Tested Applications
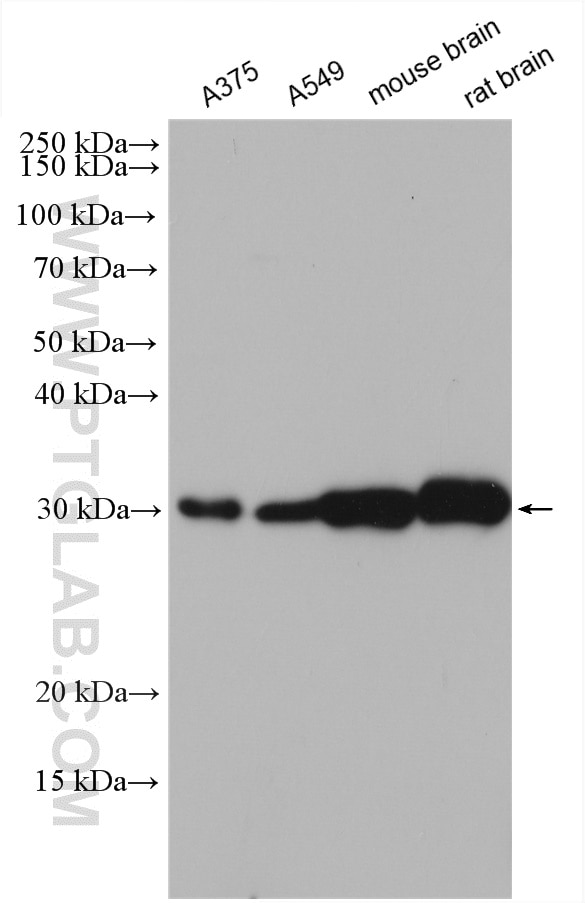
| Positive WB detected in | A375 cells, A549 cells, SH-SY5Y cells, mouse brain tissue, rat brain tissue |
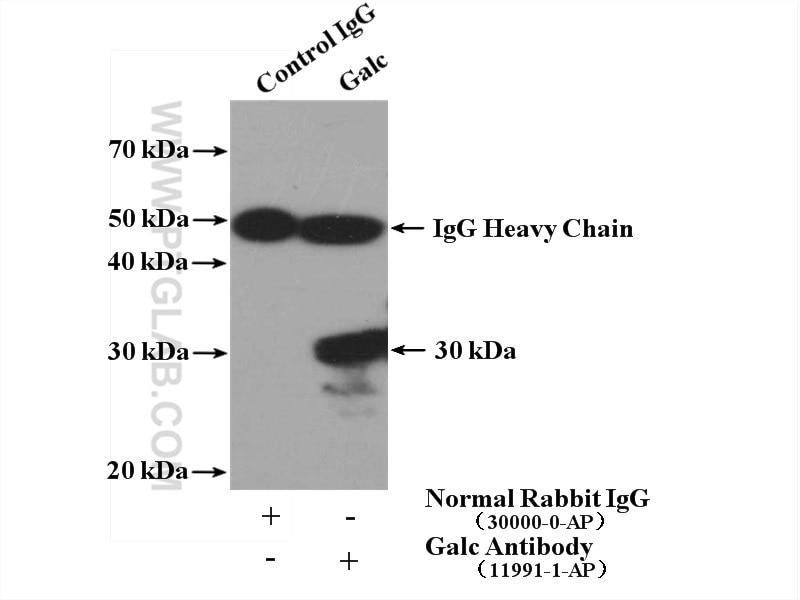
| Positive IP detected in | NIH/3T3 cells |
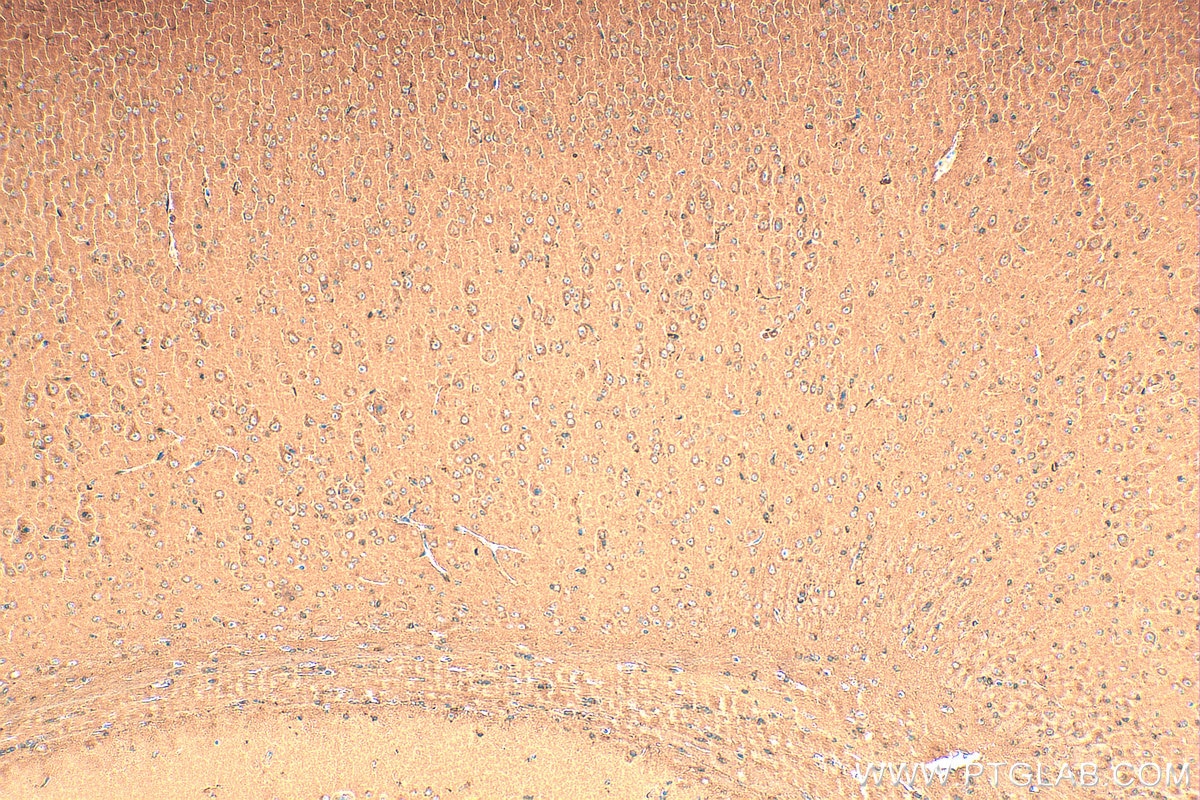
| Positive IHC detected in | mouse brain tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
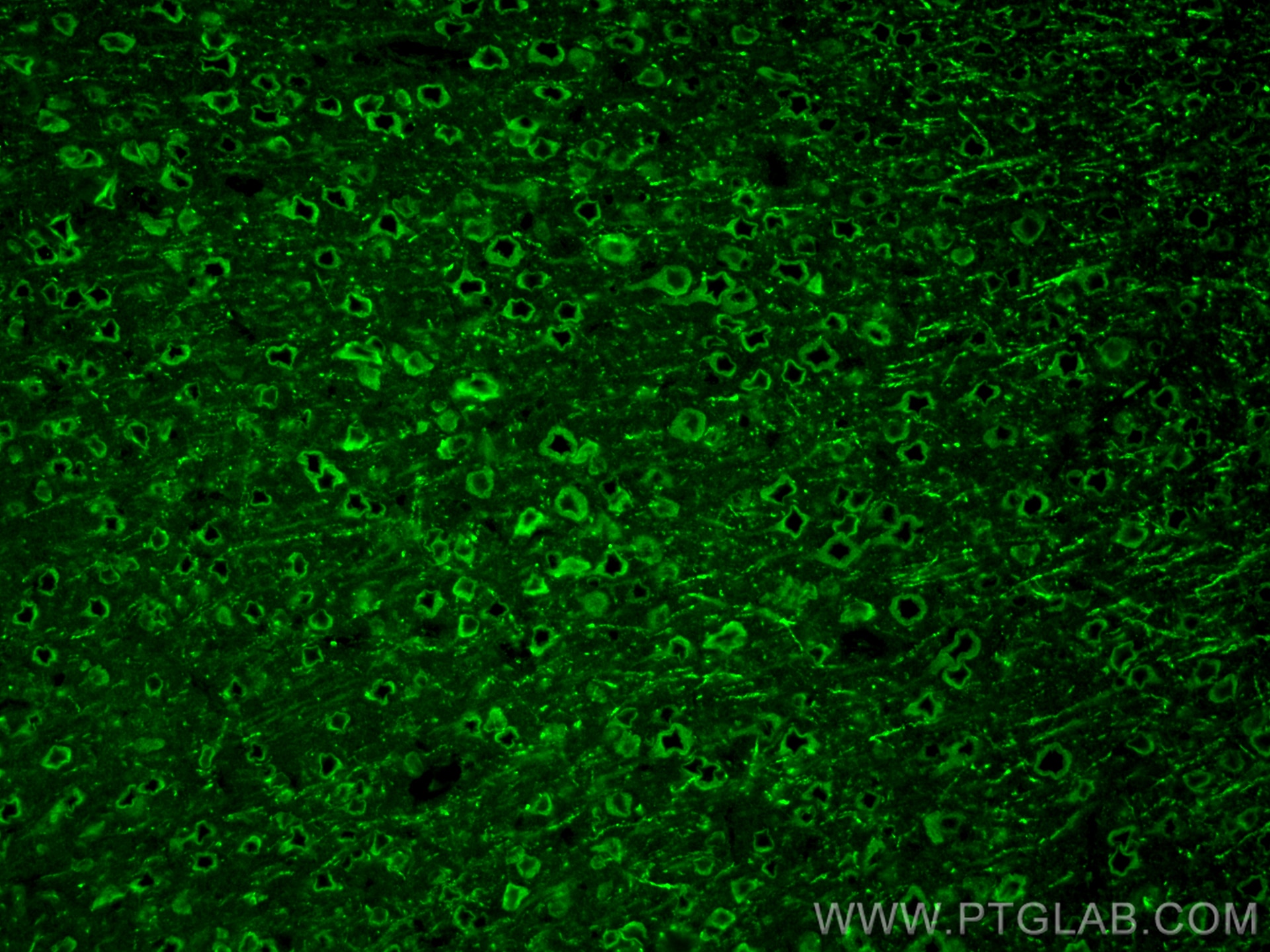
| Positive IF-P detected in | mouse brain tissue |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:1000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunohistochemistry (IHC) | IHC : 1:50-1:500 |
| Immunofluorescence (IF)-P | IF-P : 1:50-1:500 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 1 publications below |
| WB | See 11 publications below |
| IHC | See 4 publications below |
| IF | See 9 publications below |
Product Information
11991-1-AP targets Galc in WB, IHC, IF-P, IP, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse, rat, zebrafish |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag3914 Product name: Recombinant mouse Galc protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 340-513 aa of BC086671 Sequence: YYLKTVGHLEKGGSYVALTDGLGNLTIIIETMSHQHSMCIRPYLPYYNVSHQLATFTLKGSLREIQELQVWYTKLGTPQQRLHFKQLDTLWLLDGSGSFTLELEEDEIFTLTTLTTGRKGSYPPPPSSKPFPTNYKDDFNVEYPLFSEAPNFADQTGVFEYYMNNEDREHRFTL Predict reactive species |
| Full Name | galactosylceramidase |
| Calculated Molecular Weight | 77 kDa |
