Tested Applications
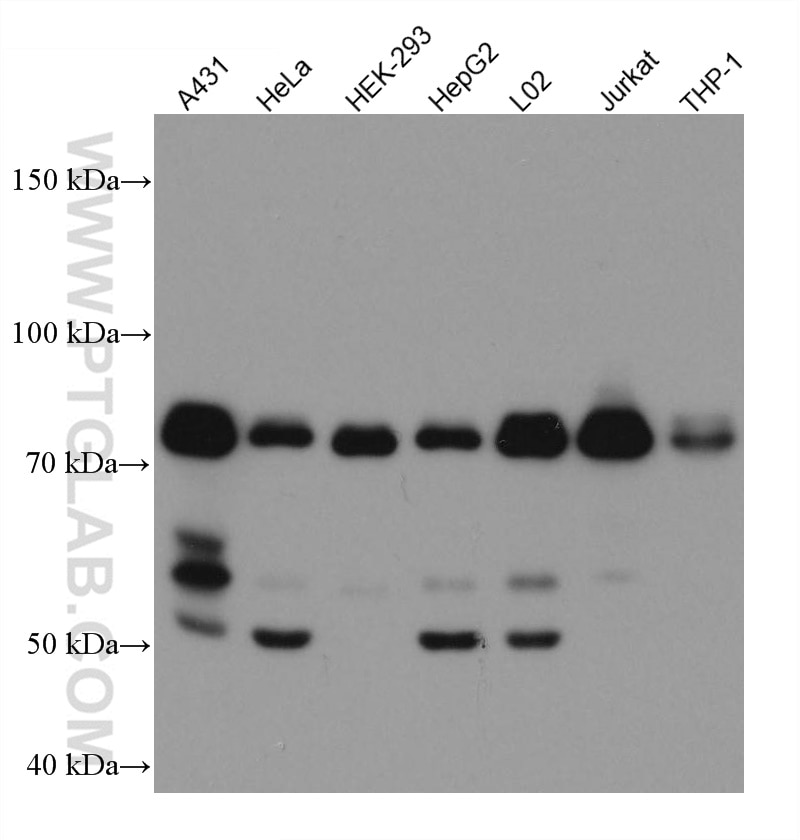
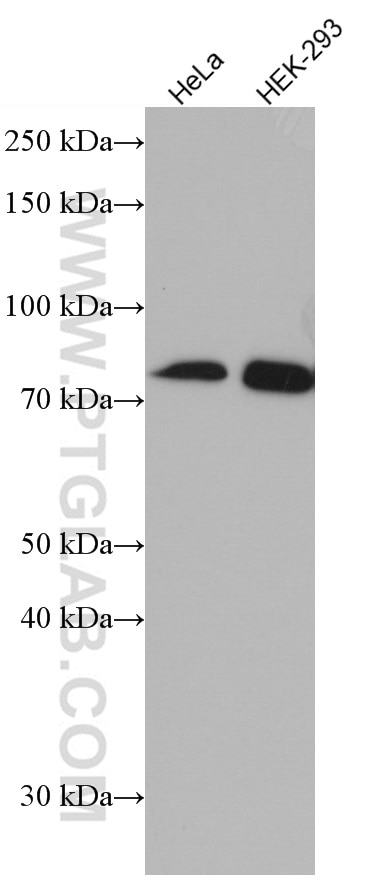
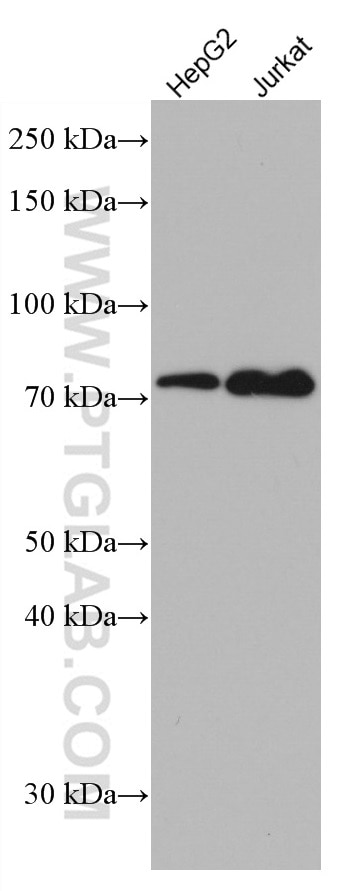
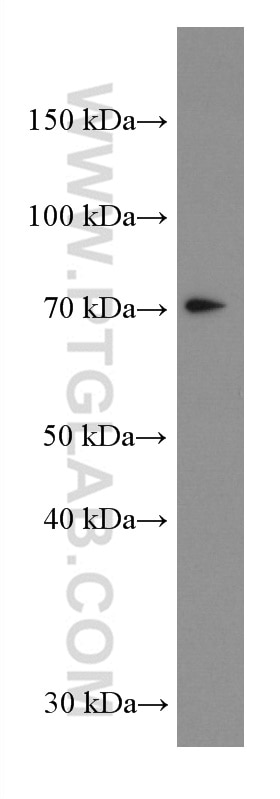
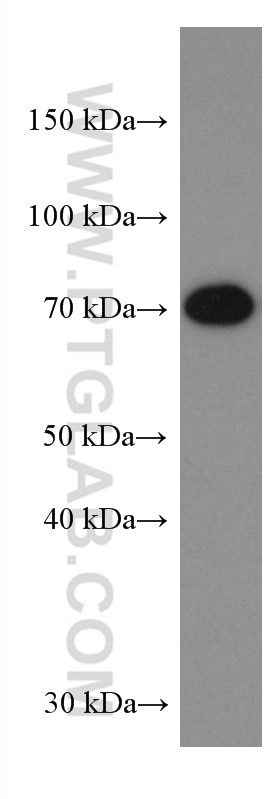
| Positive WB detected in | A431 cells, HeLa cells, HepG2 cells, Jurkat cells, L02 cells, HEK-293 cells, THP-1cells |
