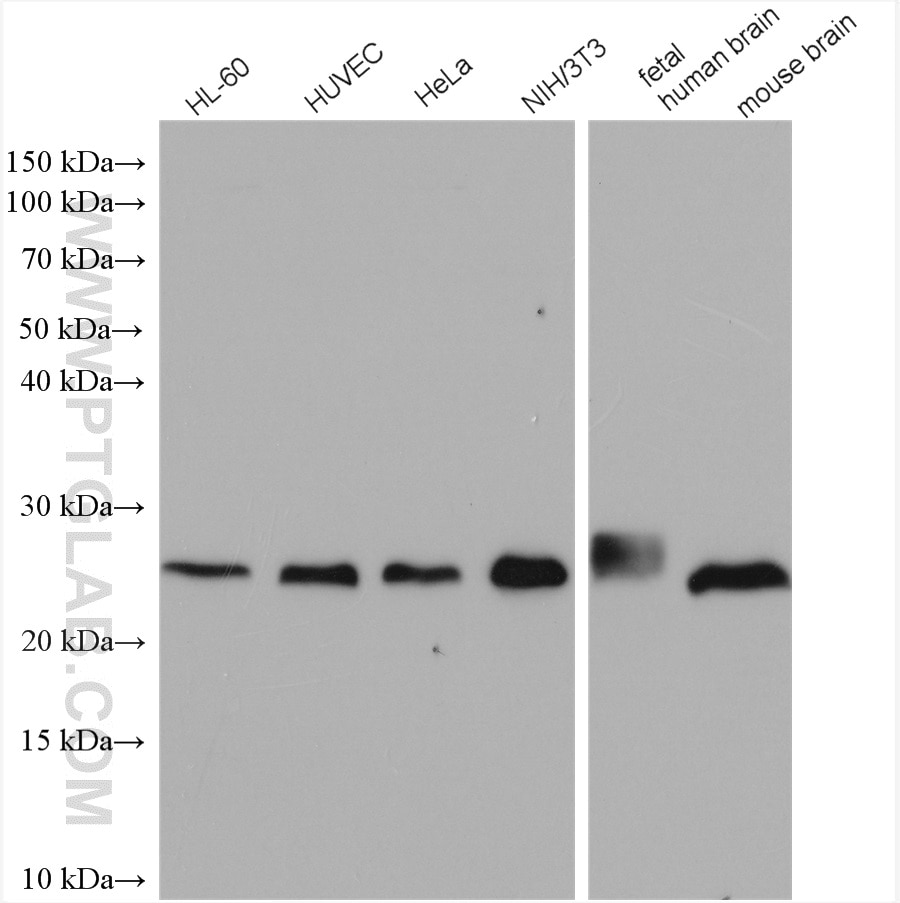
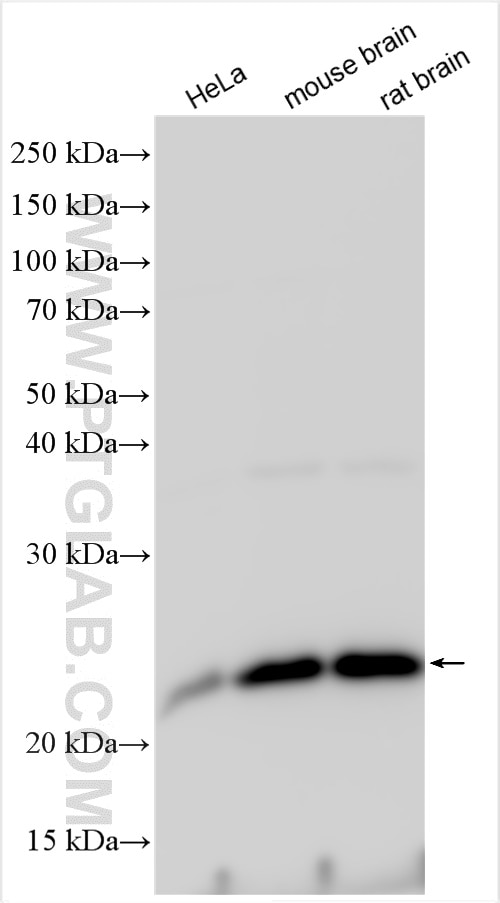
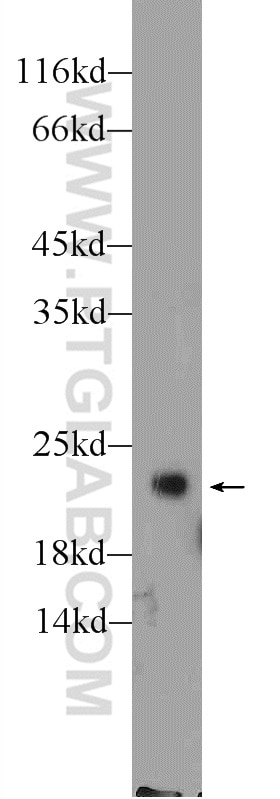
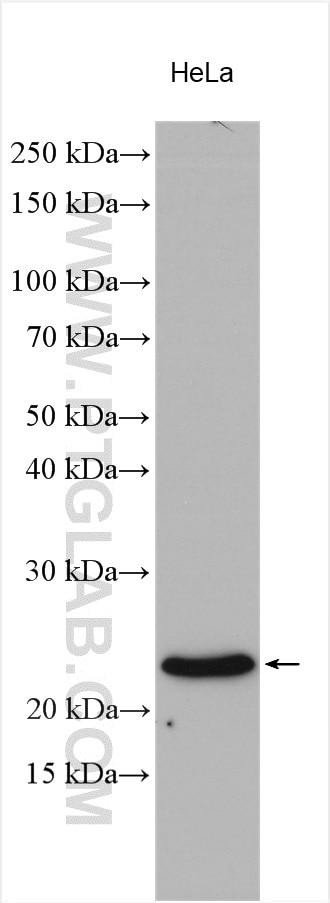
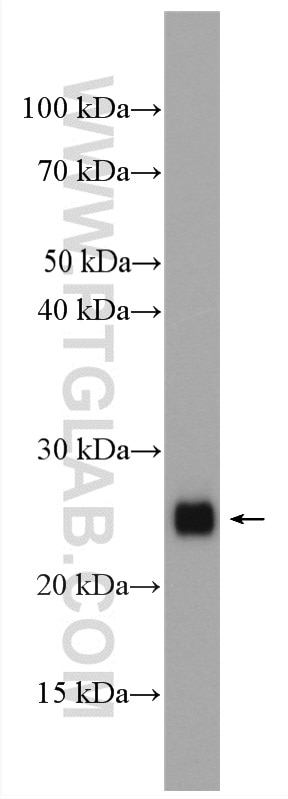
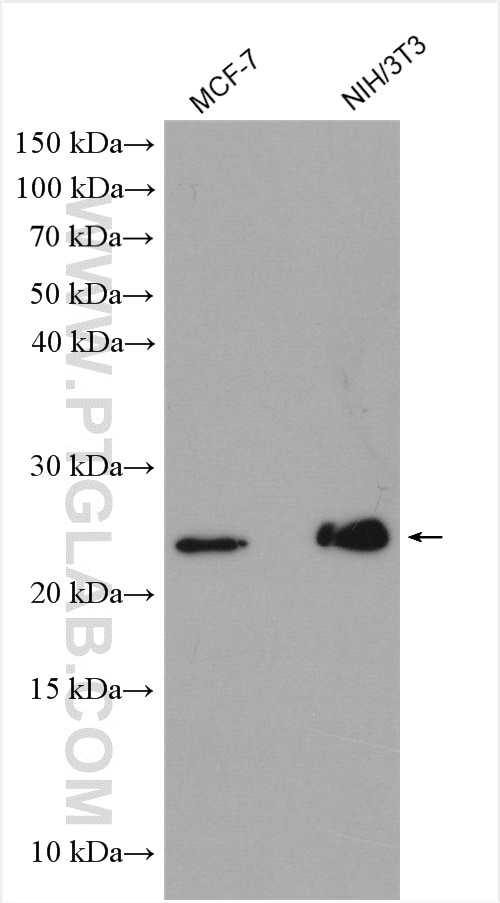
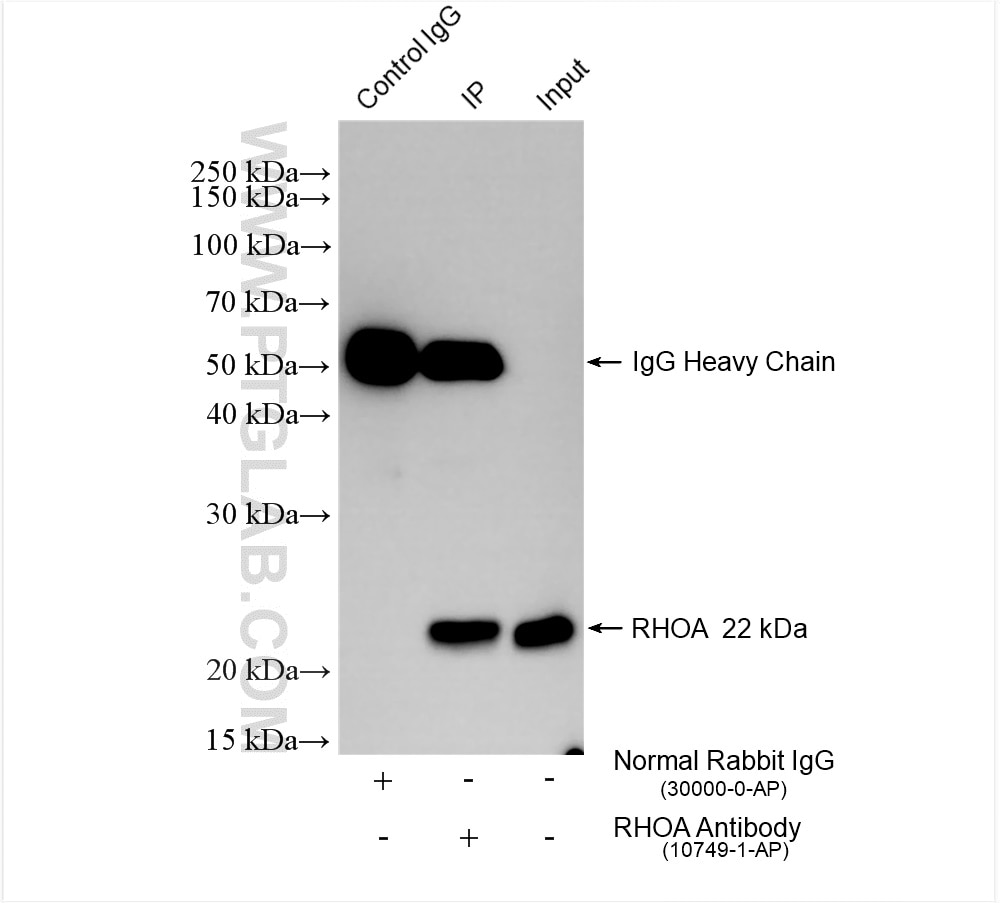
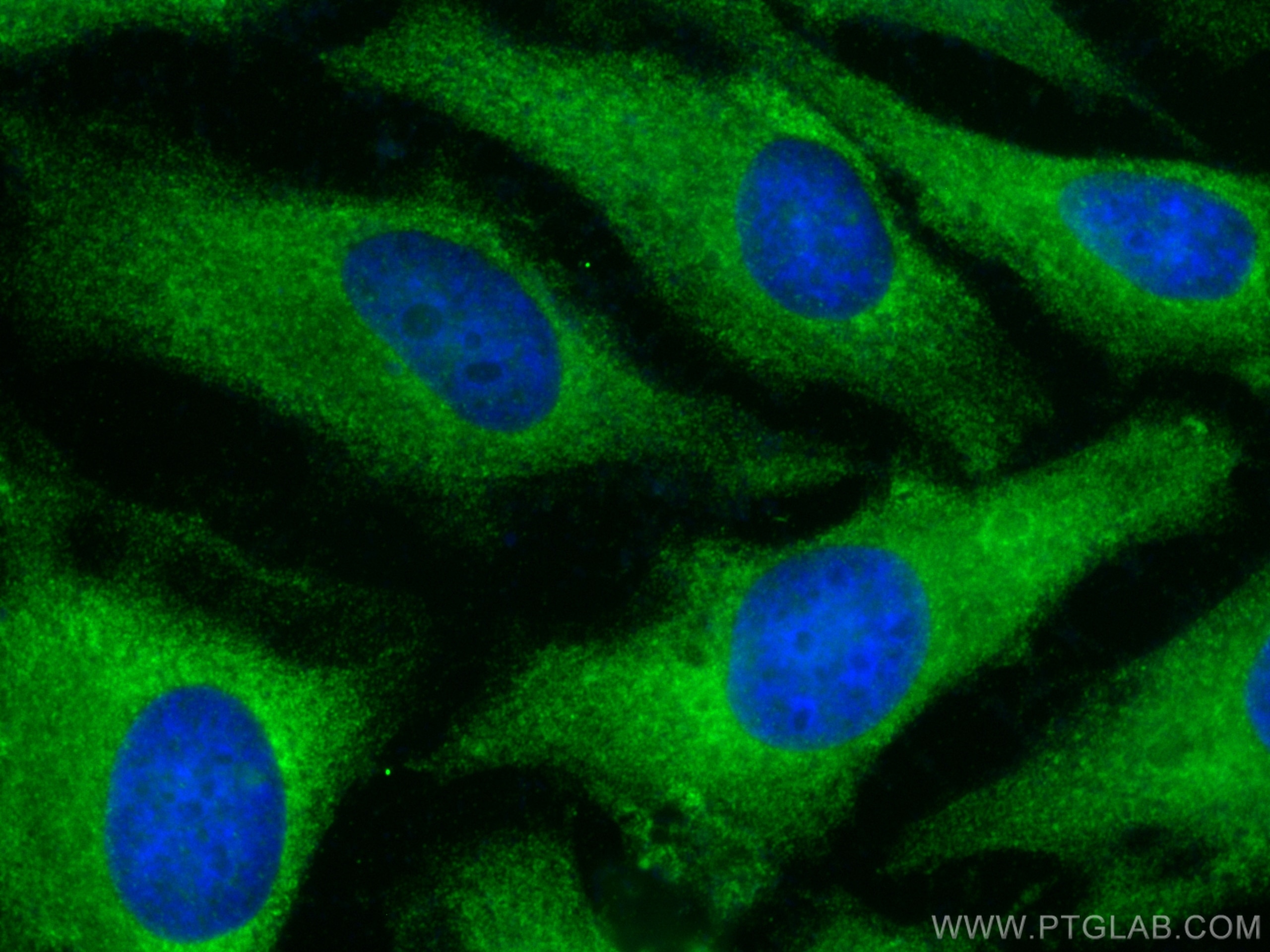
Tested Applications
