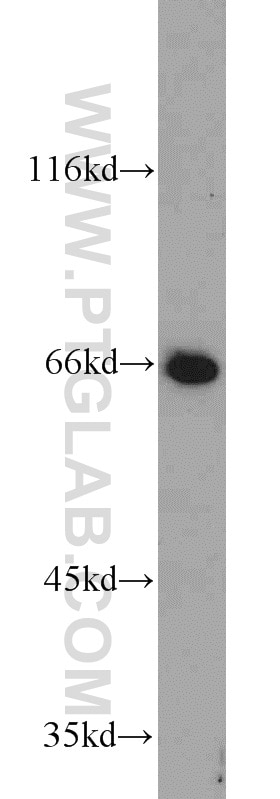
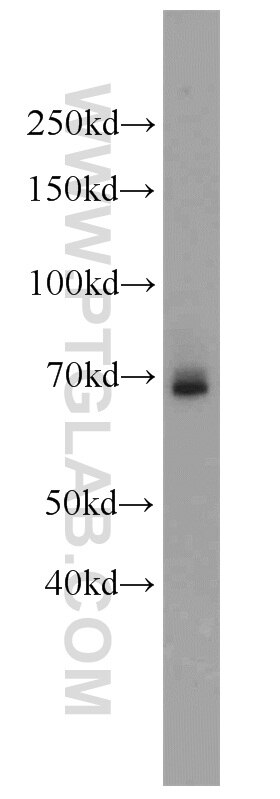
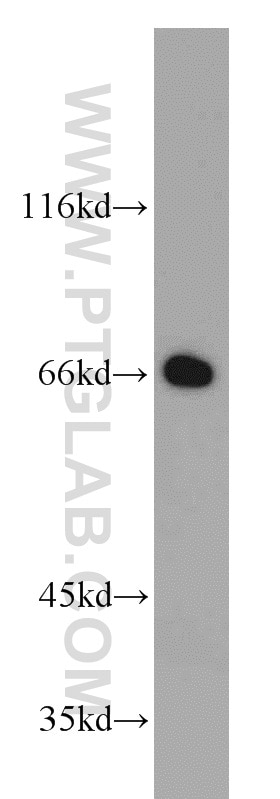
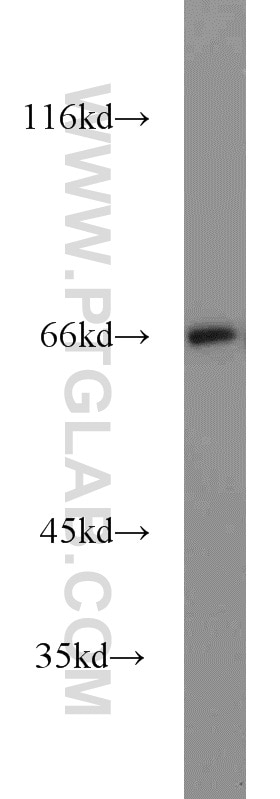
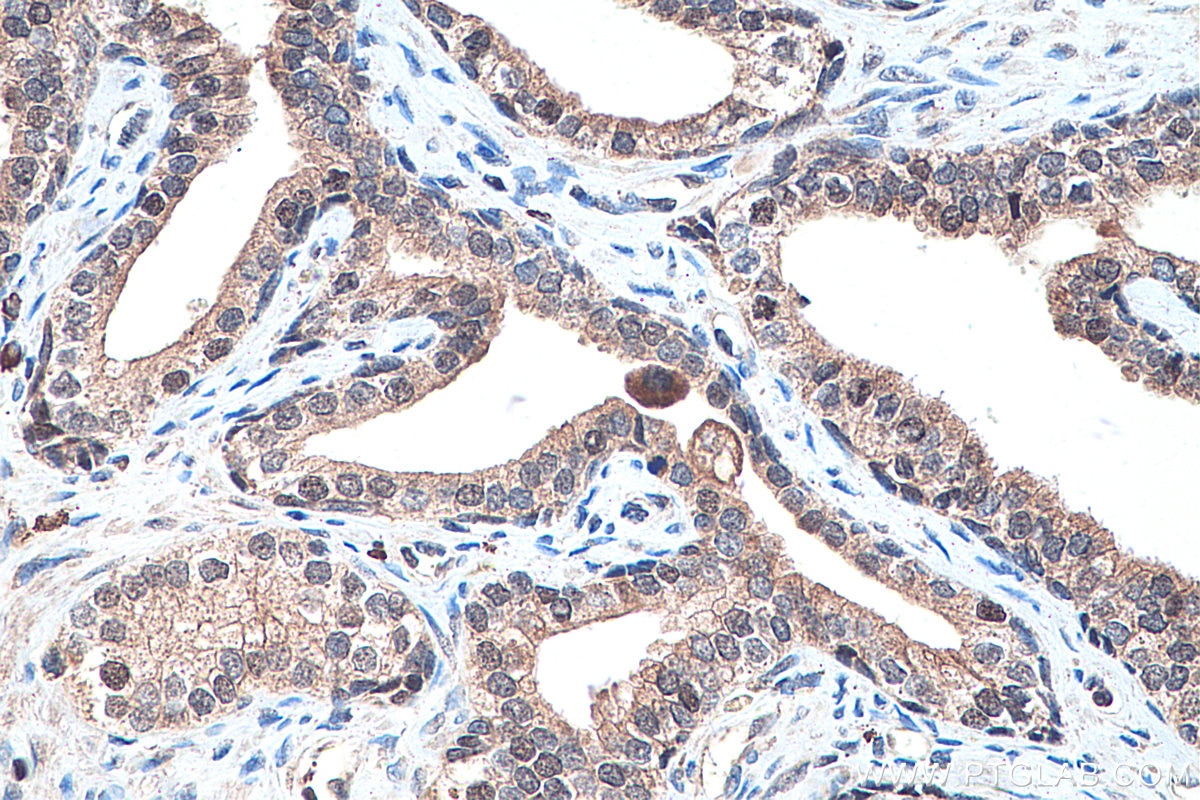
