Tested Applications
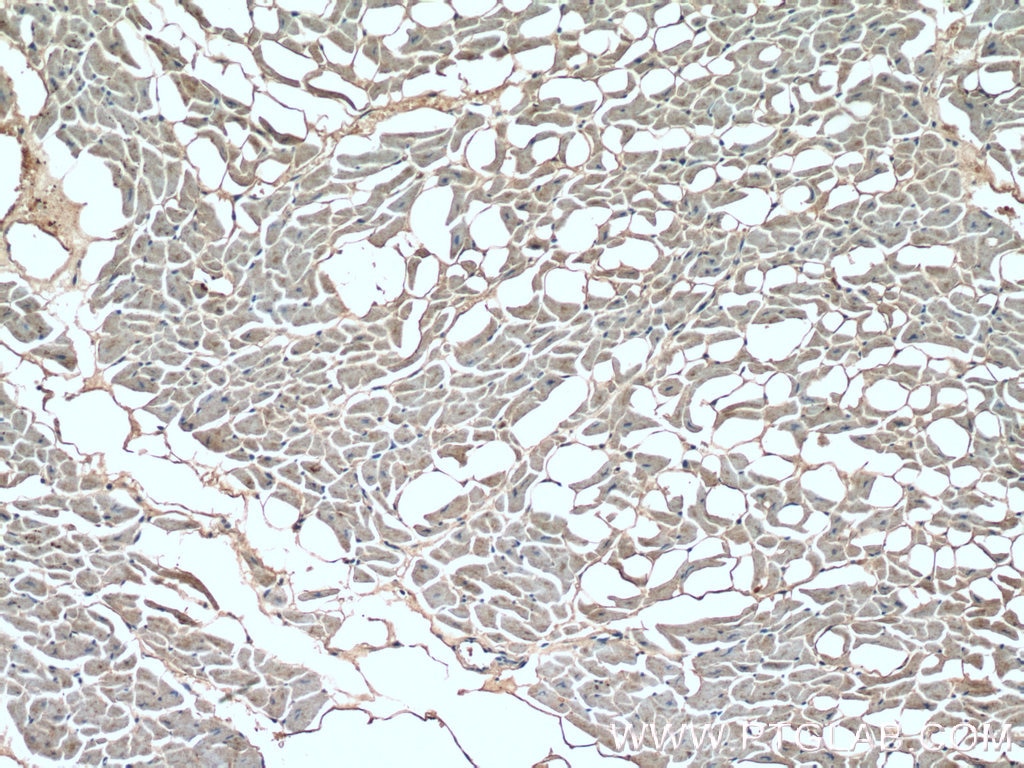
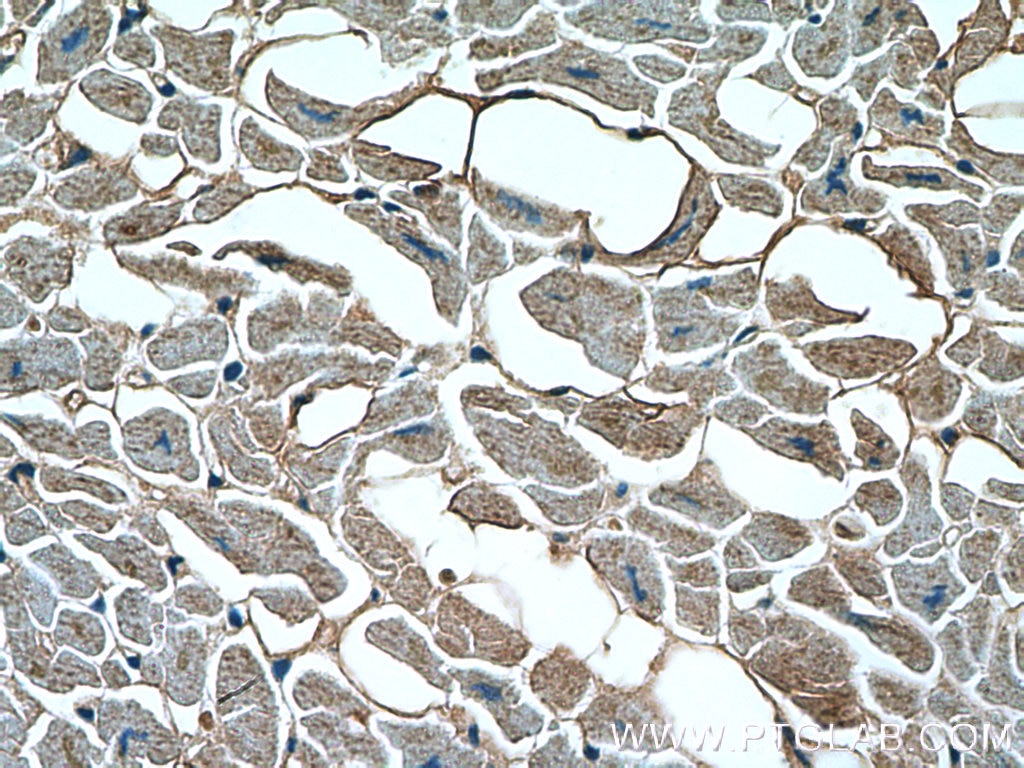
| Positive IHC detected in | human heart tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
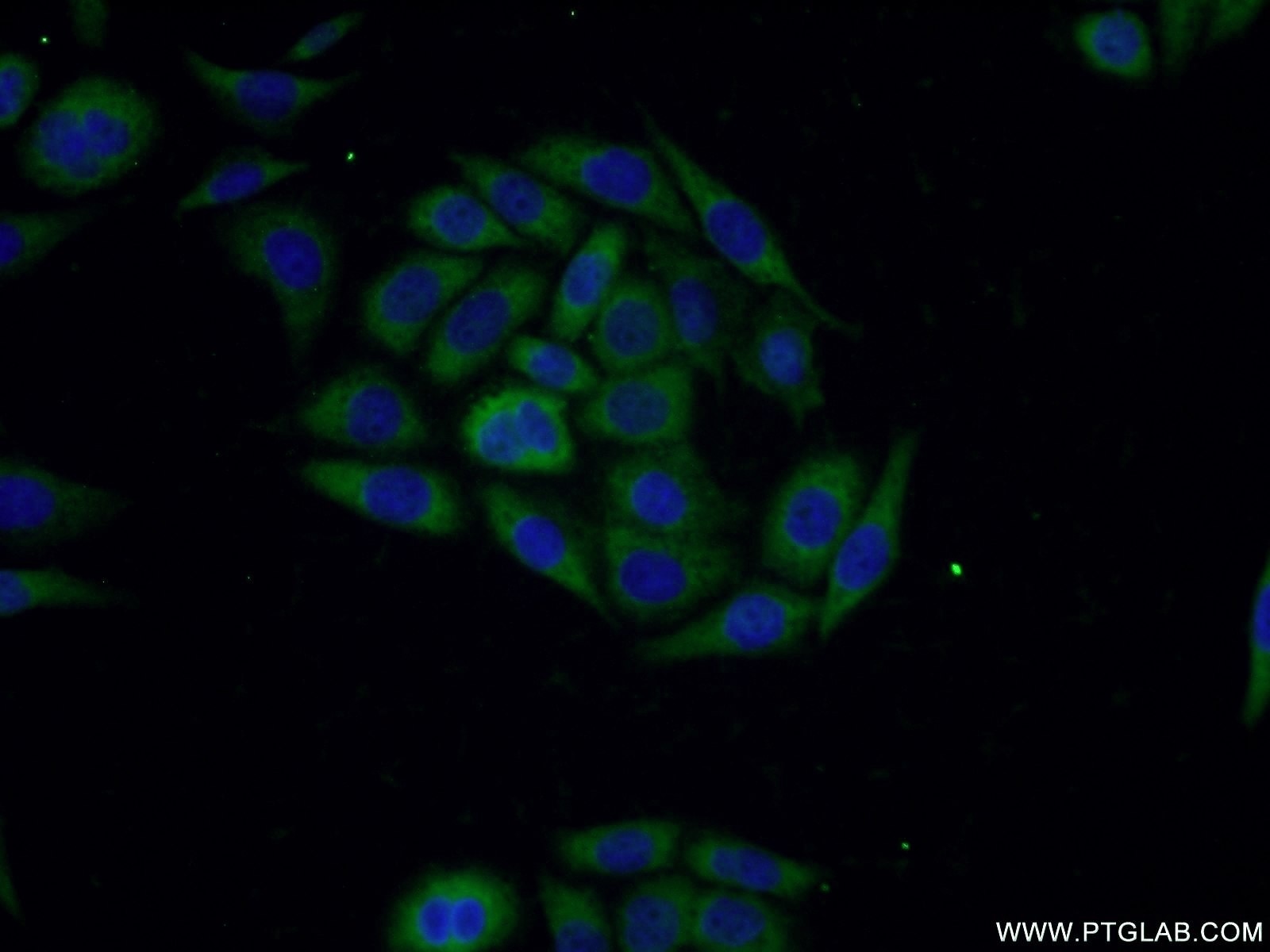
| Positive IF/ICC detected in | PC-3 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Immunohistochemistry (IHC) | IHC : 1:50-1:500 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:20-1:200 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| WB | See 9 publications below |
| IF | See 5 publications below |
Product Information
18395-1-AP targets SLN in WB, IHC, IF/ICC, ELISA applications and shows reactivity with human samples.
| Tested Reactivity | human |
| Cited Reactivity | squirrel |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag13176 Product name: Recombinant human SLN protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1-31 aa of BC005261 Sequence: MGINTRELFLNFTIVLITVILMWLLVRSYQY Predict reactive species |
| Full Name | sarcolipin |
| Calculated Molecular Weight | 31 aa, 4 kDa |
| GenBank Accession Number | BC005261 |
| Gene Symbol | SLN |
| Gene ID (NCBI) | 6588 |
| RRID | AB_2286622 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | O00631 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