| Positive WB detected in | HeLa cells, MCF-7 cells, rat brain tissue, PC-12 cells, HL-60 cells, mouse brain tissue, HUVEC cells, NIH/3T3 cells, fetal human brain tissue |
| Positive IP detected in | mouse brain tissue |
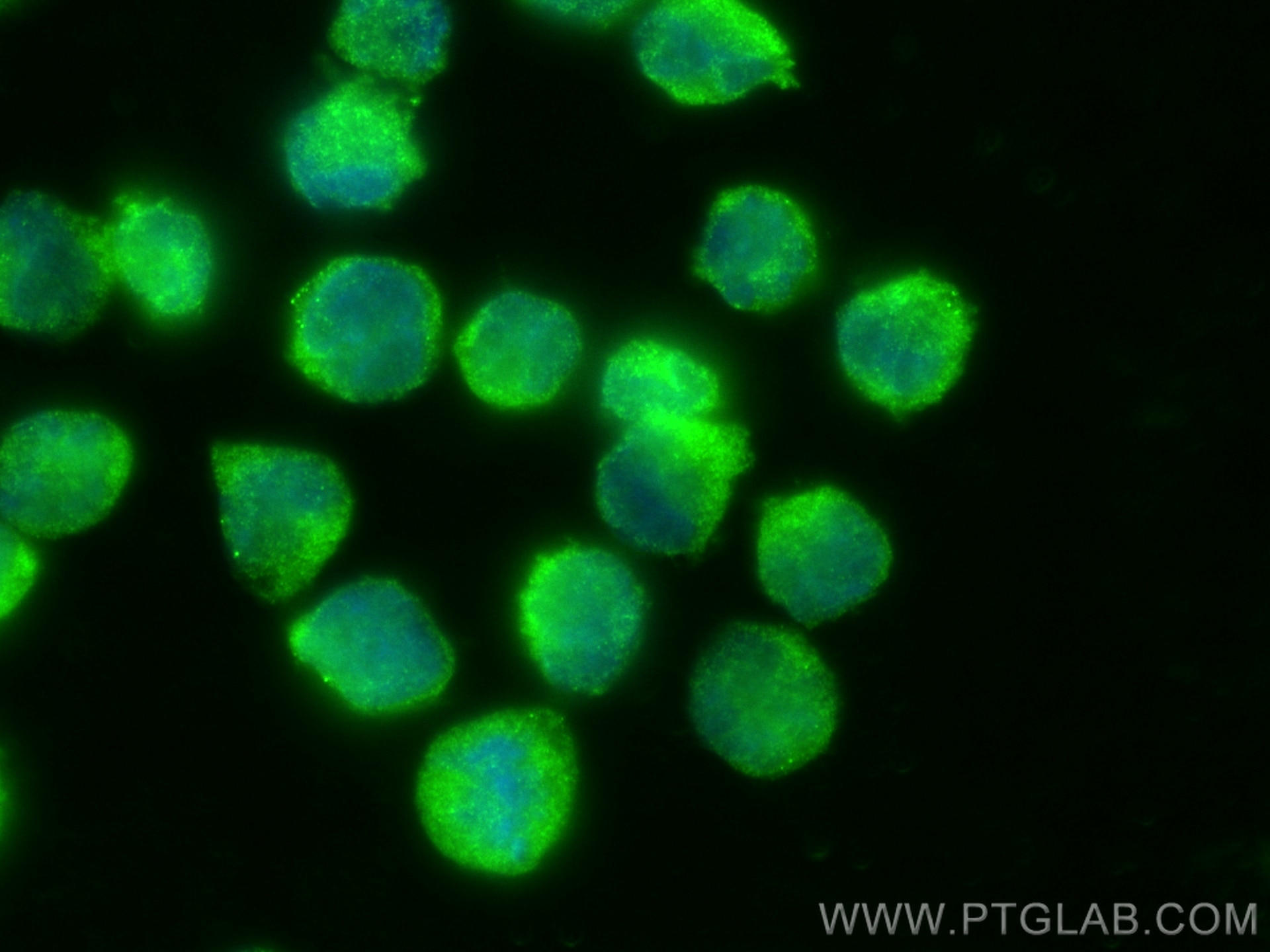
| Positive IF/ICC detected in | HeLa cells, K-562 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:1000-1:8000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:200-1:800 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 5 publications below |
| WB | See 181 publications below |
| IHC | See 17 publications below |
| IF | See 24 publications below |
| IP | See 3 publications below |
| CoIP | See 3 publications below |
Product Information
10749-1-AP targets RHOA in WB, IHC, IF/ICC, IP, CoIP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse, rat, pig, canine |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag1141 Product name: Recombinant human RHOA protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1-193 aa of BC005976 Sequence: MAAIRKKLVIVGDGACGKTCLLIVFSKDQFPEVYVPTVFENYVADIEVDGKQVELALWDTAGQEDYDRLRPLSYPDTDVILMCFSIDSPDSLENIPEKWTPEVKHFCPNVPIILVGNKKDLRNDEHTRRELAKMKQEPVKPEEGRDMANRIGAFGYMECSAKTKDGVREVFEMATRAALQARRGKKKSGCLVL Predict reactive species |
| Full Name | ras homolog gene family, member A |
| Calculated Molecular Weight | 22 kDa |
| Observed Molecular Weight | 22 kDa |
| GenBank Accession Number | BC005976 |
| Gene Symbol | RHOA |
| Gene ID (NCBI) | 387 |
| RRID | AB_2285104 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | P61586 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
What is the function of RhoA?
