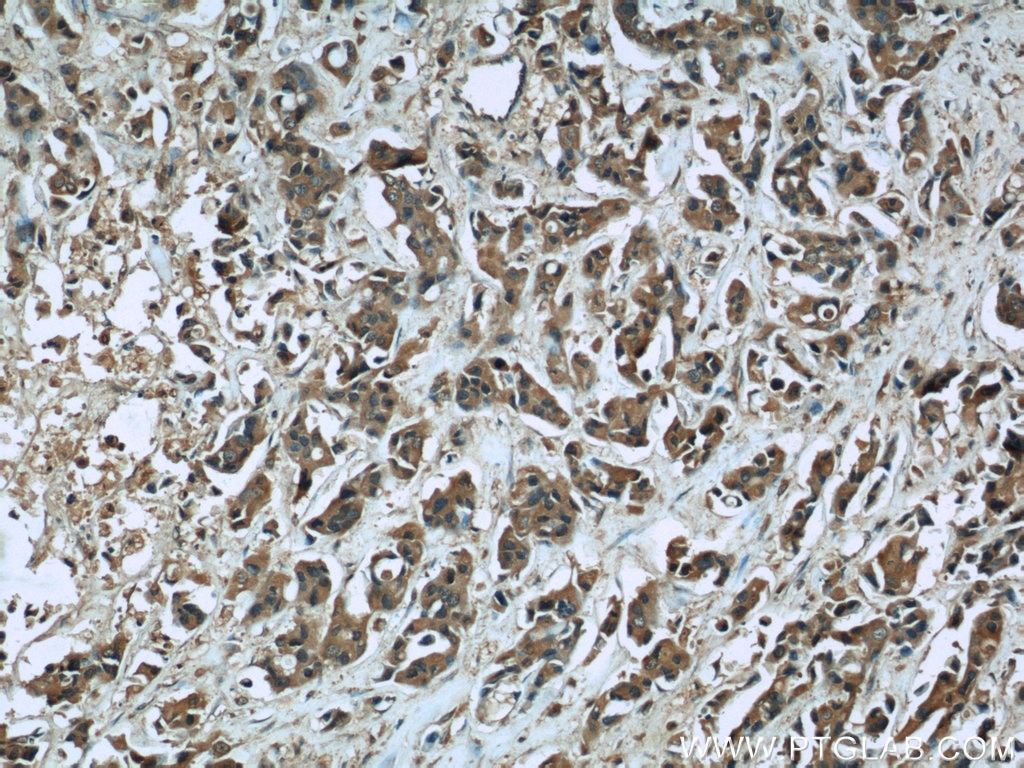
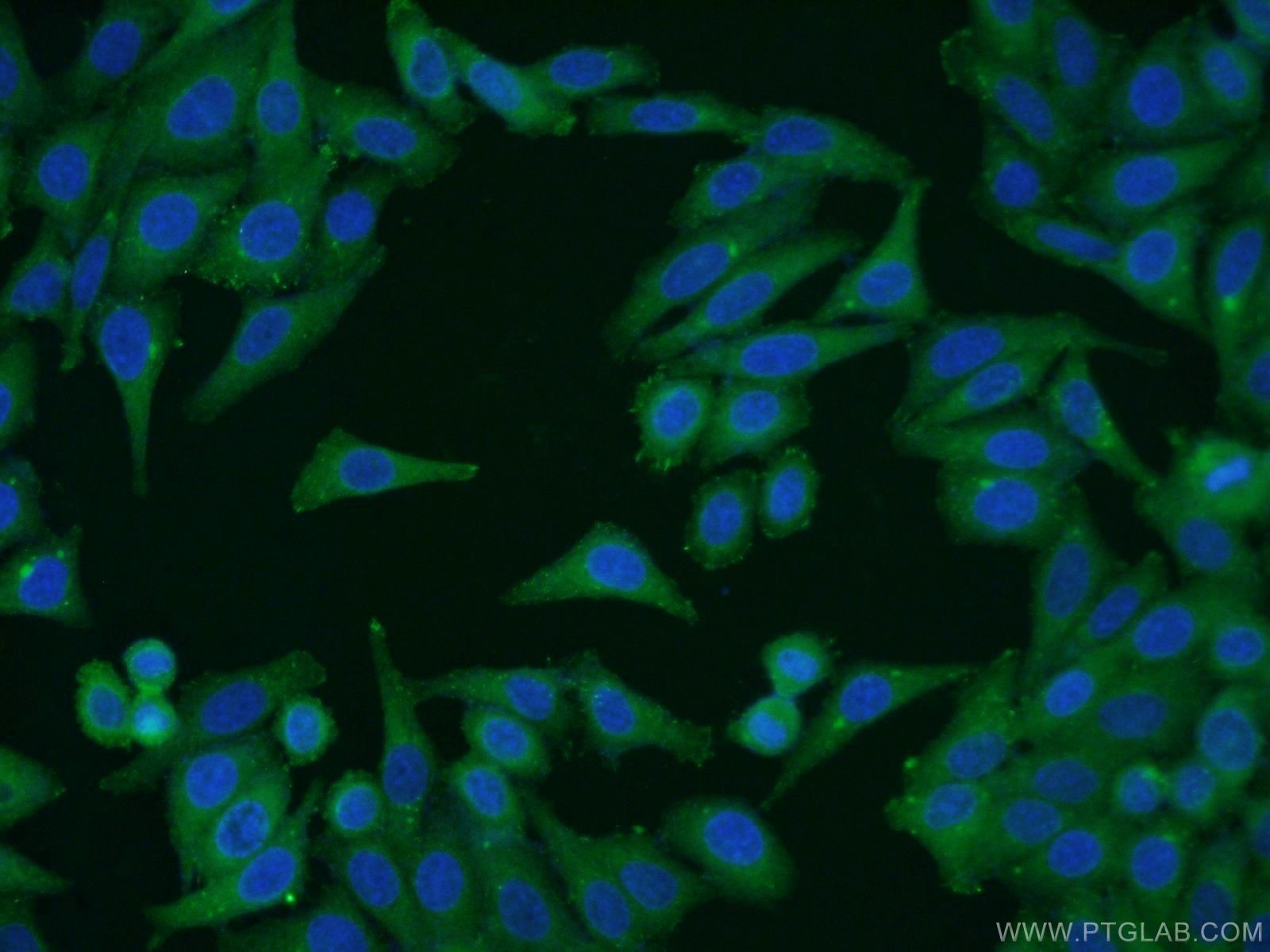
Tested Applications
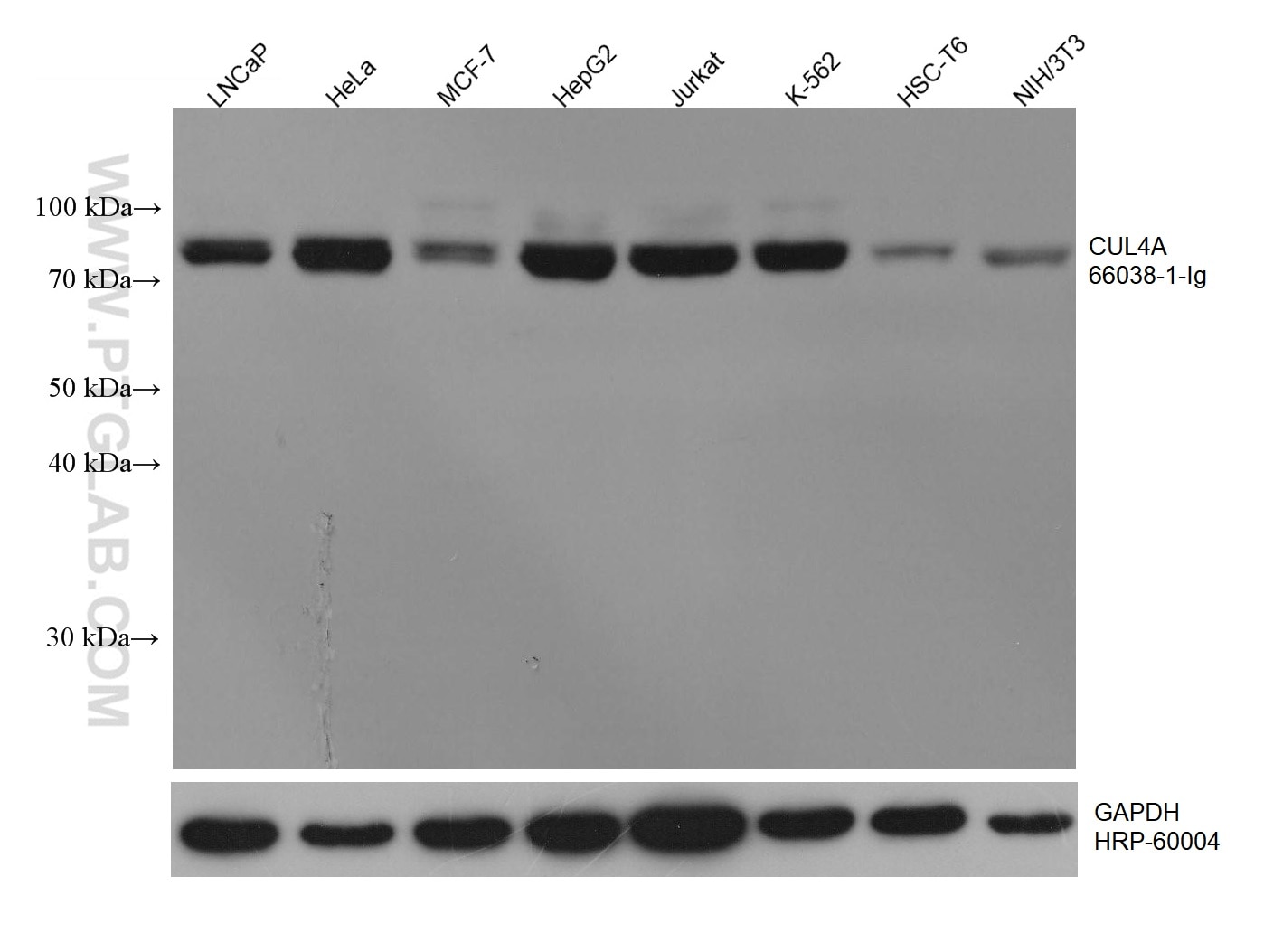
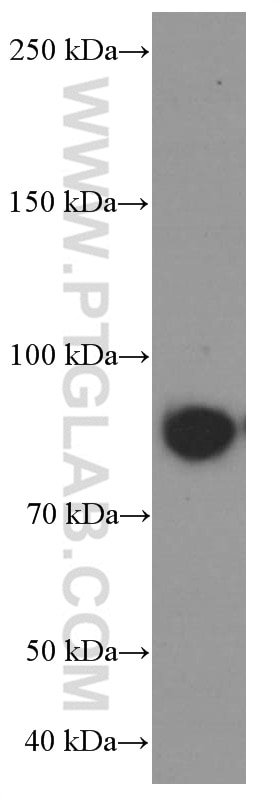
| Positive WB detected in | LNCaP cells, HeLa