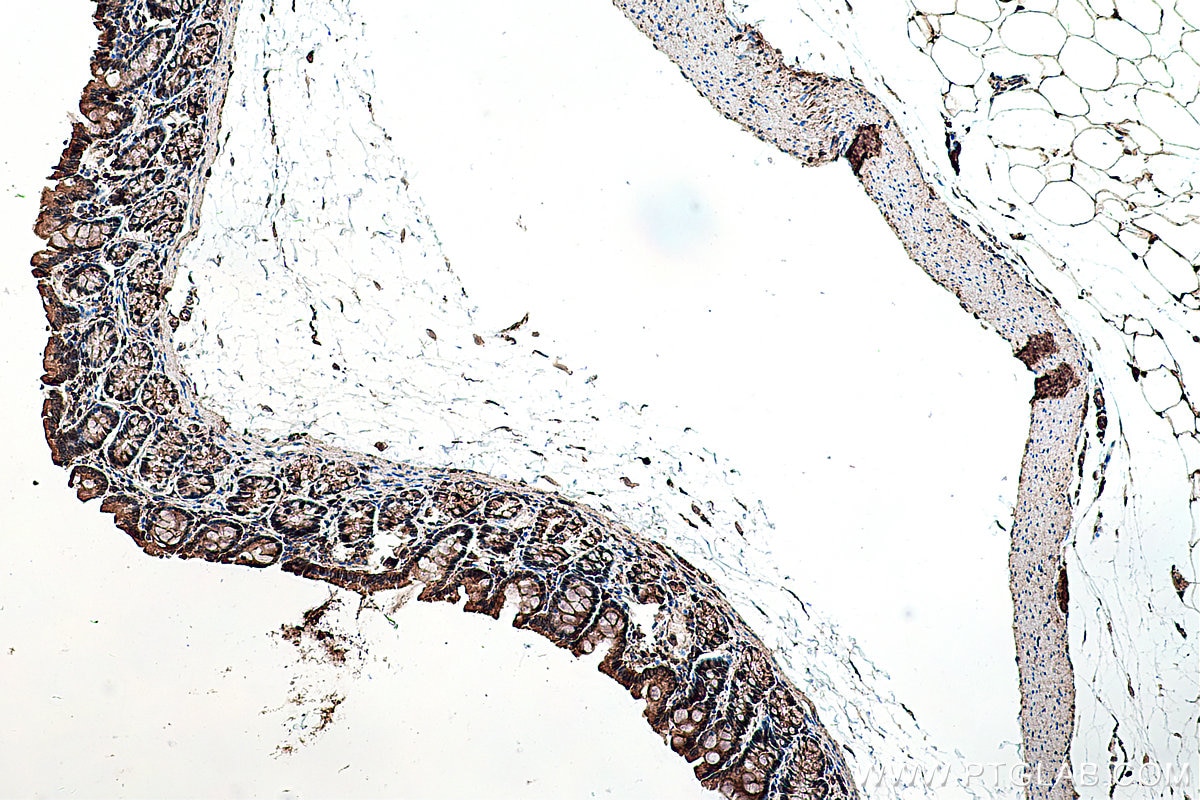
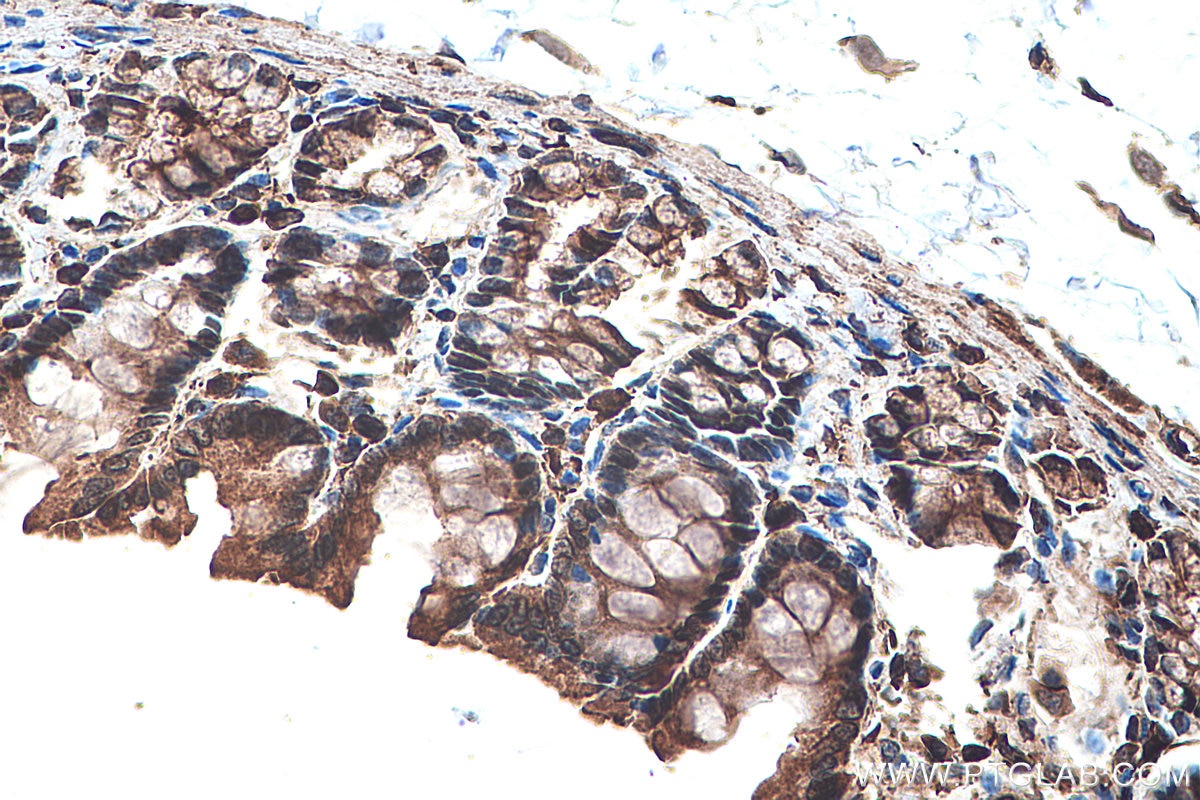
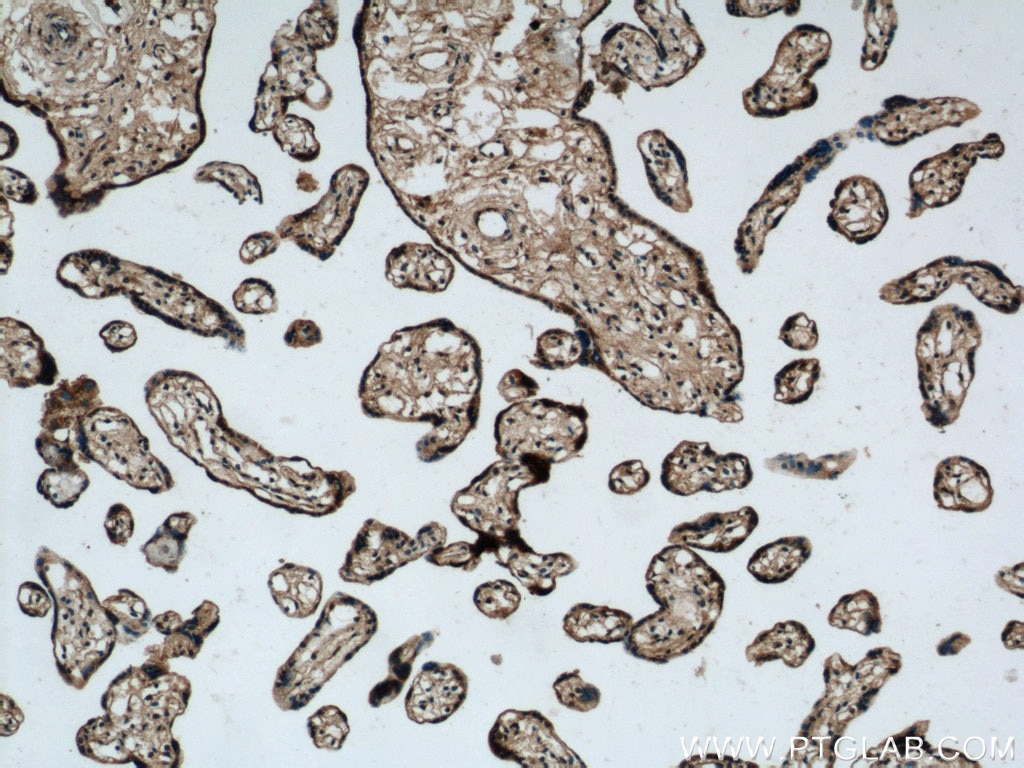
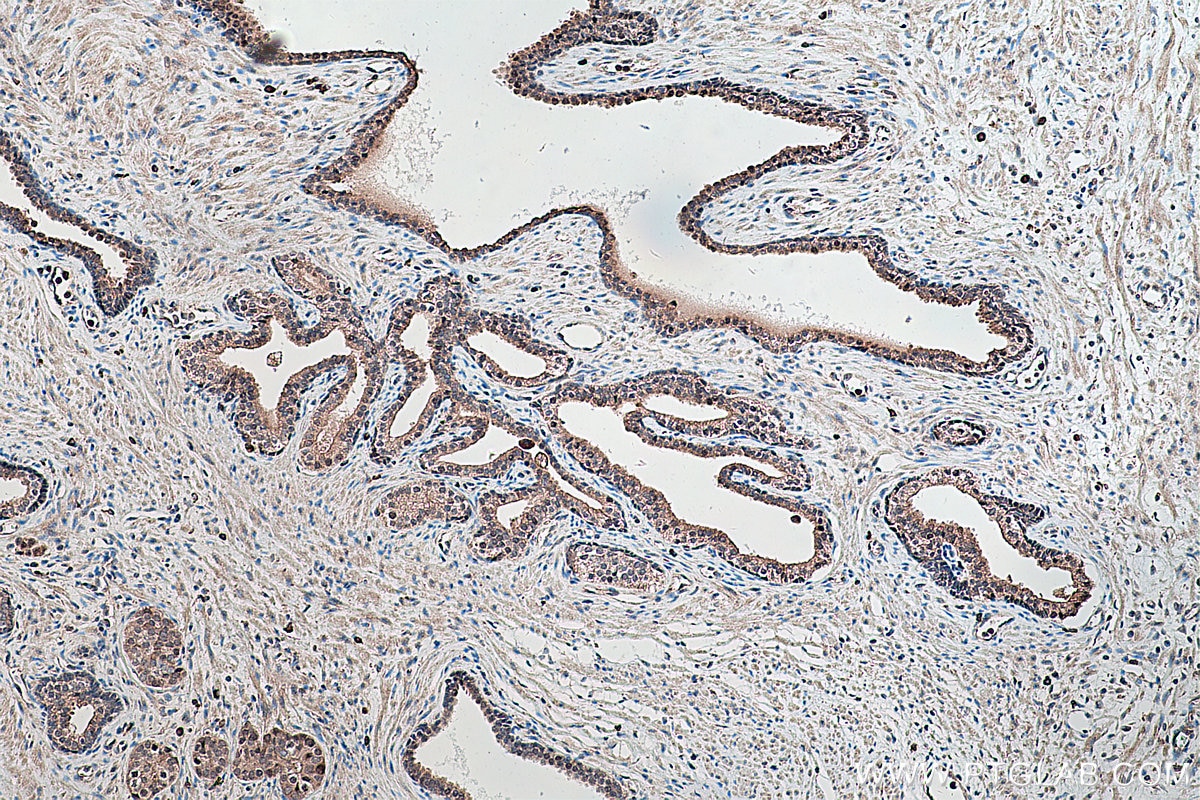
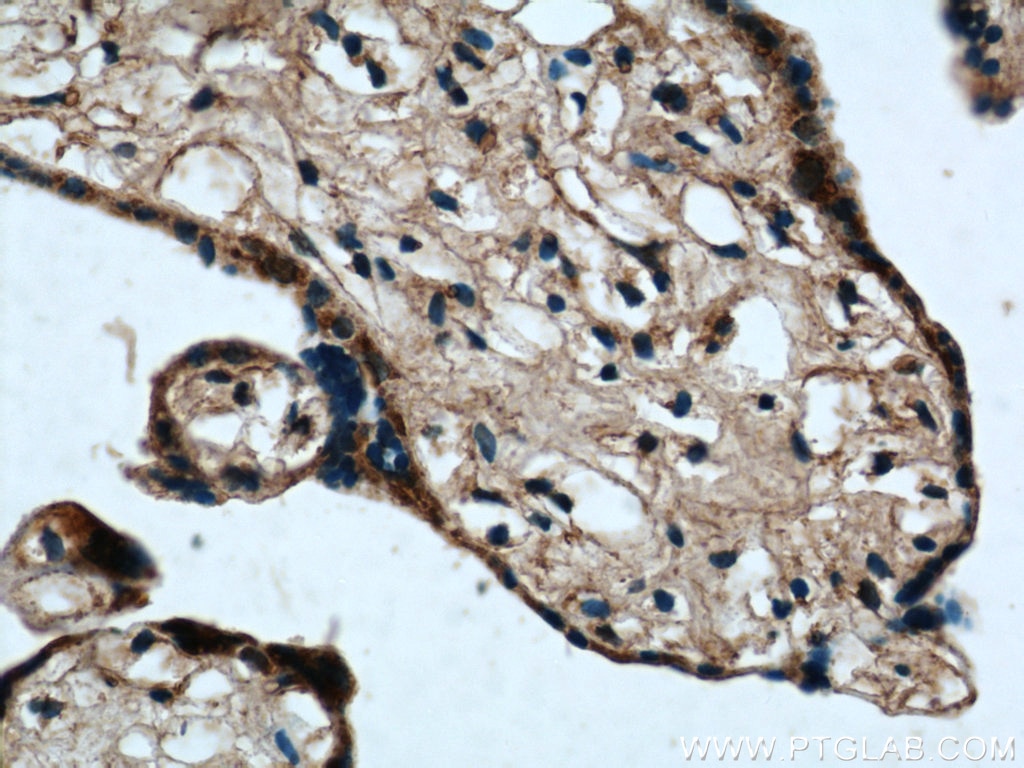
Tested Applications
