How to choose the right secondary antibody?
How to choose the best secondary antibody for your application.
Summary
- Secondary antibodies bind to the primary antibody rather than the target directly, amplifying the signal and adding flexibility across different detection methods.
- The secondary must match the host species of your primary antibody — For example, a rabbit primary requires an anti-rabbit secondary.
- For Western blot, HRP-conjugated secondaries are standard; for immunofluorescence and flow cytometry, fluorescently labelled secondaries are used instead.
- Polyclonal secondaries offer higher sensitivity for low-abundance targets, while monoclonal secondaries deliver greater specificity and batch-to-batch consistency.
- For IHC and tissue-based applications, F(ab')2 fragment secondaries are preferred as they reduce non-specific background and penetrate tissue more effectively.
Introduction to Secondary Antibodies
Secondary antibodies facilitate the indirect detection of target molecules by binding to the primary antibody, which in turn binds to the target antigen. Secondary antibodies are generally labeled with a tag to facilitate detection or purification. Indirect detection using conjugated secondary antibodies offers several advantages over the use of directly conjugated primary antibodies, including:
- Increased sensitivity – Multiple secondary antibodies binding to a single primary antibody result in signal amplification.
- Flexibility – The same secondary antibody can be used to detect multiple targets by pairing with multiple primary antibodies.
- More versatile – The same primary antibody can be paired with various secondary antibodies depending on the intended application (e.g., HRP conjugates for western blot and fluorescent conjugates for IF/FC).
- Easily available – Secondary antibodies against most species are widely available in a variety of conjugate options from multiple commercial suppliers.
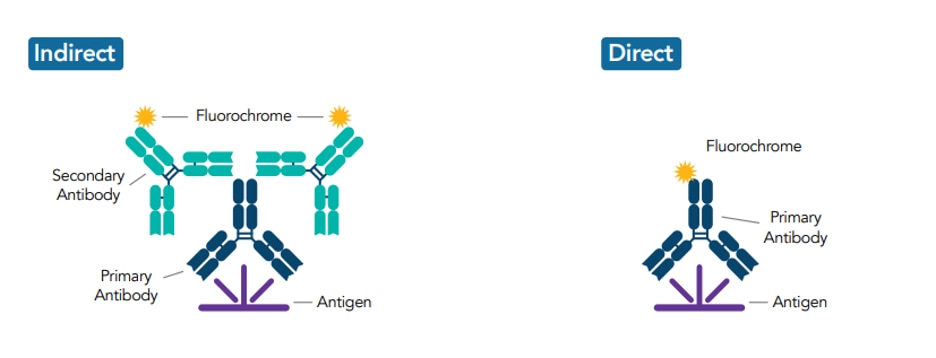 Primary antibodies can be detected by using either conjugated secondary antibodies (indirect detection) or by using directly conjugated primary antibodies (direct detection).
Primary antibodies can be detected by using either conjugated secondary antibodies (indirect detection) or by using directly conjugated primary antibodies (direct detection).
Proteintech offers a comprehensive portfolio of secondary antibodies for detecting unconjugated primary antibodies in a wide range of applications including western blot, ELISA, immunofluorescence, immunohistochemistry, and flow cytometry.
Explore All Secondary Antibodies
Choosing a Secondary Antibody
The choice of appropriate secondary antibody in an application depends on multiple factors, as highlighted below, to ensure highly sensitive and accurate detection of the target antigen.
Target specificity
The secondary antibody must be raised against the host species of the primary antibody. For example, if the primary antibody was generated in mouse, the secondary must be anti-mouse and raised in a host species other than mouse (e.g., goat anti-mouse secondary antibody). In addition, the secondary antibody should be specific for the primary antibody class or subclass. Since most polyclonal primary antibodies are raised against isotype IgG, the secondary antibody used should recognize both IgG heavy and light chains (anti-IgG H+L). Monoclonal primary antibodies on the other hand are raised against specific IgG subclasses, thereby requiring subclass-specific secondary antibodies for their detection.
Clonality
Traditionally, antibodies have been classified as polyclonal or monoclonal. While polyclonal secondary antibodies are a heterogenous population of antibodies recognizing different epitopes on the same antigen, monoclonal secondary antibodies are a homogenous antibody population that recognizes the same epitope on an antigen.