SLN, also named as Sarcolipin, belongs to the sarcolipin family. It is associated with calcium ATPase SERCA1. It inhibits SERCA pumps. SLN, a key regulator of cardiac sarco(endo)plasmic reticulum (SR) Ca(2+) ATPase, is predominantly expressed in atria and mediates β-adrenergic responses. SLN can self-assembly to dimer and oligomer for playing importantphysiological function.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for SLN antibody 18395-1-AP | Download protocol |
| IHC protocol for SLN antibody 18395-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
EBioMedicine Maternal exercise intergenerationally drives muscle-based thermogenesis via activation of apelin-AMPK signaling. | ||
J Biol Chem Identification of Small Ankyrin 1 as a Novel Sarco(endo)plasmic Reticulum Ca2+-ATPase 1 (SERCA1) Regulatory Protein in Skeletal Muscle. | ||
Skelet Muscle Ca(2+) permeation and/or binding to CaV1.1 fine-tunes skeletal muscle Ca(2+) signaling to sustain muscle function. | ||
J Biol Chem Interactions between small ankyrin 1 and sarcolipin coordinately regulate activity of the sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA1). | ||
Exp Cell Res Effect of sarcolipin-mediated cell transdifferentiation in sarcopenia-associated skeletal muscle fibrosis. |