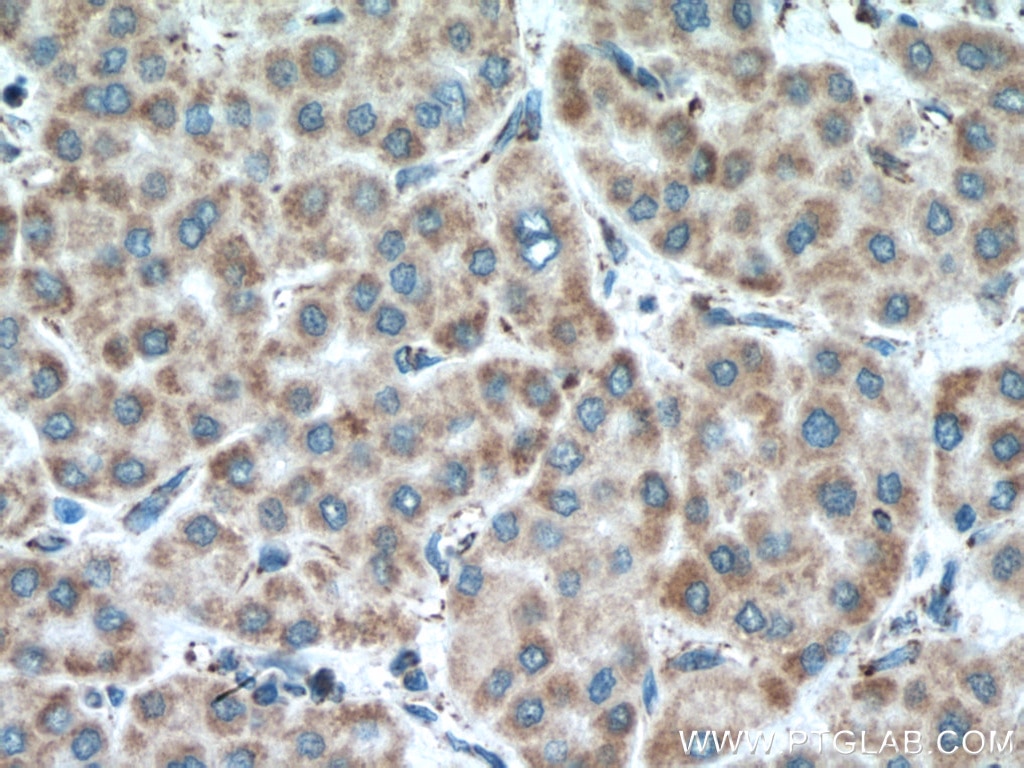
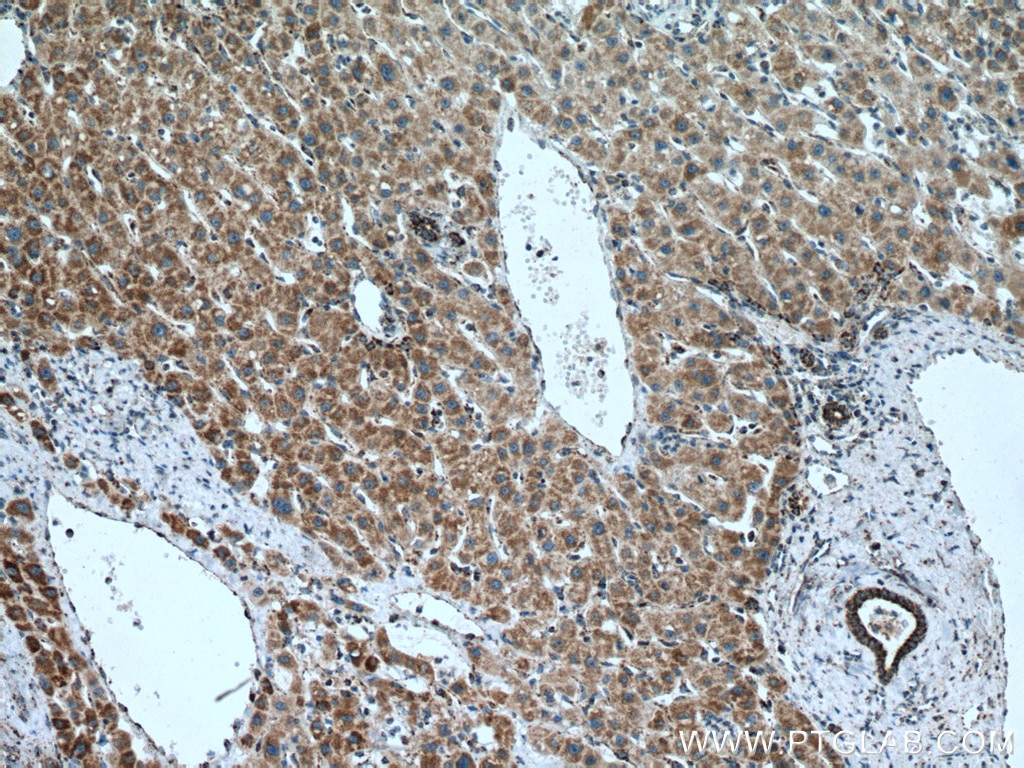
| Positive IHC detected in | human liver cancer tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:5000-1:50000 |
| Immunohistochemistry (IHC) | IHC : 1:550-1:2200 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| WB | See 15 publications below |
| IF | See 4 publications below |
| IP | See 1 publications below |
| CoIP | See 2 publications below |
Product Information
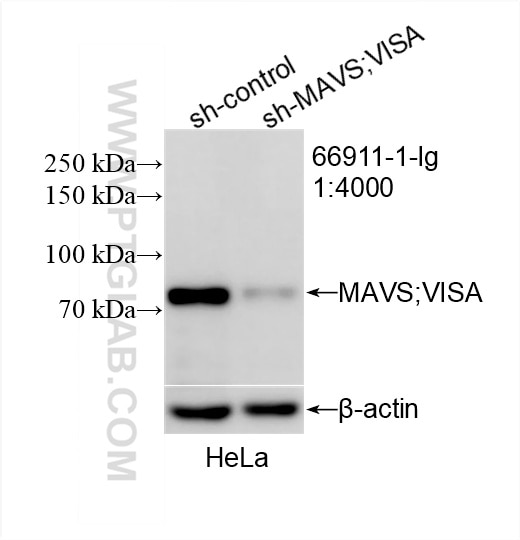
66911-1-Ig targets MAVS; VISA in WB, IHC, IF, IP, CoIP, ELISA applications and shows reactivity with human samples.