| Observed Molecular Weight | 80 kDa, 30 kDa, 50 kDa |
| GenBank Accession Number | BC086671 |
| Gene Symbol | Galc |
| Gene ID (NCBI) | 14420 |
| RRID | AB_10641987 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | P54818 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
The GALC antibody targets the liposomal enzyme Galactosylceramidase (GALC), which belongs to the glycosyl hydrolase 59 family. It hydrolyzes the galactose ester bonds of galactosylceramide, galactosylsphingosine, lactosylceramide, and monogalactosyldiglyceride. It is primarily found in the brain and kidneys where galactolipids are hydrolyzed (PMID:8634707). Deficiencies of GALC are primarily associated with the autosomal recessive Krabbe's disease. This disease is characterized by developmental delay caused by apoptosis of myelin-forming cells. GALC is responsible for hydrolyzing galactosylceramide, a cerebroside that is an important component of myelin. A deficiency in GALC causes loss of myelin to nerve cells, resulting in delayed nerve transmissions. Krabbe's disease has varying degrees of severity due to a large number of different genetic mutations in the gene. The GALC antibody can be used to detect the deletions in the GALC gene and functions of the enzyme (PMID:20886637). Normal GALC mRNA encodes the 80 kDa precursor, which is processed into 50 and 30 kDa subunits (PMID: 26865610).
