Tested Applications
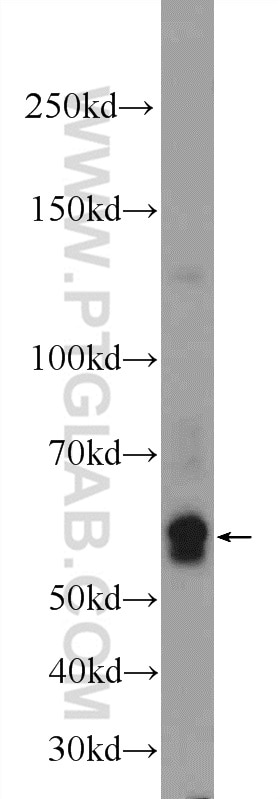
| Positive WB detected in | Tunicamycin treated HepG2 cells |
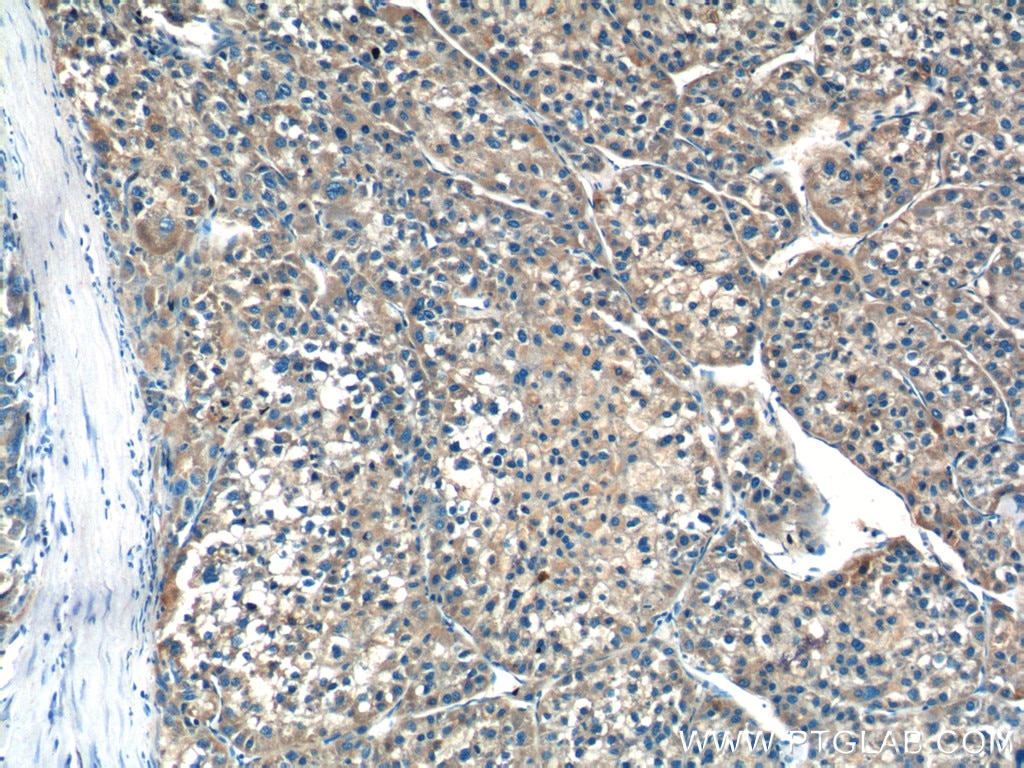
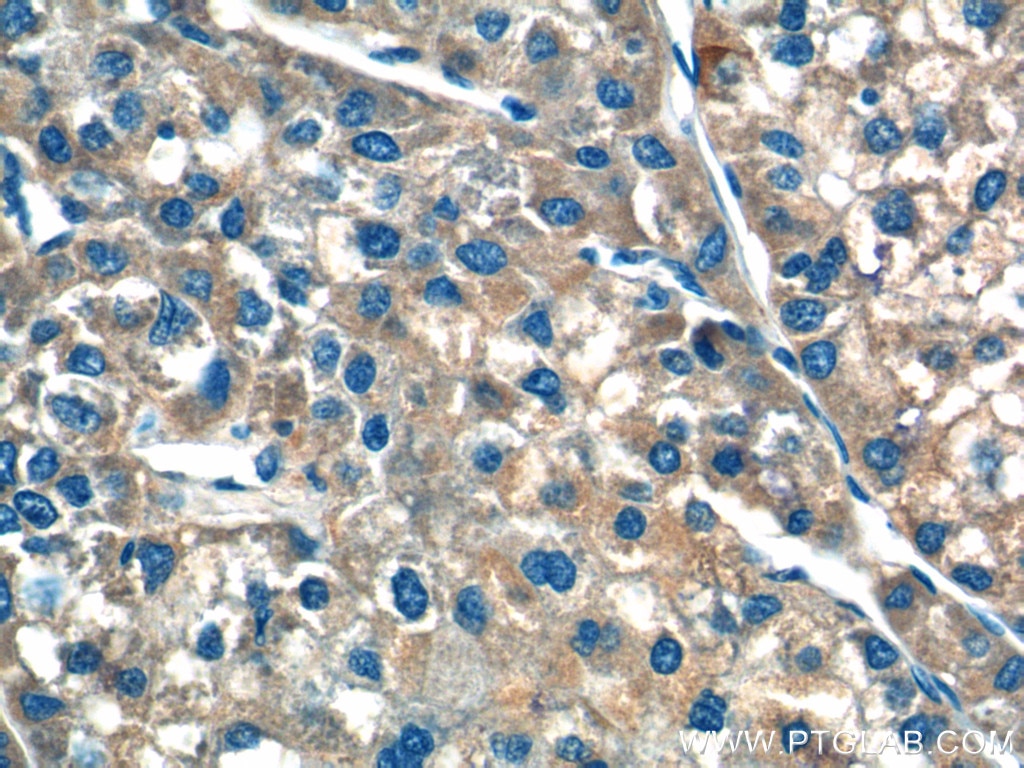
| Positive IHC detected in | human liver cancer tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:500-1:1000 |
| Immunohistochemistry (IHC) | IHC : 1:50-1:500 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 2 publications below |
| WB | See 3 publications below |
| IF | See 1 publications below |
Product Information
21481-1-AP targets Factor IX/PTC in WB, IHC, IF, ELISA applications and shows reactivity with human samples.
| Tested Reactivity | human |
| Cited Reactivity | human, mouse, pig |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag15723 Product name: Recombinant human F9 protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 129-395 aa of BC109215 Sequence: ELDVTCNIKNGRCEQFCKNSADNKVVCSCTEGYRLAENQKSCEPAVPFPCGRVSVSQTSKLTRAETVFPDVDYVNSTEAETILDNITQSTQSFNDFTRVVGGEDAKPGQFPWQVVLNGKVDAFCGGSIVNEKWIVTAAHCVETGVKITVVAGEHNIEETEHTEQKRNVIRIIPHHNYNAAINKYNHDIALLELDEPLVLNSYVTPICIADKEYTNIFLKFGSGYVSGWGRVFHKGRSALVLQYLRVPLVDRATCLRSTKFTIYNNMF Predict reactive species |
| Full Name | coagulation factor IX |
| Calculated Molecular Weight | 461 aa, 52 kDa |
| Observed Molecular Weight | 56 kDa |
| GenBank Accession Number | BC109215 |
| Gene Symbol | F9 |
| Gene ID (NCBI) | 2158 |