| Tested Reactivity | human |
| Cited Reactivity | human, mouse, pig, monkey |
| Host / Isotype | Mouse / IgG1 |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag5949 Product name: Recombinant human MAVS; VISA protein Source: e coli.-derived, PET28a Tag: 6*His Domain: 1-347 aa of BC044952 Sequence: MPFAEDKTYKYICRNFSNFCNVDVVEILPYLPCLTARDQDRLRATCTLSGNRDTLWHLFNTLQRRPGWVEYFIAALRGCELVDLADEVASVYESYQPRTSDRPPDPLEPPSLPAERPGPPTPAAAHSIPYNSCREKEPSYPMPVQETQAPESPGENSEQALQTLSPRAIPRNPDGGPLESSSDLAALSPLTSSGHQEKDTELGSTHTAGATSSLTPSRGPVSPSVSFQPLARSTPRASRLPGPTGSVVSTGTSFSSSSPGLASAGAAEGKQGAESDQAEPIICSSGAEAPANSLPSKVPTTLMPVNTVALKVPANPASVSTVPSKLPTSSKPPGAVPSNALTNPAPS Predict reactive species |
| Full Name | mitochondrial antiviral signaling protein |
| Calculated Molecular Weight | 57 kDa |
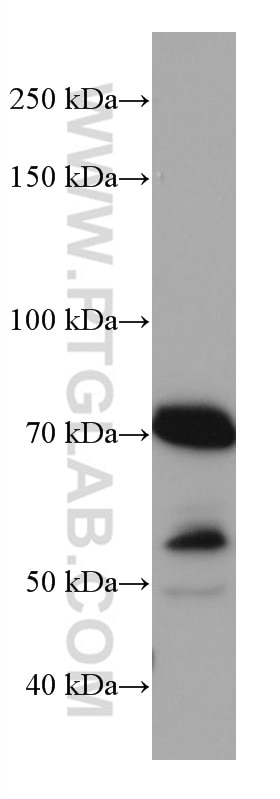
| Observed Molecular Weight | 50-55 kDa, 70-75 kDa |
| GenBank Accession Number | BC044952 |
| Gene Symbol | MAVS |
| Gene ID (NCBI) | 57506 |
| RRID | AB_2882238 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Protein G purification |
| UNIPROT ID | Q7Z434 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
Mitochondrial antiviral-signaling protein (MAVS) is also known as virus-induced-signaling adapter (VISA) or IFN beta promoter stimulator protein 1 (IPS-1), it is widely involved and required for innate immune defense against viruses. MAVS, present in T cells, monocytes, epithelial cells and hepatocytes, contains CARD and transmembrane domains which are essential for antiviral functions. MAVS is able to interact with various cellular proteins including DDX58/RIG-I, IFIH1/MDA5, TRAF2, TRAF6, TMEM173/MITA, IFIT3 and etc. It can undergoe phosphorylation on multiple sites and ubiquitination, which may together cause the molecular weight migrate to about 70 kDa despite the predicated 57 kDa.
Protocols
| Product Specific Protocols | |
|---|---|
| IHC protocol for MAVS; VISA antibody 66911-1-Ig | Download protocol |
| WB protocol for MAVS; VISA antibody 66911-1-Ig | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Mol Cell An Epstein-Barr virus protein interaction map reveals NLRP3 inflammasome evasion via MAVS UFMylation | ||
J Immunother Cancer Targeting KDM4C enhances CD8+ T cell mediated antitumor immunity by activating chemokine CXCL10 transcription in lung cancer. | ||
Front Immunol Swine Acute Diarrhea Syndrome Coronavirus Nucleocapsid Protein Antagonizes Interferon-β Production via Blocking the Interaction Between TRAF3 and TBK1. | ||
J Virol Encephalomyocarditis virus abrogates the IFN-β signaling pathway via its structural protein VP2. | ||
Vet Microbiol DDX56 antagonizes IFN-β production to enhance EMCV replication by inhibiting IRF3 nuclear translocation | ||
Virus Res Host protein, HSP90β, antagonizes IFN-β signaling pathway and facilitates the proliferation of encephalomyocarditis virus in vitro. |