Proteintech’s Multi-rAb Recombinant Secondary Antibodies are the newest generation of secondary antibodies that combine the high sensitivity of polyclonals with the superior specificity of monoclonals. Multi-rAb secondaries consist of mixtures of recombinant monoclonal antibodies that recognize multiple complementary epitopes on the same IgG.
Learn more about Multi-rAb Recombinant Secondary Antibodies
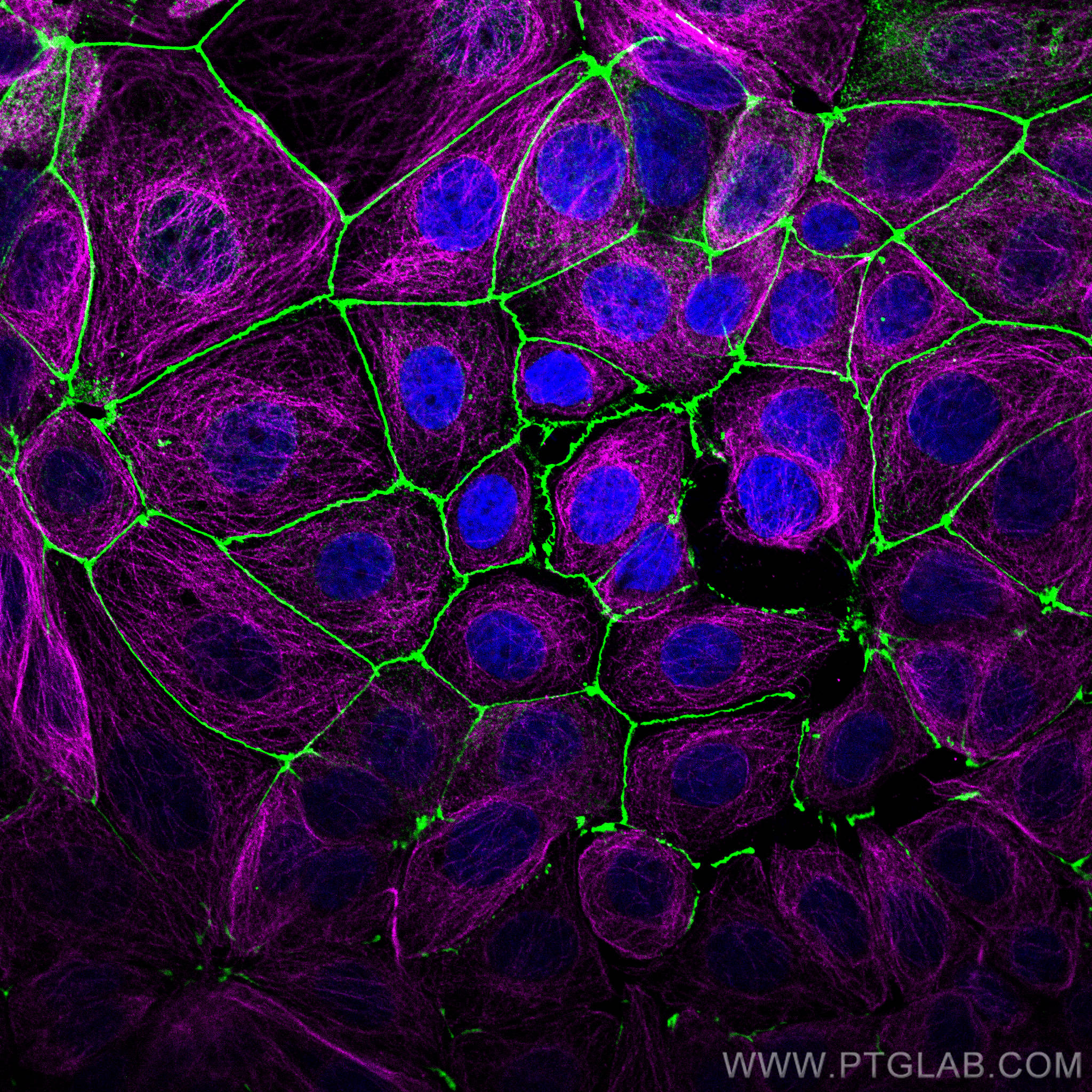
IF analysis of MCF-7 stained with anti-ZO1 antibody (21773-1-AP, green) and anti-alpha tubulin antibody (66031-1-Ig, magenta). Multi-rAb CoraLite® Plus 488 and 647 were used for detection. |
