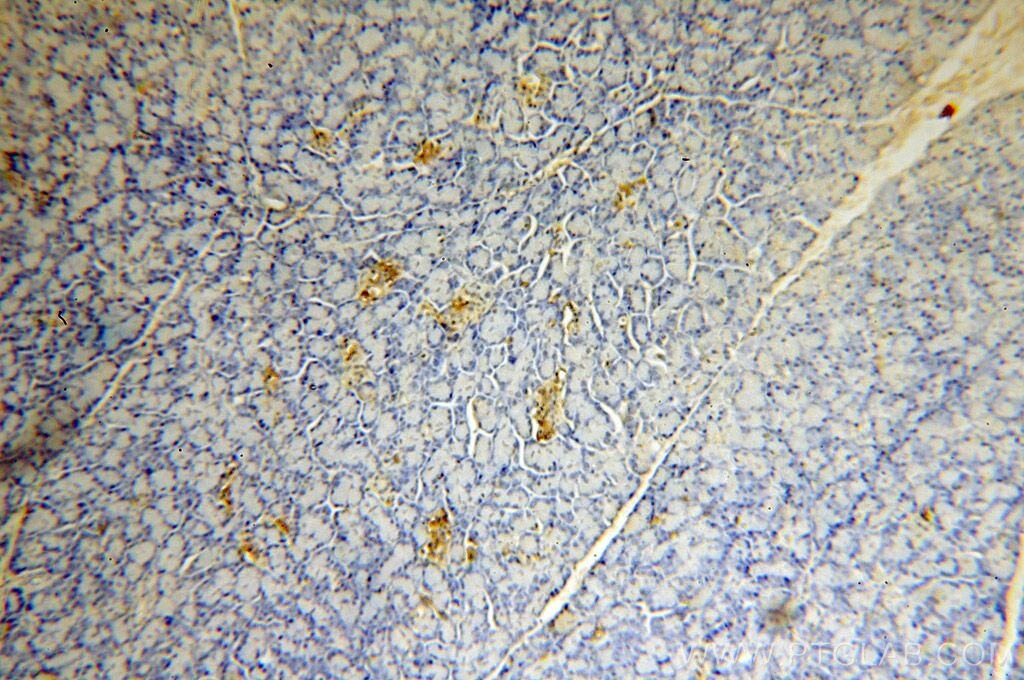
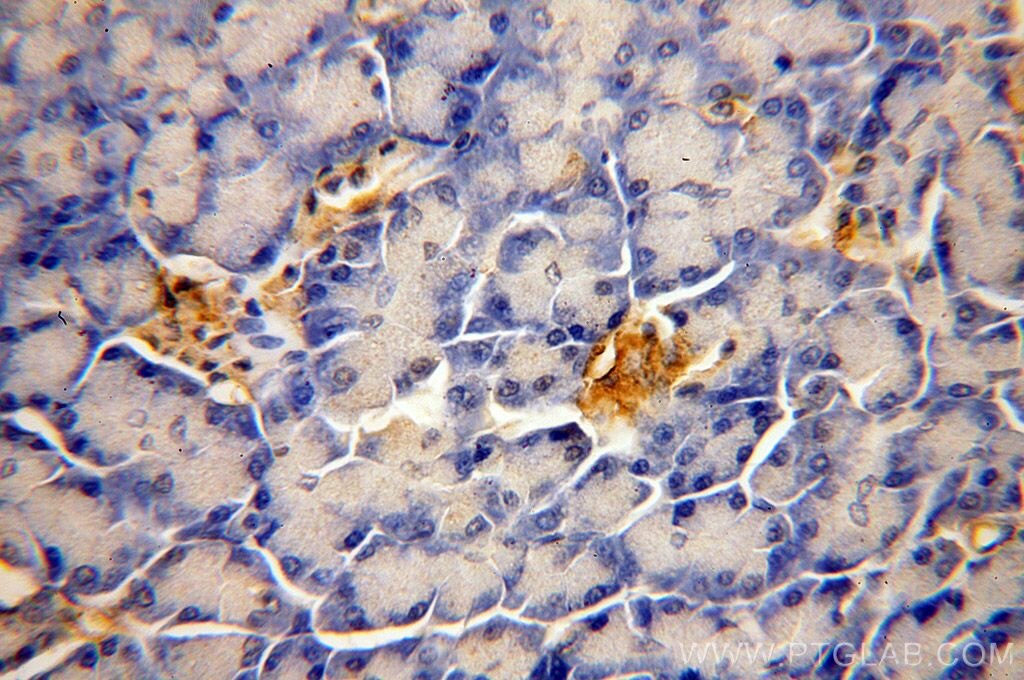
Tested Applications
| Positive IHC detected in | human pancreas tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
Recommended dilution
| Application | Dilution |
|---|---|
| Immunohistochemistry (IHC) | IHC : 1:20-1:200 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Product Information
18313-1-AP targets ERO1L in IHC, ELISA applications and shows reactivity with human, mouse samples.
| Tested Reactivity | human, mouse |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
Peptide Predict reactive species |
| Full Name | ERO1-like (S. cerevisiae) |
| Calculated Molecular Weight | 54 kDa |
| GenBank Accession Number | BC008674 |
| Gene Symbol | ERO1L |
| Gene ID (NCBI) | 30001 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q96HE7 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
ERO1L, also named as ERO1-alpha, is an essential oxidoreductase that oxidizes proteins in the endoplasmic reticulum to produce disulfide bonds. It acts by oxidizing directly P4HB/PDI isomerase through a direct disulfide exchange. It does not act as a direct oxidant of folding substrate, but relies on P4HB/PDI to transfer oxidizing equivalent. Associates with ERP44 but not with GRP54, demonstrating that it does not oxidize all PDI related proteins and can discriminate between PDI and related proteins. Its reoxidation probably involves electron transfer to molecular oxygen via FAD. Glutathione may be required to regulate its activity in the endoplasmic reticulum. It may be responsible for a significant proportion of reactive oxygen species (ROS) in the cell, thereby being a source of oxidative stress. It is required for the folding of immunoglobulin proteins. Responsible for the release of the unfolded cholera toxin from reduced P4HB/PDI in case of infection by V.cholerae, thereby playing a role in retrotranslocation of the toxin. This antibody has no cross reaction to ERO1L.