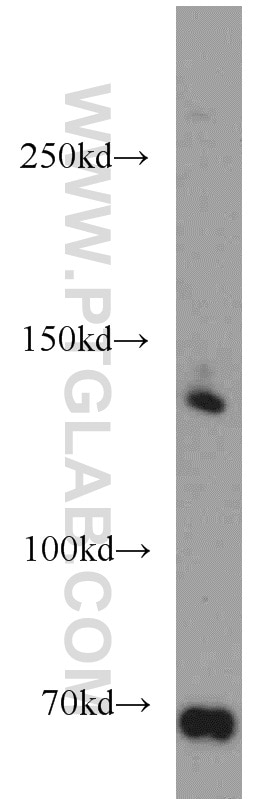
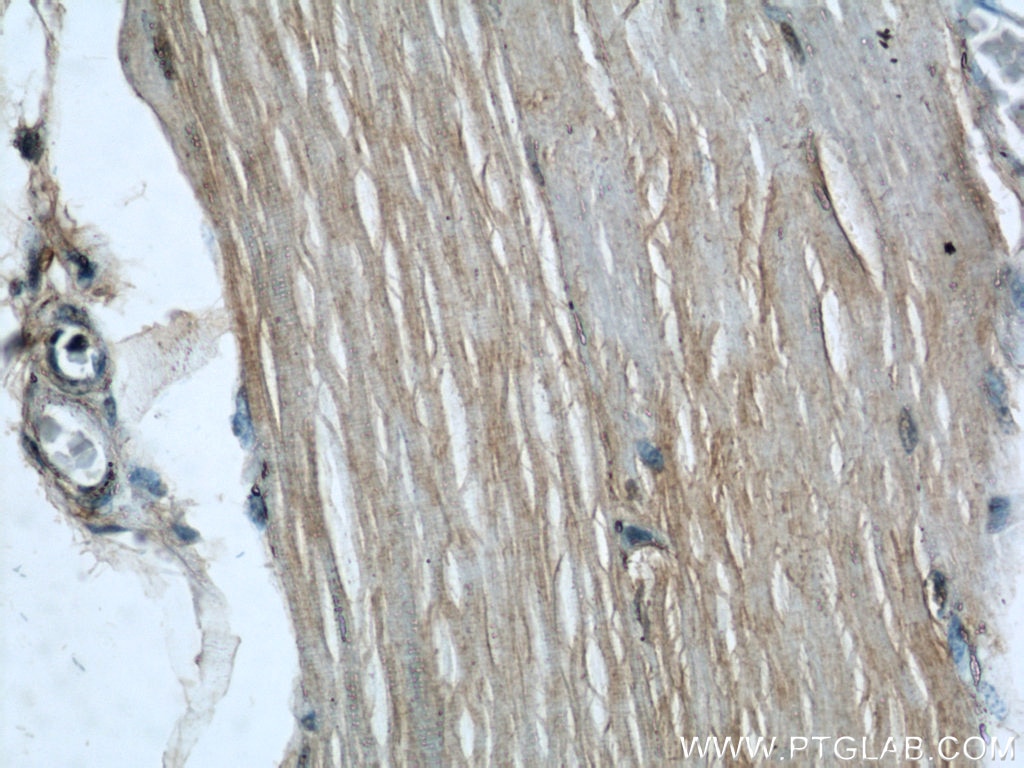
Tested Applications
| Positive WB detected in | HeLa cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:2000 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 5 publications below |
| WB | See 11 publications below |
| IHC | See 1 publications below |
| IP | See 1 publications below |
Product Information
20988-1-AP targets Hamartin in WB, IHC, IP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
Peptide Predict reactive species |
| Full Name | tuberous sclerosis 1 |
| Calculated Molecular Weight | 130 kDa |
| Observed Molecular Weight | |
| GenBank Accession Number | NM_000368 |
| Gene Symbol | Hamartin/TSC1 |
| Gene ID (NCBI) | 7248 |
| RRID | AB_10950398 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q92574 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
TSC1, also named as KIAA0243 and TSC, is implicated as a tumor suppressor. It is involved in microtubule-mediated protein transport, but this seems to be due to unregulated mTOR signaling. In complex with TSC2, TSC1 inhibits the nutrient-mediated or growth factor-stimulated phosphorylation of S6K1 and EIF4EBP1 by negatively regulating mTORC1 signaling. It seems not to be required for TSC2 GAP activity towards RHEB. It is involved in microtubule-mediated protein transport, but this seems to be due to unregulated mTOR signaling. Defects in TSC1 are the cause of tuberous sclerosis complex (TSC). Defects in TSC1 may be a cause of focal cortical dysplasia of Taylor balloon cell type (FCDBC). The antibody is specific to TSC1. This antibody can recognize two isoforms: 70 kDa (667aa) and 130 kDa.
Publications
| Species | Application | Title |
|---|---|---|
Sci Adv Synthetic lethality by targeting the RUVBL1/2-TTT complex in mTORC1-hyperactive cancer cells. | ||
Theranostics Combination therapy with ropivacaine-loaded liposomes and nutrient deprivation for simultaneous cancer therapy and cancer pain relief.
| ||
Mol Ther Nucleic Acids miR-301a Suppression within Fibroblasts Limits the Progression of Fibrosis through the TSC1/mTOR Pathway.
| ||
Sci Rep Tsc1 deficiency impairs mammary development in mice by suppression of AKT, nuclear ERα, and cell-cycle-driving proteins.
| ||
Biochem Biophys Res Commun UBE2S exerts oncogenic activities in urinary bladder cancer by ubiquitinating TSC1.
|