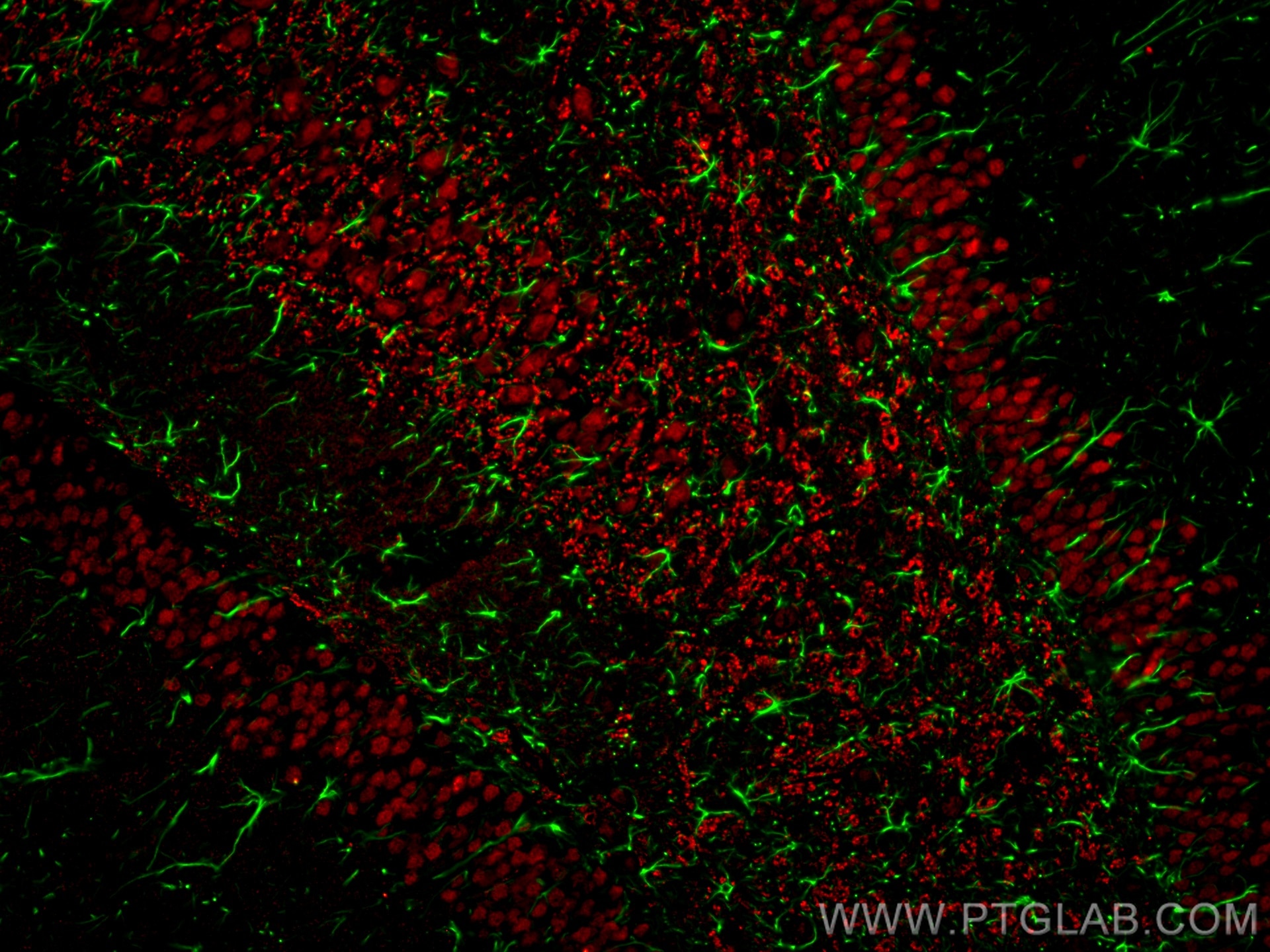
IF analysis of rat brain FFPE section stained with anti-GFAP antibody (16825-1-AP, green) and anti-NeuN antibody (66836-1-Ig, red). Multi-rAb CoraLite® Plus 488 and 594 were used for detection. |
Host species
The host species refers to the animal in which the secondary antibody was raised. It is important to ensure that the secondary antibody host is different from the host animal in which the primary antibody was raised. While the majority of primary antibodies are raised in rabbits or mice, goats are the most widely used host animals for commercially available secondary antibodies.
