Tested Applications
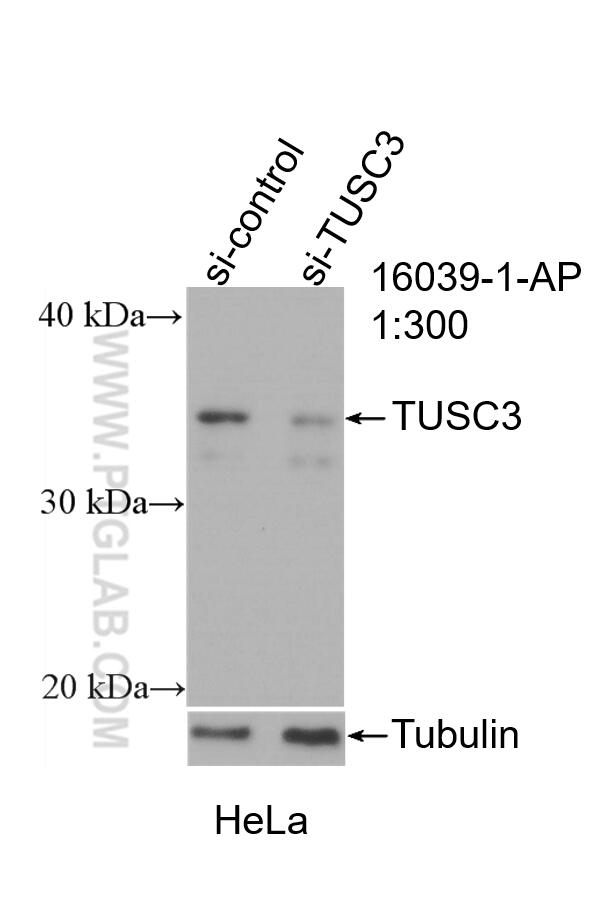
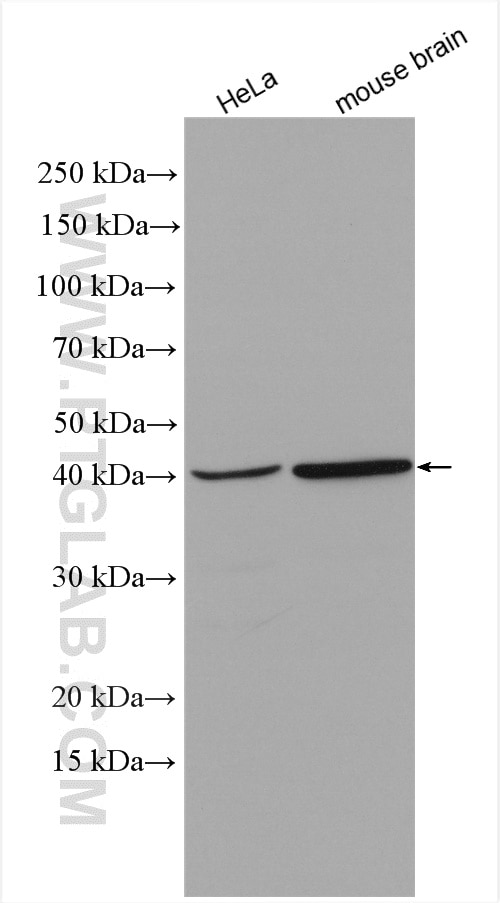
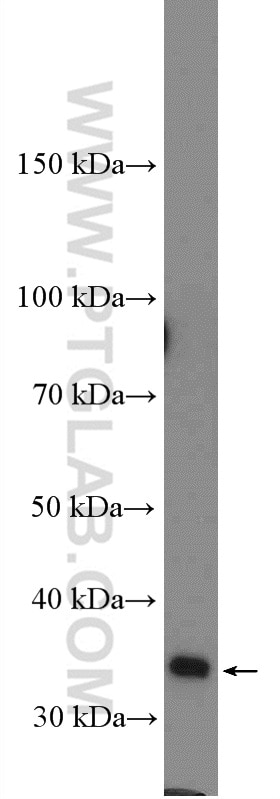
| Positive WB detected in | HeLa cells, rat testis tissue, mouse brain tissue |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:3000 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 4 publications below |
| WB | See 7 publications below |
| IHC | See 2 publications below |
| IP | See 1 publications below |
Product Information
16039-1-AP targets TUSC3 in WB, IHC, IP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag8912 Product name: Recombinant human TUSC3 protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 45-199 aa of BC010370 Sequence: KENLLAEKVEQLMEWSSRRSIFRMNGDKFRKFIKAPPRNYSMIVMFTALQPQRQCSVCRQANEEYQILANSWRYSSAFCNKLFFSMVDYDEGTDVFQQLNMNSAPTFMHFPPKGRPKRADTFDLQRIGFAAEQLAKWIADRTDVHIRVFRPPNYS Predict reactive species |
| Full Name | tumor suppressor candidate 3 |
| Calculated Molecular Weight | 347 aa, 40 kDa |
| Observed Molecular Weight | 35-40 kDa |
| GenBank Accession Number | BC010370 |
| Gene Symbol | TUSC3 |
| Gene ID (NCBI) | 7991 |
| RRID | AB_2878208 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q13454 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
TUSC3 (tumor suppressor candidate 3), originally named N33, is a potential tumor supressor gene. Decreased expression of TUSC3 has been found in various cancers, including prostate cancer, pancreas cancer and ovary cancer. TUSC3 also known as OST3A, is identified as a part of the oligosaccharyl-transferase (OST) complex and plays a crucial role in protein N-glycosylation. TUSC3 mutations have been found in families with non-syndromic autosomal recessive mental retardation.
Protocols
| Product Specific Protocols | |
|---|---|
| WB protocol for TUSC3 antibody 16039-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Nat Commun miRNA-mediated TUSC3 deficiency enhances UPR and ERAD to promote metastatic potential of NSCLC. | ||
J Cell Biol Oxidoreductase activity is necessary for N-glycosylation of cysteine-proximal acceptor sites in glycoproteins.
| ||
Sci Rep Mammalian cells lacking either the cotranslational or posttranslocational oligosaccharyltransferase complex display substrate-dependent defects in asparagine linked glycosylation.
| ||
J Biol Chem ER entry pathway and glycosylation of GPI-anchored proteins are determined by N-terminal signal sequence and C-terminal GPI-attachment sequence | ||
J Transl Med Downregulation of TUSC3 promotes EMT and hepatocellular carcinoma progression through LIPC/AKT axis
| ||
J Allergy Clin Immunol Epigenetic activation of the TUSC3 gene as a potential therapy for XMEN disease |