cells, pig brain tissue, MCF-7 cells, HepG2 cells, Jurkat cells, K-562 cells, HSC-T6 cells, NIH/3T3 cells |
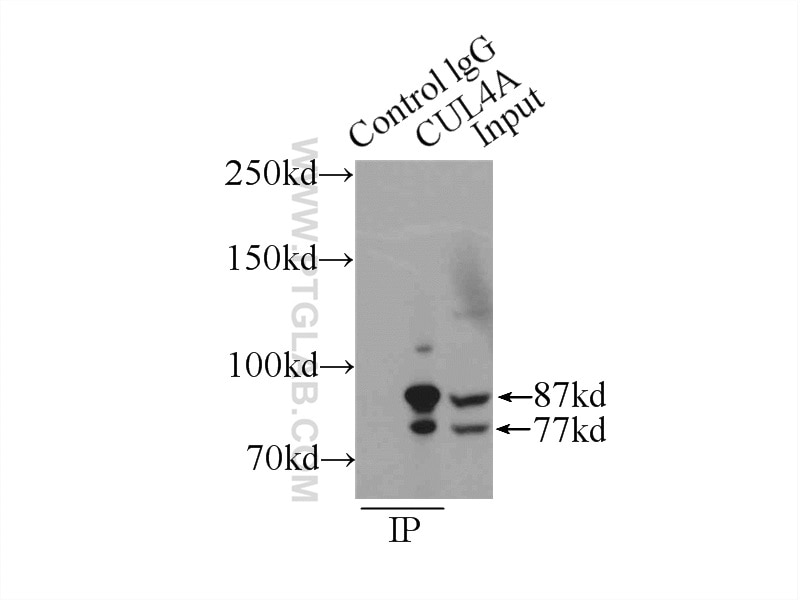
| Positive IP detected in | MCF-7 cells |
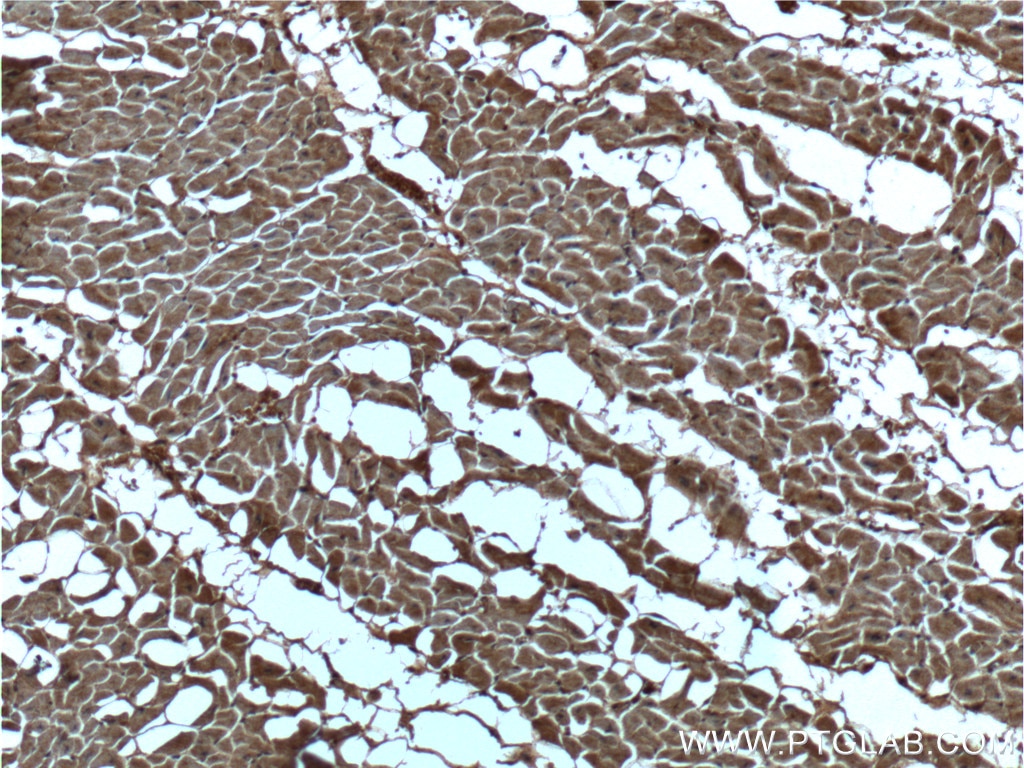
