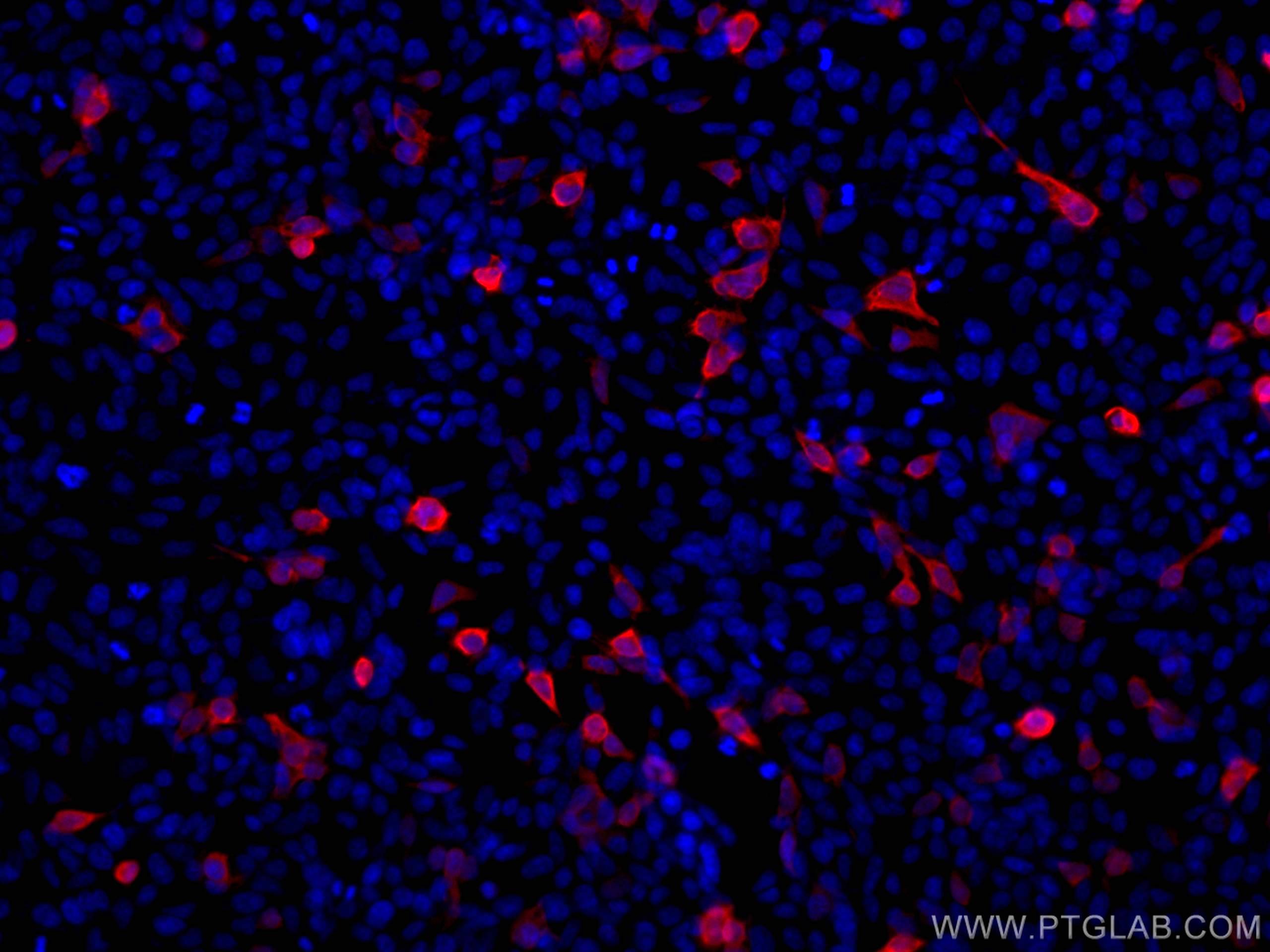
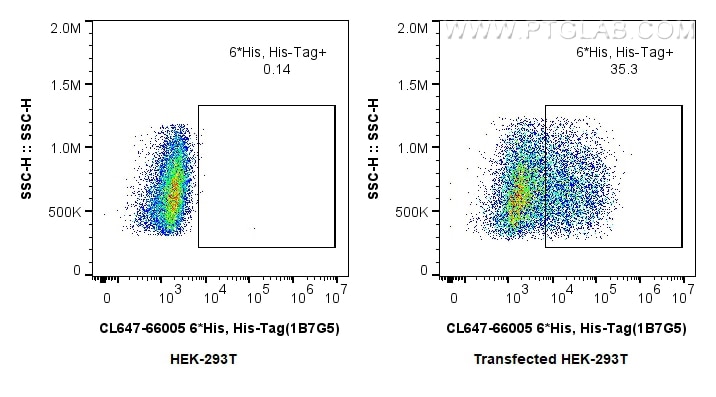
Tested Applications
| Positive IF/ICC detected in | Transfected HEK-293 cells |
| Positive FC (Intra) detected in | Transfected HEK-293 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Immunofluorescence (IF)/ICC | IF/ICC : 1:250-1:1000 |
| Flow Cytometry (FC) (INTRA) | FC (INTRA) : 0.80 ug per 10^6 cells in a 100 µl suspension |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| IF | See 3 publications below |
Product Information
CL647-66005 targets 6*His, His-Tag in IF/ICC, FC (Intra) applications and shows reactivity with recombinant protein samples.
