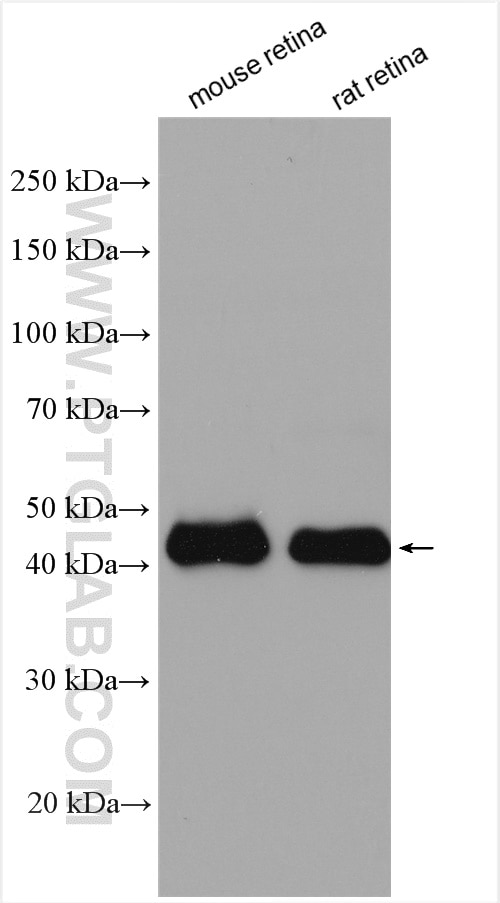
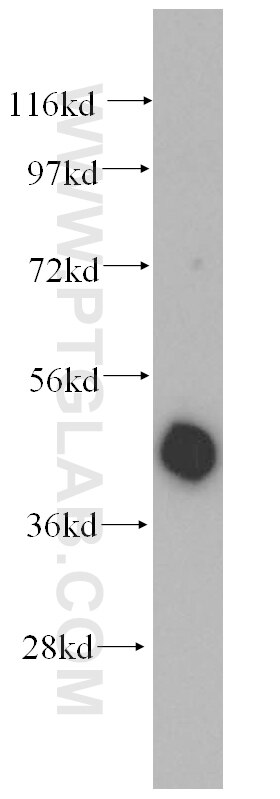
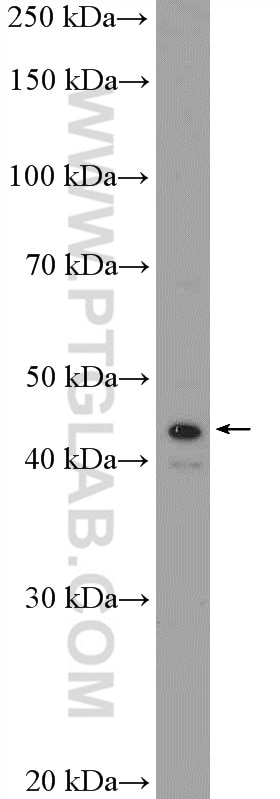
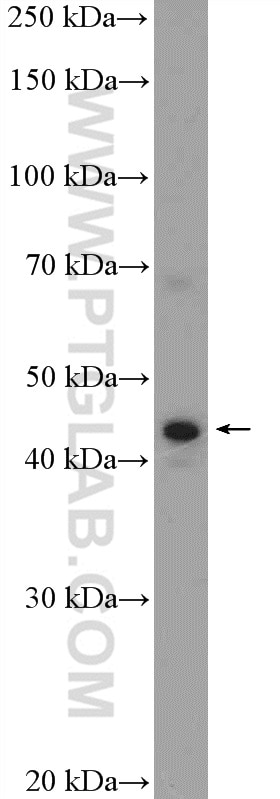
Tested Applications
| Positive WB detected in | mouse retina tissue, Y79 cells, HepG2 cells, rat retina tissue |
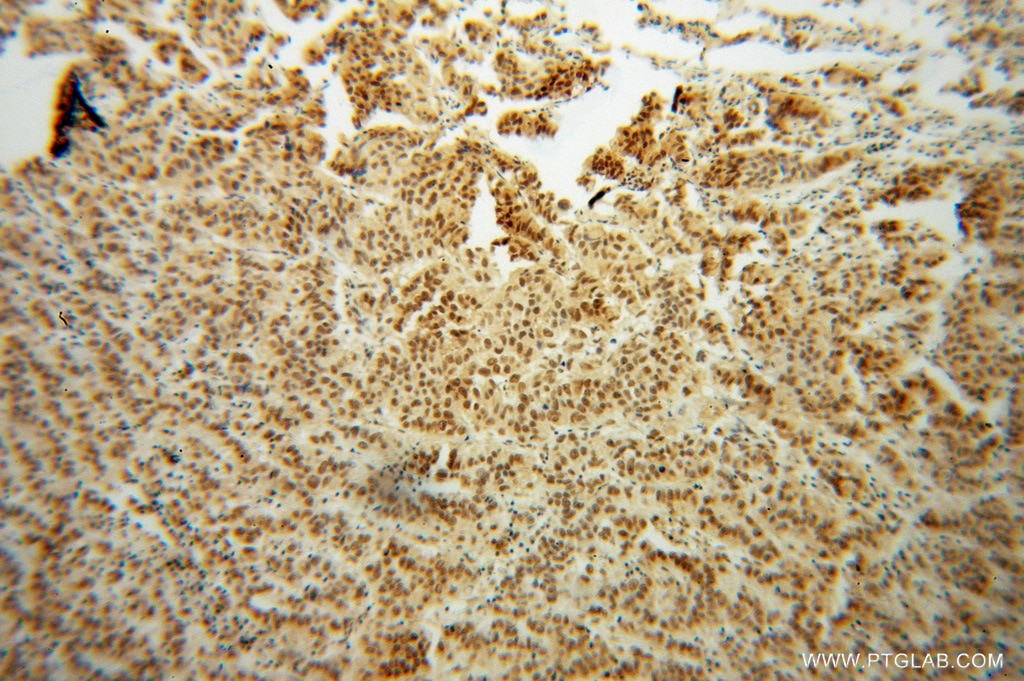
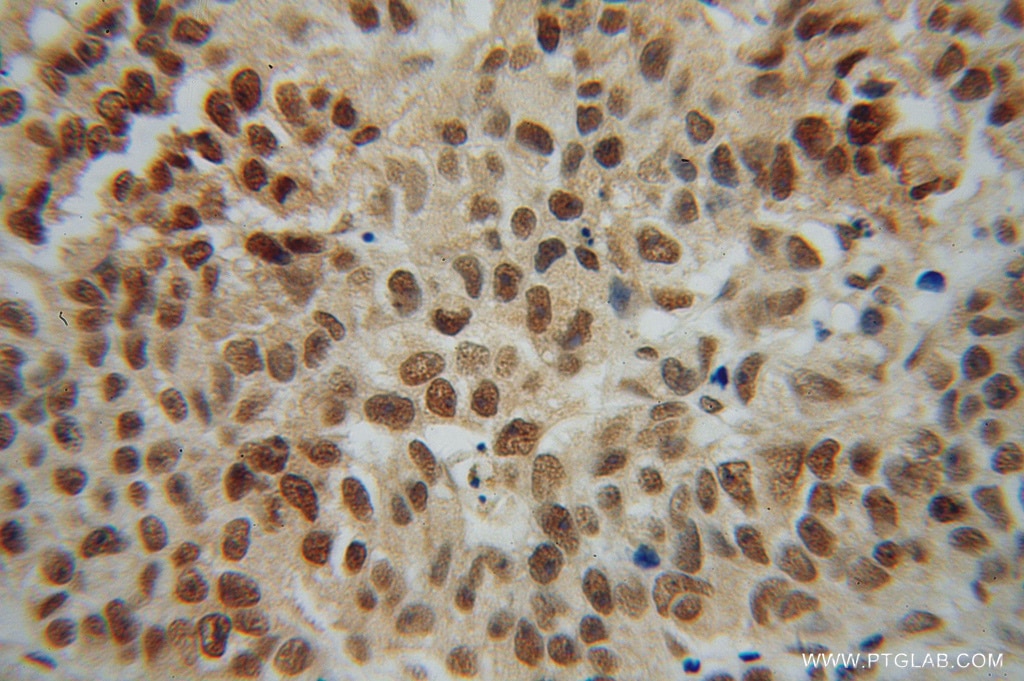
| Positive IHC detected in | human prostate cancer tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
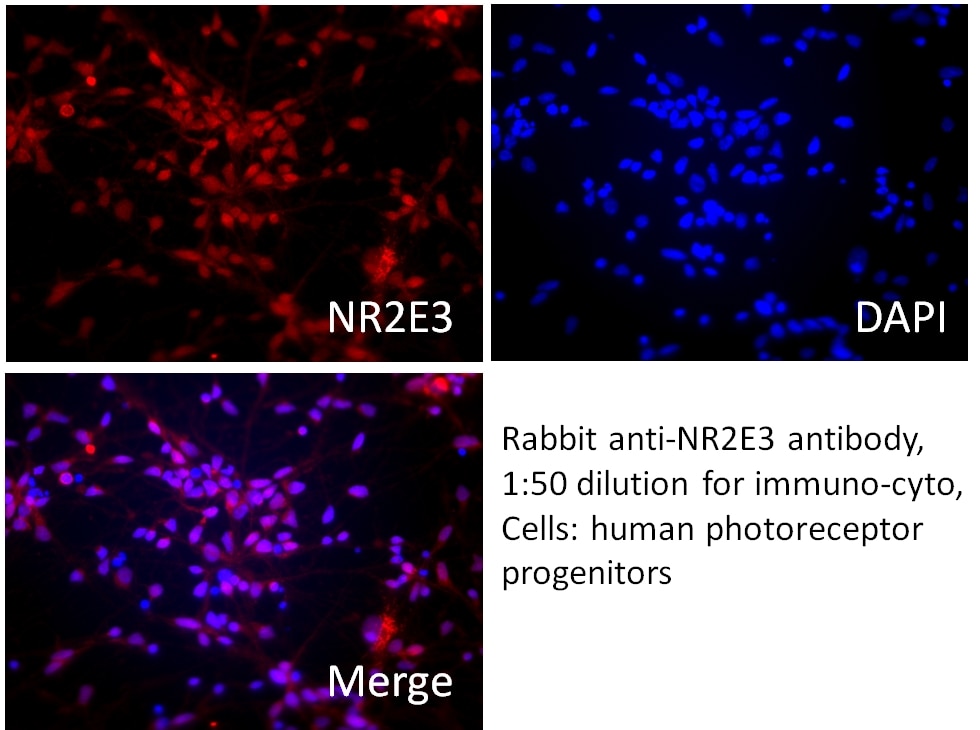
| Positive IF/ICC detected in | photoreceptor progenitor cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:2000-1:12000 |
| Immunohistochemistry (IHC) | IHC : 1:20-1:200 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:20-1:100 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 2 publications below |
| WB | See 6 publications below |
| IHC | See 3 publications below |
Product Information
14246-1-AP targets NR2E3 in WB, IHC, IF/ICC, ELISA applications and shows reactivity with human, mouse, rat, pig samples.
| Tested Reactivity | human, mouse, rat, pig |
| Cited Reactivity | human, mouse, zebrafish |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag5503 Product name: Recombinant human NR2E3 protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1-322 