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for Galc antibody 11991-1-AP | Download protocol |
| IHC protocol for Galc antibody 11991-1-AP | Download protocol |
| IP protocol for Galc antibody 11991-1-AP | Download protocol |
| WB protocol for Galc antibody 11991-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Dev Cell NPC1-mTORC1 Signaling Couples Cholesterol Sensing to Organelle Homeostasis and Is a Targetable Pathway in Niemann-Pick Type C. | ||
Stem Cells Multipotent Stromal Cells Alleviate Inflammation, Neuropathology, and Symptoms Associated with Globoid Cell Leukodystrophy in the Twitcher Mouse. | ||
Stem Cell Res Ther Enhanced neuromorphogenesis of neural stem cells via the optimization of physical stimulus-responsive signaling pathways | ||
Mol Ther Extended normal life after AAVrh10-mediated gene therapy in the mouse model of Krabbe disease. | ||
Mol Ther AAV-mediated expression of galactocerebrosidase in brain results in attenuated symptoms and extended life span in murine models of globoid cell leukodystrophy. | ||
Glia Ncx3 gene ablation impairs oligodendrocyte precursor response and increases susceptibility to experimental autoimmune encephalomyelitis. |
Reviews
The reviews below have been submitted by verified Proteintech customers who received an incentive for providing their feedback.
FH Celia (Verified Customer) (01-14-2025) | Antibody worked well, arrived promptly and the team were really helpful getting the order sorted with my university.
|