| Tested Reactivity | recombinant protein |
| Cited Reactivity | human |
| Host / Isotype | Mouse / IgG1 |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
Peptide Predict reactive species |
| Full Name | 6*His, His-Tag |
| Calculated Molecular Weight | 0.84 kDa |
| Gene Symbol | |
| Gene ID (NCBI) | |
| RRID | AB_2920271 |
| Conjugate | CoraLite® Plus 647 Fluorescent Dye |
| Excitation/Emission Maxima Wavelengths | 654 nm / 674 nm |
| Excitation Laser | Red Laser (633 nm) |
| Form | Liquid |
| Purification Method | Protein G purification |
| UNIPROT ID | HISTAG |
| Storage Buffer | PBS with 50% glycerol, 0.05% Proclin300, 0.5% BSA, pH 7.3. |
| Storage Conditions | Store at -20°C. Avoid exposure to light. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. |
Background Information
Protein tags are a protein or peptide sequences located either on the C- or N- terminal of the target protein. His-tag is often used for affinity purification and binding assays. Expressed. The His-tag antibody is a useful tool for monitoring of the His-tagged proteins and recognizes His-tags placed at N-terminal, C-terminal, and internal regions of fusion proteins expressed in bacteria, insect, and mammalian cells. A His-tag (polyhistidine tag) consists of at least six histidine residues that are located at the N- or C-terminus of recombinant proteins. It is commonly used for affinity purification and protein binding experiments.
The recombinant protein has only 6 histidine residues as a tag. Is this sufficient to be detected by your antibody? Is the His-tag antibody able to detect N-terminal and C-terminal His tags?
The His-tag antibody was raised using a 6-His tag peptide as the immunogen, which is ~1 kDa. Some scientists use more than 6 histidine residues as a tag for recombinant proteins to increase affinity to metal ions. A 6-histidine tag is sufficient for specific recognition by the His-tag antibody. It is able to recognize both N-terminal and C-terminal His tags of recombinant proteins produced in various expression systems, including bacteria, yeasts, insect, and mammalian cells.
After purification, I can detect my protein by western blotting using the His-tag antibody but can also see additional bands running lower. What might they represent?
They may represent cleavage products of the recombinant protein. Unspecific protein cleavage can occur during protein production (e.g., in bacteria) or protein purification post the cell lysis steps. Changing expression conditions (temperature, time, induction methods, medium) and using protease inhibitors during purification helps to minimize protein degradation. Additional steps, such as ion exchange, size exclusion, or chromatography, allow isolation of the full-length product.
Can a His-tag antibody be used in applications other than western blotting?
Yes, this antibody has been successfully used for immunofluorescence (IF), immunoprecipitation (IP), and indirect ELISA analysis.
Protocols
| Product Specific Protocols | |
|---|---|
| FC protocol for CL Plus 647 6*His, His-Tag antibody CL647-66005 | Download protocol |
| IF protocol for CL Plus 647 6*His, His-Tag antibody CL647-66005 | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Int J Mol Sci Identifying Exifone as a Dual-Target Agent Targeting Both SARS-CoV-2 3CL Protease and the ACE2/S-RBD Interaction Among Clinical Polyphenolic Compounds | ||
J Virol Herpes simplex virus 1 envelope glycoprotein C shields glycoprotein D to protect virions from entry-blocking antibodies | ||
bioRxiv Herpes Simplex Virus 1 Envelope Glycoprotein C Shields Glycoprotein D to Protect Virions from Entry-Blocking Antibodies | ||
Cell Mol Life Sci Eeukaryotic-like Sppsk1 from Vibrio splendidus AJ01 mediates phagosome escape via inhibiting phagosome acidification and maturation | ||
Nat Prod Bioprospect Exploring anti-SARS-CoV-2 natural products: dual-viral target inhibition by delphinidin and the anti-coronaviral efficacy of deapio platycodin D | ||
J Extracell Vesicles Targeted Intracellular Delivery via Precision Programming of ARRDC1-Mediated Microvesicles. |
Reviews
The reviews below have been submitted by verified Proteintech customers who received an incentive for providing their feedback.
FH Zhi (Verified Customer) (03-22-2024) | Good and easy to use.
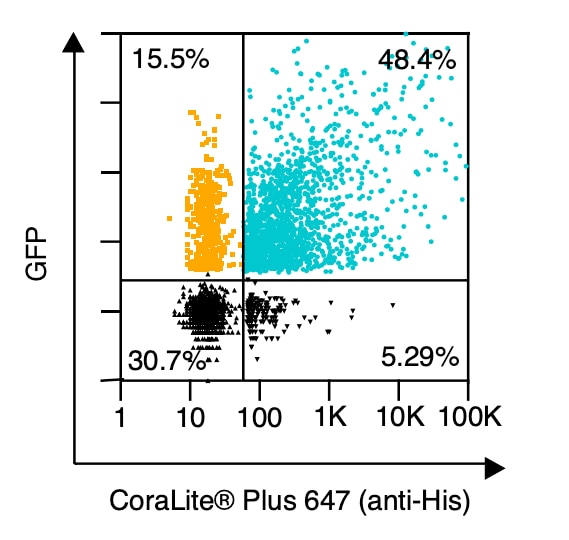 |