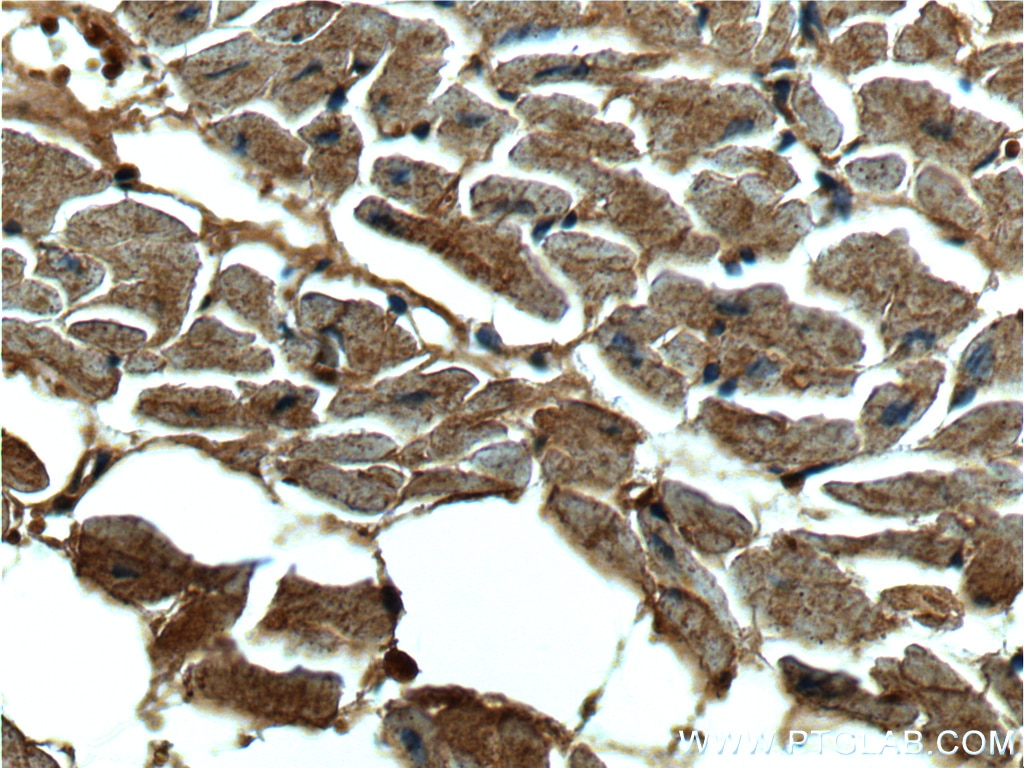
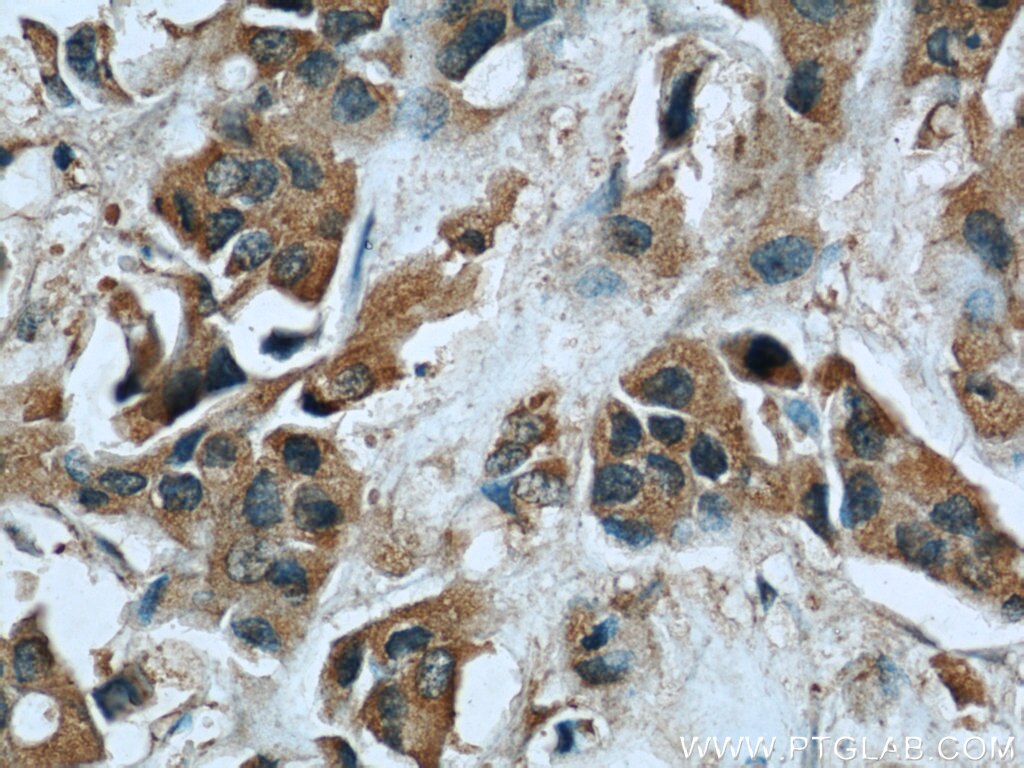
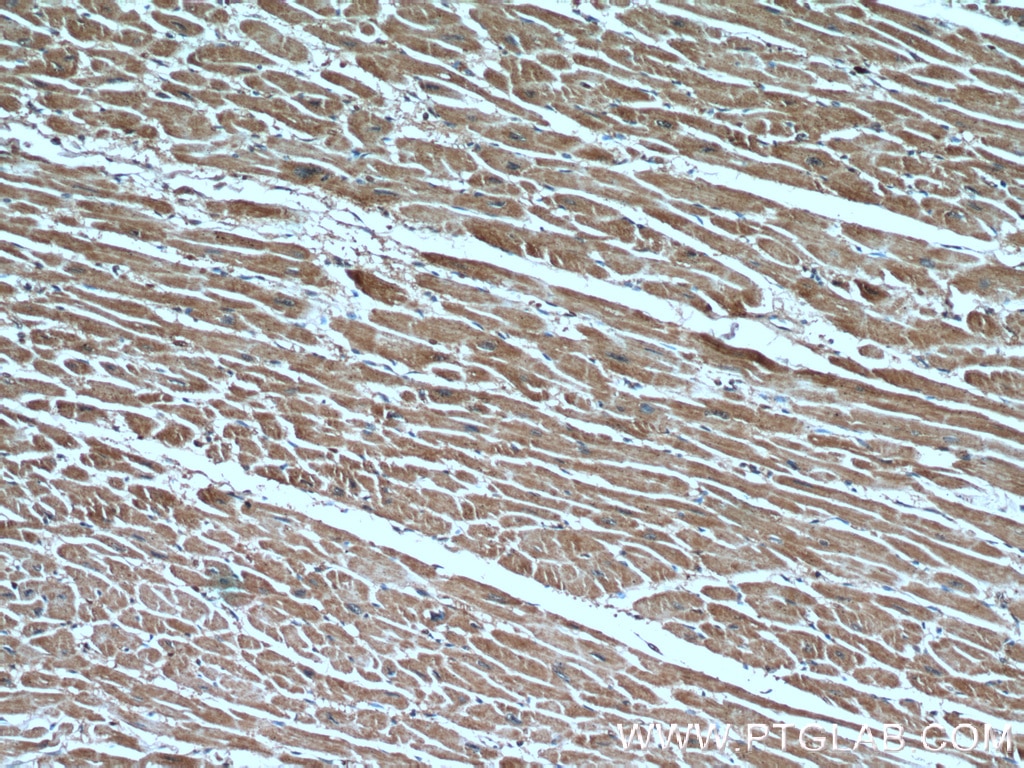
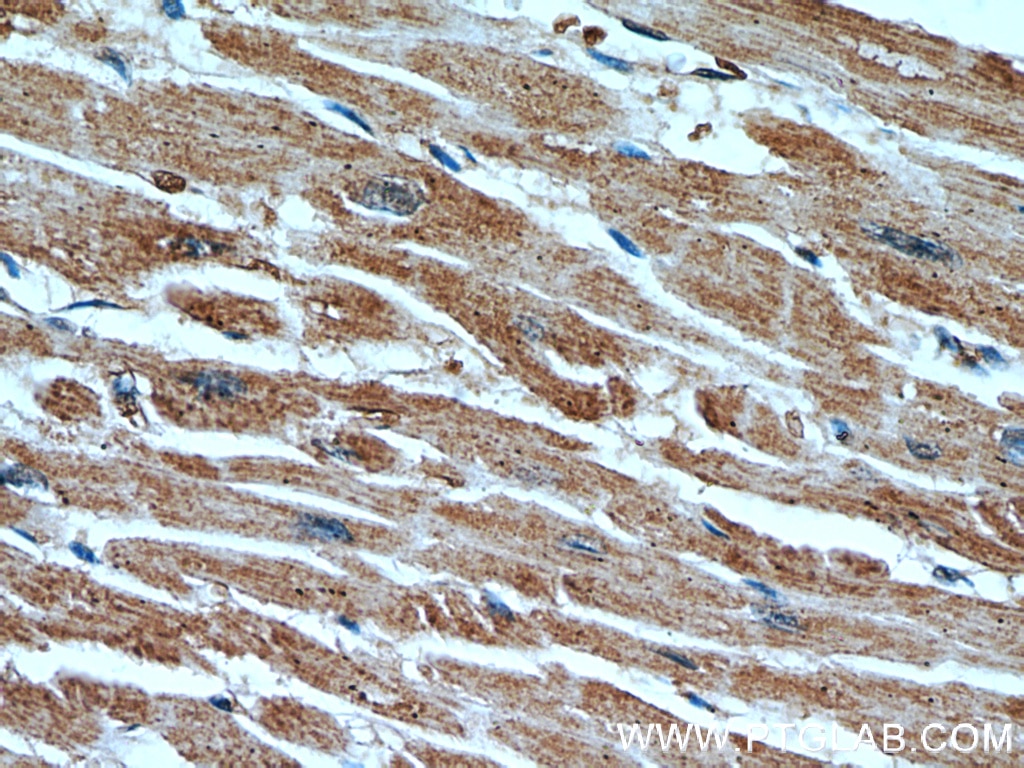
| Positive IHC detected in | human heart tissue, human breast cancer tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
| Positive IF/ICC detected in | HepG2 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:5000-1:50000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunohistochemistry (IHC) | IHC : 1:20-1:200 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:20-1:200 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 1 publications below |
| WB | See 4 publications below |
| IF | See 1 publications below |
| IP | See 2 publications below |
Product Information
66038-1-Ig targets CUL4A in WB, IHC, IF/ICC, IP, ELISA applications and shows reactivity with human, mouse, rat, pig, monkey samples.