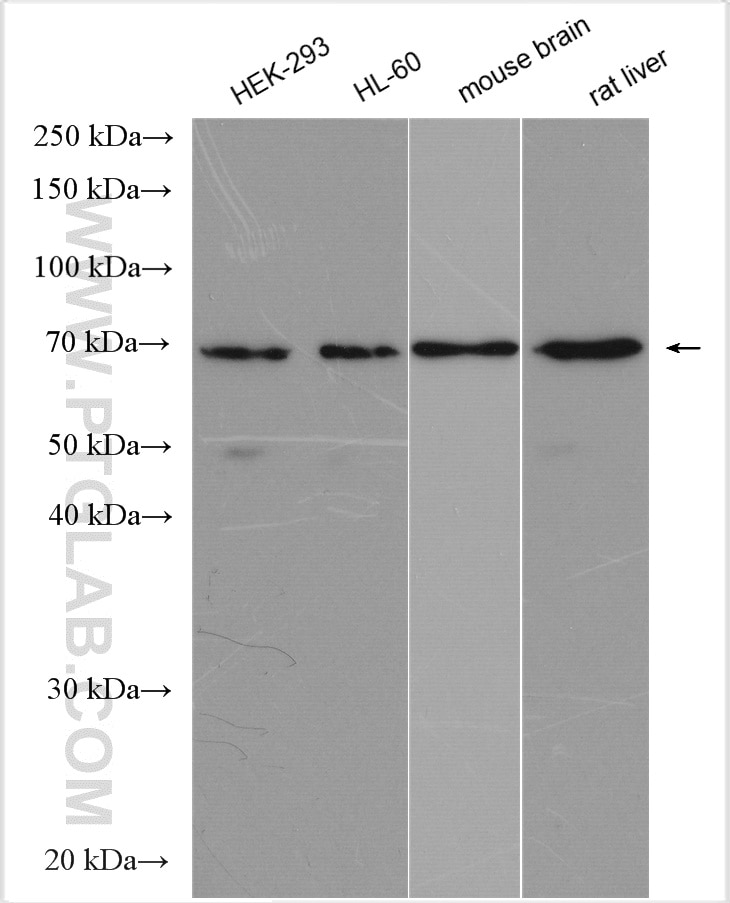
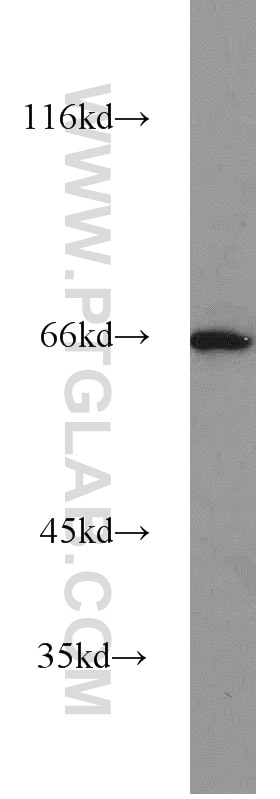
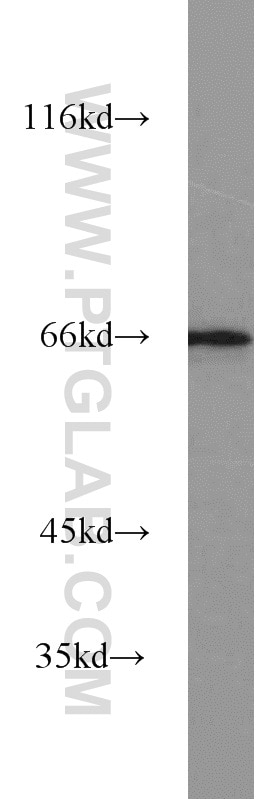
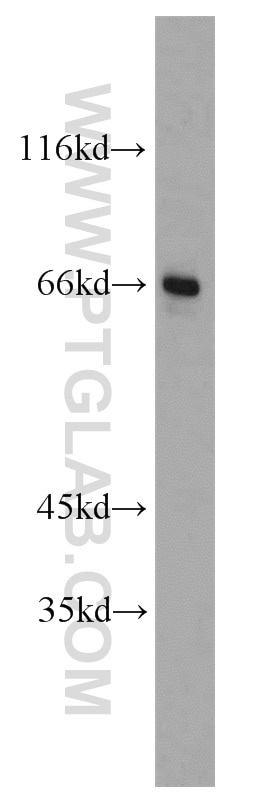
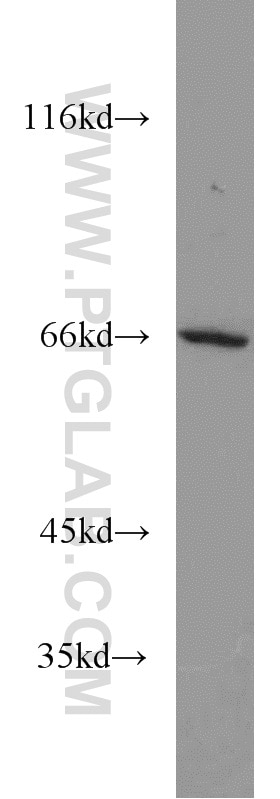
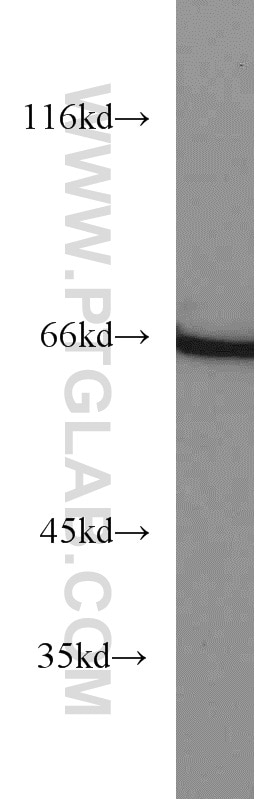
