New ways to study the elusive exosome
Written by Poppy Nathan, Proteintech Field Marketing Representative and PhD Student at the University of Birmingham
Exosomes: From garbage to glory
Exosomes are tiny extracellular vesicles (30-150 nm) which play key roles in immune regulation, tumour growth, and cellular communication. For years they were thought to simply be cellular waste products, yet today they stand at the forefront of cellular research. In 2007, it was discovered by Valadi et al that exosomes can modify recipient cell behaviour through the transfer of functional mRNA and microRNA between cells, which created a new understanding of the importance of these key communicators. Since then, the past decade has seen an explosion of interest in exosome research and they are now implicated in diseases including cancer, diabetes, and neurodegenerative conditions.
Importantly, exosomes have promising therapeutic potential, due to their low immunogenicity, biocompatibility, and their ability to cross the blood brain barrier, making them a potentially invaluable drug-delivery system.Exosomes also contain proteins, DNA/RNA, lipids and other cellular content from their origin cell, making them potentially exciting candidates for disease biomarkers. Despite these promising findings, research remains difficult due to the elusive nature of the exosome. The biggest limitation in both exosome research and their use in diagnostics is the challenge of exosome isolation, measurement and characterisation, which is why novel methods to research exosomes are vital.
Why is Exosome Isolation So Hard?
A big problem in current exosome research is the need for exosome isolation or enrichment, using methods which are often difficult, expensive, and can lead to sample biases. The gold standard method of exosome isolation has been ultracentrifugation, in which the sample undergoes a series of spin steps to remove varying sizes of cells and debris, leaving only the smallest nanoparticles. As centrifugation is based on size separation, the nanoparticles left over also include lipoproteins and are not specifically exosomes. Quantification and characterisation of these nanoparticles then has to be performed separately, using a mixture of electron microscopy, nanoparticle tracking analysis, immunoblotting, or high resolution flow cytometry, all of which are time-consuming and require a level of expertise.
Novel Methods of Exosome Isolation: The Exoview™
In order to overcome these difficulties, novel technologies are available which selectively capture and isolate exosomes. The Exoview™ is an affinity-based technology which allows for specific capture, quantification and characterisation of different populations of exosomes all from the same sample. It works by capturing exosomes on a chip coated in antibodies against extracellular vesicle (EV)-specific surface markers, namely CD63, CD9 and CD81. Once captured, exosome size is then measured using Single Particle Interferometric Reflectance Imaging Sensor (SP-IRIS) technology, which can detect extracellular vesicles (EVs) as small as 50nm in diameter. The EVs are then stained for up to 3 fluorescent markers against surface proteins, or can be permeabilised and probed for intracellular proteins and cargo.
This piece of technology is incredibly exciting to the EV field as it allows capture of exosomes from samples such as plasma and serum without needing to purify first. This also means much less sample is needed for analysis, which has significant promises for disease research where patient samples are limited. The downside of the Exoview™ (as with most novel technologies!) is it is very expensive to run; a standard tetraspanin chip which contains 16 sample spots will set you back $800, and the chips themselves have a very short shelf life of one month, limiting the potential for this technology to be widely implicated.
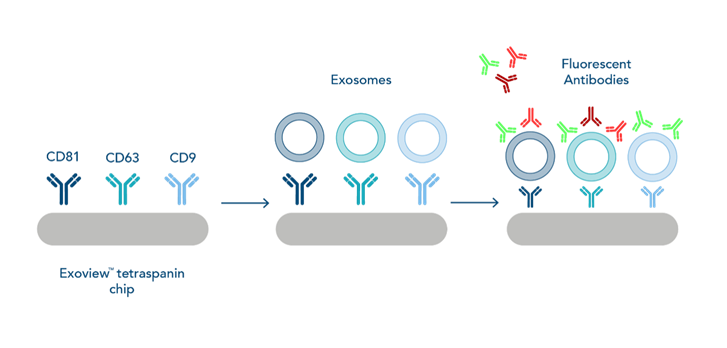 |
| Figure 1. Schematic outlining the workflow of the Exoview™ |
Antibodies In Exosome Research
Antibodies against exosomal markers are essential for identification and characterisation of exosomes. Exosome location, protein expression, and morphology can be revealed using antibodies in western blots, flow cytometry, immunofluorescence staining and immunoaffinity capture techniques. The Exoview™ utilises antibodies in two different ways - both for capture of exosomes using antibodies against surface markers, and for staining to visualise different populations of exosomes. This highlights the need for antibodies which are specific for exosomes and sensitive enough to pick up the whole population. Proteintech offers a range of antibodies which are all manufactured and validated in house. Find out more about the antibodies available from Proteintech here
| Alix | CD63 | TSG101 | CD44 |
| CD9 | CD81 | Caveolin-1 | Rab5 |
| CD24 | CD82 | CD41 |
Read more about the Exoview™
-
Local administration of stem cell-derived extracellular vesicles in a thermoresponsive hydrogel promotes a pro-healing effect in a rat model of colo-cutaneous post-surgical fistula - Berger, A. et al. (2021)
-
Diverse populations of extracellular vesicles with opposite functions during herpes simplex virus 1 infection - Dogrammatzis, C. et al. (2020)
-
Spinal cord injury alters microRNA and CD81+ exosome levels in plasma extracellular nanoparticles with neuroinflammatory potential - Khan, N. Z. et al. (2020)
-
Exosomes and GPI-anchored proteins: Judicious pairs for investigating biomarkers from body fluids - Vidal, M. (2020)
-
Cytokine profiling in serum-derived exosomes isolated by different methods – Jung, HH et al., 2020
References
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659. doi:10.1038/ncb1596
Kusuma G, Lim R. Characterisation of extracellular vesicles surface markers and co-expression studies with single particle interferometric imaging platform. Cytotherapy. 2020;22(5):S50-S51. doi:10.1016/j.jcyt.2020.03.063
Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science (80- ). 2020;367(6478). doi:10.1126/science.aau6977
Xing Y, Cheng Z, Wang R, Lv C, James TD, Yu F. Analysis of extracellular vesicles as emerging theranostic nanoplatforms. Coord Chem Rev. 2020;424:213506. doi:10.1016/j.ccr.2020.213506
Characterize Exosomes | Extracellular Vesicles | NanoView Biosciences. Accessed March 29, 2021. https://www.nanoviewbio.com/