aa of BC041421 Sequence: MCPVDKAHRNQCQACRLKKCLQAGMNQDAVQNERQPRSTAQVHLDSMESNTESRPESLVAPPAPAGRSPRGPTPMSAARALGHHFMASLITAETCAKLEPEDADENIDVTSNDPEFPSSPYSSSSPCGLDSIHETSARLLFMAVKWAKNLPVFSSLPFRDQVILLEEAWSELFLLGAIQWSLPLDSCPLLAPPEASAAGGAQGRLTLASMETRVLQETISRFRALAVDPTEFACMKALVLFKPETRGLKDPEHVEALQDQSQVMLSQHSKAHHPSQPVRFGKLLLLLPSLRFITAERIELLFFRKTIGNTPMEKLLCDMFKN Predict reactive species |
| Full Name | nuclear receptor subfamily 2, group E, member 3 |
| Calculated Molecular Weight | 45 kDa |
| Observed Molecular Weight | 43-45 kDa |
| GenBank Accession Number | BC041421 |
| Gene Symbol | NR2E3 |
| Gene ID (NCBI) | 10002 |
| RRID | AB_2155476 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q9Y5X4 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
NR2E3, also known as PNR, encodes a retinal nuclear receptor that is a ligand-dependent transcription factor. This protein is part of a large family of nuclear receptor transcription factors involved in signaling pathways. NR2E3 influences the development of photoreceptors and their differentiation into rod and cone types, and acts as a ranscriptional factor that is an activator of rod development and repressor of cone development [PMID:20725840]. It binds the promoter region of a number of rod- and cone-specific genes, including rhodopsin, M- and S-opsin and rod-specific phosphodiesterase beta subunit. [PMID:15689355]