| Positive WB detected in | HEK-293 cells, human brain tissue, human heart tissue, mouse ovary tissue, mouse heart tissue, mouse testis tissue, mouse liver tissue, MCF-7 cells, 3T3-L1 cells, rat brain tissue, HL-60 cells, mouse brain tissue, rat liver tissue |
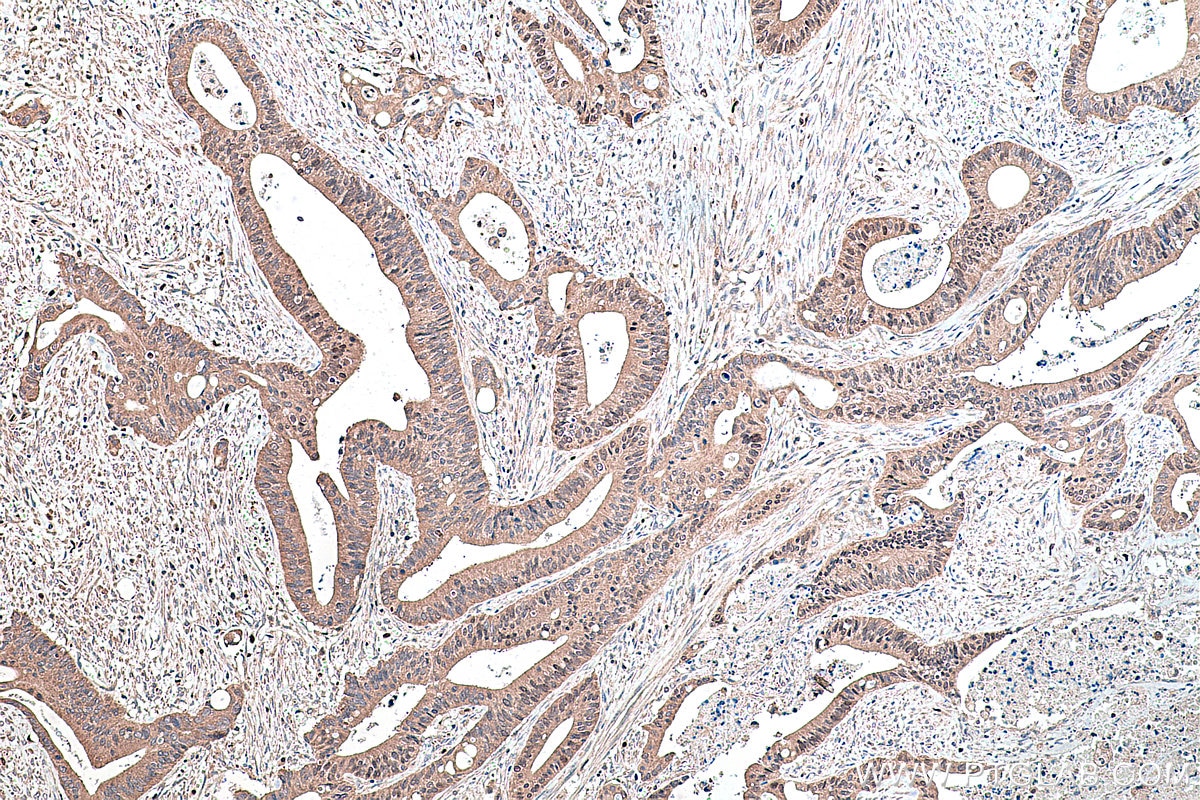
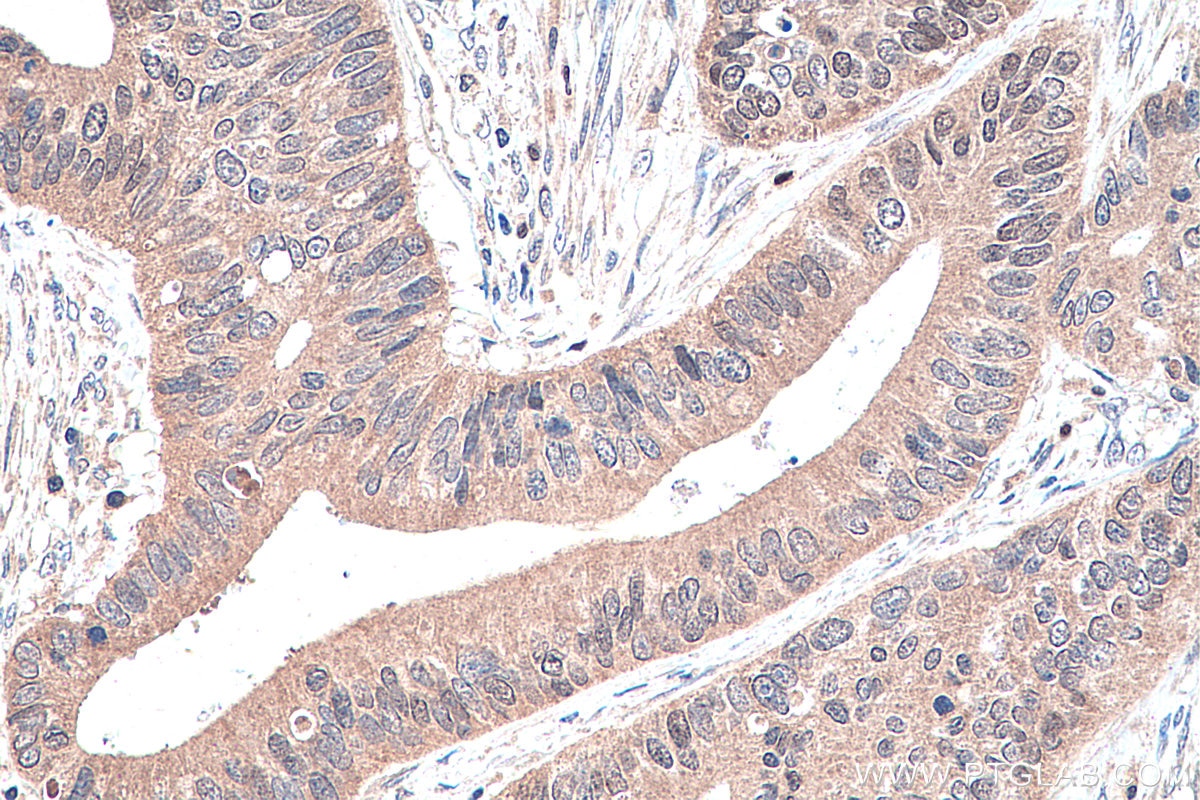
| Positive IHC detected in | human prostate cancer tissue, human colon cancer tissue, rat colon tissue, human placenta tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:1000-1:6000 |
| Immunohistochemistry (IHC) | IHC : 1:500-1:2000 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 1 publications below |
| WB | See 10 publications below |
| IHC | See 2 publications below |
Product Information
22061-1-AP targets PPAR Gamma in WB, IHC, ELISA applications and shows reactivity with human, mouse, rat samples.