| Tested Reactivity | human, mouse, rat, pig, monkey |
| Cited Reactivity | human, mouse |
| Host / Isotype | Mouse / IgG1 |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag18089 Product name: Recombinant human CUL4A protein Source: e coli.-derived, PET28a Tag: 6*His Domain: 460-759 aa of BC008308 Sequence: RLLVGKSASVDAEKSMLSKLKHECGAAFTSKLEGMFKDMELSKDIMVHFKQHMQNQSDSGPIDLTVNILTMGYWPTYTPMEVHLTPEMIKLQEVFKAFYLGKHSGRKLQWQTTLGHAVLKAEFKEGKKEFQVSLFQTLVLLMFNEGDGFSFEEIKMATGIEDSELRRTLQSLACGKARVLIKSPKGKEVEDGDKFIFNGEFKHKLFRIKINQIQMKETVEEQVSTTERVFQDRQYQIDAAIVRIMKMRKTLGHNLLVSELYNQLKFPVKPGDLKKRIESLIDRDYMERDKDNPNQYHYVA Predict reactive species |
| Full Name | cullin 4A |
| Calculated Molecular Weight | 77 kDa |
| Observed Molecular Weight | 77 kDa, 88 kDa |
| GenBank Accession Number | BC008308 |
| Gene Symbol | CUL4A |
| Gene ID (NCBI) | 8451 |
| RRID | AB_11042446 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Protein G purification |
| UNIPROT ID | Q13619 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
Cullin proteins assemble a large number of RING E3 ubiquitin ligases, participating in the proteolysis through the ubiquitin-proteasome pathway. Two cullin 4 (CUL4) proteins, CUL4A (87 kDa) and CUL4B(104 kDa), have been identified. The two CUL4 sequences are 83% identical. They target certain proteins for degradation by binding protein DDB1 to form a CUL4-DDB1 ubiquitin ligase complex with DDB. They form two individual E3 ligases, DDB1-CUL4ADDB2 and DDB1-CUL4BDDB2 in this process. CUL4A appeared in both the nucleus and the cytosol, suggesting a more complex mechanism for entering the nucleus. CUL4B is localized in the nucleus and facilitates the transfer of DDB1 into the nucleus independently of DDB2.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for CUL4A antibody 66038-1-Ig | Download protocol |
| IHC protocol for CUL4A antibody 66038-1-Ig | Download protocol |
| IP protocol for CUL4A antibody 66038-1-Ig | Download protocol |
| WB protocol for CUL4A antibody 66038-1-Ig | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Curr Biol Temporal Regulation of ESCO2 Degradation by the MCM Complex, the CUL4-DDB1-VPRBP Complex, and the Anaphase-Promoting Complex. | ||
Oncogene GSK3β-driven SOX2 overexpression is a targetable vulnerability in esophageal squamous cell carcinoma
| ||
Stem Cell Res Ther KAP1 phosphorylation promotes the survival of neural stem cells after ischemia/reperfusion by maintaining the stability of PCNA. | ||
Cell Death Differ Cullin 4B-RING E3 ligase negatively regulates the immunosuppressive capacity of mesenchymal stem cells by suppressing iNOS | ||
Cell Death Differ AMBRA1 promotes intestinal inflammation by antagonizing PP4R1/PP4c mediated IKK dephosphorylation in an autophagy-independent manner |