Antibody format
Secondary antibodies are available in various formats including whole antibodies and antibody fragments. The choice of the secondary antibody format is dependent on the intended application. For example, antibody fragments including F(ab’)2 and Fab’ are preferred over whole antibodies for IHC and IF in tissue samples due to their smaller size allowing for better tissue penetration.
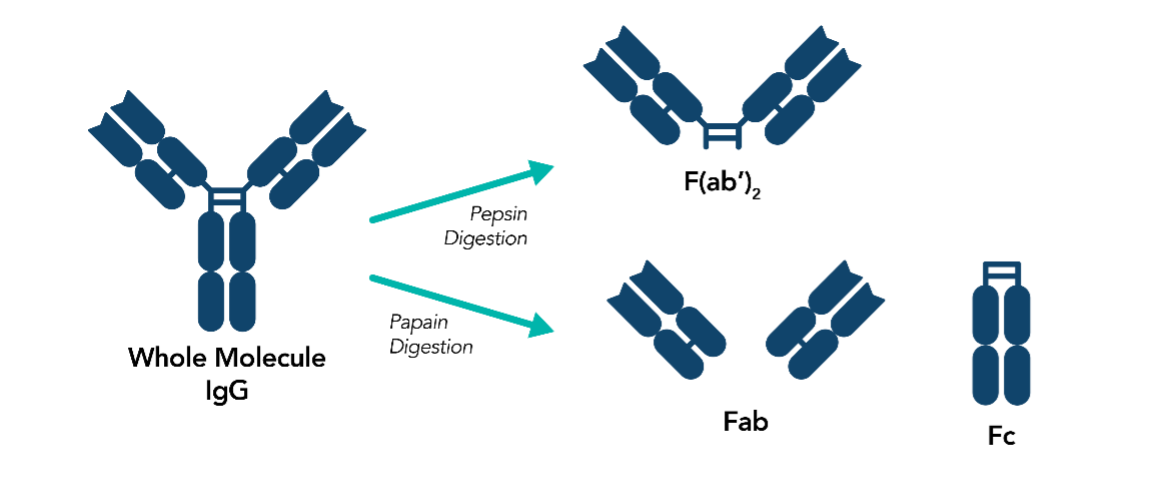 Whole IgG antibodies are digested with pepsin and papain to generate F(ab’)2 and Fab’ fragment antibodies, respectively.
Whole IgG antibodies are digested with pepsin and papain to generate F(ab’)2 and Fab’ fragment antibodies, respectively.
Proteintech offers a novel class of recombinant secondary antibodies known as Nano-secondary reagents. Nano-Secondary reagents consist of recombinantly generated alpaca nanobodies/VHHs that can achieve higher resolution and better tissue penetration due to their smaller size.
 Left: primary antibody (dark blue) & Nano-secondary (green) complex. Right: conventional polyclonal secondary antibody (light blue) complex.
Left: primary antibody (dark blue) & Nano-secondary (green) complex. Right: conventional polyclonal secondary antibody (light blue) complex.
Explore All Nano-Secondary Reagents
Type of conjugate
The choice of the conjugate primarily depends on the application and the detection method. The table below highlights suggested secondary antibody conjugates based on the intended application.
| Application | Secondary Antibody Conjugates |
|---|---|
| Western blot | • Enzyme labeled (HRP/AP) • Fluorescently labeled |
| IHC | • Enzyme labeled (HRP/HRP-polymer) • Biotinylated secondaries • Fluorescently labeled |
| Immunofluorescence | • Fluorescently labeled • HRP-labeled for TSA-based detection |
| Flow cytometry | • Fluorescently labeled • Enzyme labeled • Biotinylated (for use with streptavidin) |
| ELISA | • Biotin labeled • Enzyme labeled |
Cross-adsorbed antibodies
Cross-adsorbed secondary antibodies have undergone additional purification steps to eliminate cross-reactivity against unintended IgGs from non-target species. Proteintech’s Multi-rAb Recombinant Secondary Antibodies are a mixture of recombinant monoclonal antibodies selected for minimal cross-reactivity, achieving the same enhanced specificity as highly cross-adsorbed traditional polyclonals.