| Tested Reactivity | human, mouse, rat |
| Cited Reactivity | human, mouse, rat |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag17136 Product name: Recombinant human PPARG protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1-111 aa of BC006811 Sequence: MTMVDTEMPFWPTNFGISSVDLSVMEDHSHSFDIKPFTTVDFSSISTPHYEDIPFTRTDPVVADYKYDLKLQEYQSAIKVEPASPPYYSEKTQLYNKPHEEPSNSLMAIEC Predict reactive species |
| Full Name | peroxisome proliferator-activated receptor gamma |
| Calculated Molecular Weight | 505 aa, 58 kDa |
| Observed Molecular Weight | 66-70 kDa |
| GenBank Accession Number | BC006811 |
| Gene Symbol | PPARG |
| Gene ID (NCBI) | 5468 |
| RRID | AB_11182268 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | P37231 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
Peroxisome Proliferator-Activated Receptors (PPARs) are ligand-activated intracellular transcription factors, members of the nuclear hormone receptor superfamily (NR), that includes estrogen, thyroid hormone receptors, retinoic acid, Vitamin D3 as well as retinoid X receptors (RXRs). The PPAR subfamily consists of three subtypes encoded by distinct genes denoted PPARα (NR1C1), PPARβ/δ (NR1C2) and PPARγ (NR1C3), which are activated by selective ligands. PPARγ, also named as PPARG, contains one nuclear receptor DNA-binding domain and is a receptor that binds peroxisome proliferators such as hypolipidemic drugs and fatty acids. It plays an important role in the regulation of lipid homeostasis, adipogenesis, INS resistance, and development of various organs. Defects in PPARG are the cause of familial partial lipodystrophy type 3 (FPLD3) and may be associated with susceptibility to obesity. Defects in PPARG can lead to type 2 INS-resistant diabetes and hypertension. PPARG mutations may be associated with colon cancer. Genetic variations in PPARG are associated with susceptibility to glioma type 1 (GLM1). PPARG has two isoforms with molecular weight 57 kDa and 54 kDa (PMID: 9831621), but modified PPARG is about 67 KDa (PMID: 16809887). PPARG2 is a splice variant and has an additional 30 amino acids at the N-terminus (PMID: 15689403). Experimental data indicate that a 45 kDa protein displaying three different sequences immunologically related to the nuclear receptor PPARG2 is located in mitochondria (mt-PPAR). However, the molecular weight of this protein is clearly less when compared to that of PPARG2 (57 kDa). (PMID: 10922459). PPARG has been reported to be localized mainly (but not always) in the nucleus. PPARG can also be detected in the cytoplasm and was reported to possess extra-nuclear/non-genomic actions (PMID: 17611413; 19432669; 14681322).
Publications
| Species | Application | Title |
|---|---|---|
Genes Dev Loss of TLE3 promotes the mitochondrial program in beige adipocytes and improves glucose metabolism. | ||
Free Radic Biol Med Hydrogen sulfide alleviates particulate matter-induced emphysema and airway inflammation by suppressing ferroptosis. | ||
Biochem Pharmacol A novel pyrazole-containing indolizine derivative suppresses NF-κB activation and protects against TNBS-induced colitis via a PPAR-γ-dependent pathway. | ||
Front Pharmacol The Active Components of Fuzheng Huayu Formula and Their Potential Mechanism of Action in Inhibiting the Hepatic Stellate Cells Viability - A Network Pharmacology and Transcriptomics Approach. | ||
Front Pharmacol Alpha-Asaronol Alleviates Dysmyelination by Enhancing Glutamate Transport Through the Activation of PPARγ-GLT-1 Signaling in Hypoxia-Ischemia Neonatal Rats. |