Ras homolog gene family, member A (RhoA) is a small GTPase that is involved in cytoskeleton organization.1 It is important in actomyosin contractility, as when RhoA binds to GTP it initiates a cascade of events mediating stress fiber or contractile ring formation, where actin bundles crosslink with other proteins. It is also associated with actin polymerization, making RhoA important in membrane ruffling and cell motility.2 The role of RhoA is essential in cell migration and adhesion.
What is the cellular localization of RhoA?
In motile cells, a leading edge stretches ahead, stabilizes, the cytoskeleton is reorganized, and the tail of the cell retracts. The role of RhoA in contracting actomyosin has linked RhoA with the tail retraction in this process, but studies into the spatiotemporal kinetics of GTPases have revealed that RhoA is also located at the leading edge of migrating cells.3 This highlights the dual roles of this protein in actin contractility and polymerization.
What is the role of RhoA in disease?
Though it is not an oncogene, the role of RhoA in adhesion and migration has implicated this protein in cancer biology. Expression of RhoA has been found to be higher in malignant tumors compared to benign tumors or in non-tumor tissue, and more invasive tumors have been found to overexpress RhoA.4 This protein is also implicated in neurodegenerative diseases and aging due to its role in axon guidance, with abnormal localization of RhoA associated with Alzheimer's and Parkinson's pathology.5,6
1. Ridley, A. J. & Hall, A. The small GTP-binding protein rho regulates the assembly of focal adhesions and actin stress fibers in response to growth factors. Cell 70, 389-399 (1992).
2. 21. Kurokawa, K. & Matsuda, M. Localized RhoA Activation as a Requirement for the Induction of Membrane Ruffling. Mol. Biol. Cell 16, 4294-4303 (2005).
3. 22. Struckhoff, A. P., Rana, M. K. & Worthylake, R. A. RhoA can lead the way in tumor cell invasion and metastasis. Front. Biosci. (Landmark Ed. 16, 1915-26 (2011).
4. 23. Mcglasson, S. et al. Rare variants of the 3'-5' DNA exonuclease in early onset TREX1 small vessel stroke [version 1; referees: awaiting peer review]. doi:10.12688/wellcomeopenres.12631.1
5. 24. Huesa, G. et al. Altered Distribution of RhoA in Alzheimer's Disease and AβPP Overexpressing Mice. J. Alzheimer's Dis. 19, 37-56 (2010).
6. 25. Hynds, D. L. Subcellular localization of Rho GTPases: implications for axon regeneration. Neural Regen. Res. 10, 1032-3 (2015).
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for RHOA antibody 10749-1-AP | Download protocol |
| IP protocol for RHOA antibody 10749-1-AP | Download protocol |
| WB protocol for RHOA antibody 10749-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Cancer Cell Targeting the mevalonate pathway suppresses ARID1A-inactivated cancers by promoting pyroptosis | ||
Cell Death Differ Endothelial cells-derived SEMA3G suppresses glioblastoma stem cells by inducing c-Myc degradation | ||
Nat Commun A mosquito salivary protein promotes flavivirus transmission by activation of autophagy. | ||
Circ. Res. Inhibition of KLF5-Myo9b-RhoA Pathway-Mediated Podosome Formation in Macrophages Ameliorates Abdominal Aortic Aneurysm. | ||
Biomater Res Near-Infrared Light-Controlled Dynamic Hydrogel for Modulating Mechanosensitive Ion Channels in 3-Dimensional Environment | ||
Acta Biomater Adipose-derived stem cells with miR-150-5p inhibition laden in hydroxyapatite/tricalcium phosphate ceramic powders promote osteogenesis via regulating Notch3 and activating FAK/ERK and RhoA |