Protocols
| Product Specific Protocols | |
|---|---|
| IHC protocol for NR2E3 antibody 14246-1-AP | Download protocol |
| WB protocol for NR2E3 antibody 14246-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
PLoS Genet Distinct and Atypical Intrinsic and Extrinsic Cell Death Pathways between Photoreceptor Cell Types upon Specific Ablation of Ranbp2 in Cone Photoreceptors. | ||
J Biol Chem Differential Loss of Prolyl-Isomerase or Chaperone Activity of Ran-binding protein 2 (Ranbp2) Unveils Distinct Physiological Roles of Its Cyclophilin Domain in Proteostasis. | ||
Sci Rep Loss of NR2E3 represses AHR by LSD1 reprogramming, is associated with poor prognosis in liver cancer. | ||
Sci Rep Proteomics identifies multiple retinitis pigmentosa associated proteins involved in retinal degeneration in a mouse model bearing a Pde6b mutation | ||
Int Ophthalmol Low expression of NR1D1 and NR2E3 is associated with advanced features of retinoblastoma | ||
Adv Sci (Weinh) The Loss of an Orphan Nuclear Receptor NR2E3 Augments Wnt/β-catenin Signaling via Epigenetic Dysregulation that Enhances Sp1-β catenin-p300 Interactions in Hepatocellular Carcinoma
|