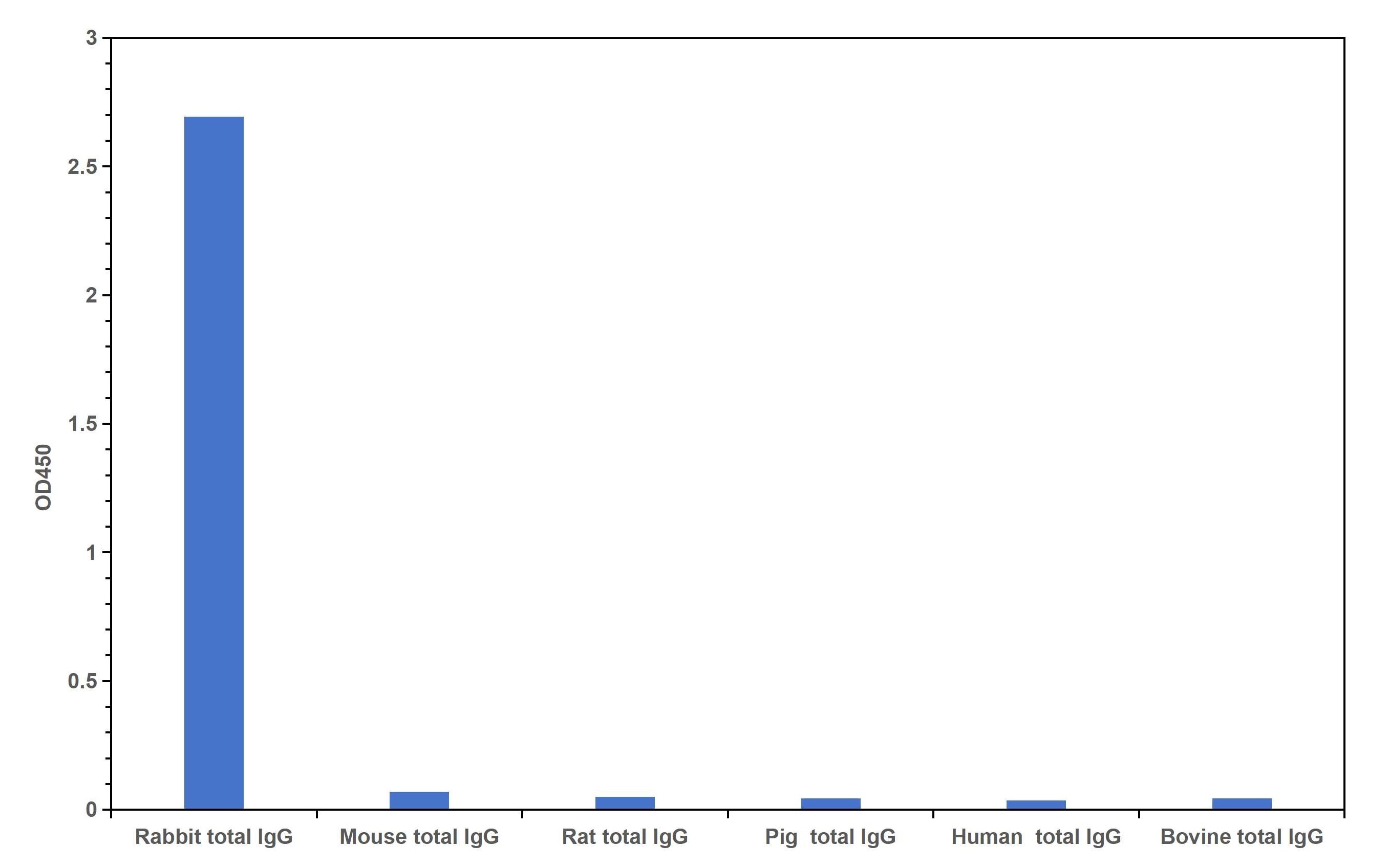 Multi-rAb HRP-Goat Anti-Rabbit Recombinant Secondary Antibody (RGAR001) is highly specific for rabbit IgG and does not react with other tested species.
Multi-rAb HRP-Goat Anti-Rabbit Recombinant Secondary Antibody (RGAR001) is highly specific for rabbit IgG and does not react with other tested species.