| RRID | AB_2878866 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | P00740 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
The F9 gene encodes coagulation factor IX, which circulates as an inactive zymogen until proteolytic release of its activation peptide allows it to assume the conformation of an active serine protease. Its role in the blood coagulation cascade is to activate factor X (F10) through interactions with calcium, membrane phospholipids, and factor VIII (F8). Factor IX and factor X both consist of 2 polypeptide chains referred to as the L (light) and H (heavy) chains. The H chain bears a structural resemblance to the polypeptide chain of the pancreatic serine protease trypsin (PRSS1). The L chain is covalently linked to the H chain by a single disulfide bond.
Protocols
| Product Specific Protocols | |
|---|---|
| IHC protocol for Factor IX/PTC antibody 21481-1-AP | Download protocol |
| WB protocol for Factor IX/PTC antibody 21481-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
EMBO Mol Med CRISPR/Cas9-mediated somatic correction of a novel coagulator factor IX gene mutation ameliorates hemophilia in mouse. | ||
Front Neurosci Serum Exosomal Proteins F9 and TSP-1 as Potential Diagnostic Biomarkers for Newly Diagnosed Epilepsy. | ||
Cell Death Dis Genome wide CRISPR/Cas9 screen identifies the coagulation factor IX (F9) as a regulator of senescence.
| ||
Vet Res TRIM28 regulates the coagulation cascade inhibited by p72 of African swine fever virus
|