IHC Sample Visualization
Learn the difference between direct and indirect visualization and when to use each method.
Immunohistochemistry Sample Visualization
After incubation with the primary antibody, the staining can be visualized using different detection methods. The detection technique can be direct or indirect and the signal generation can be chromogenic or fluorescent.
Direct Versus Indirect Detection
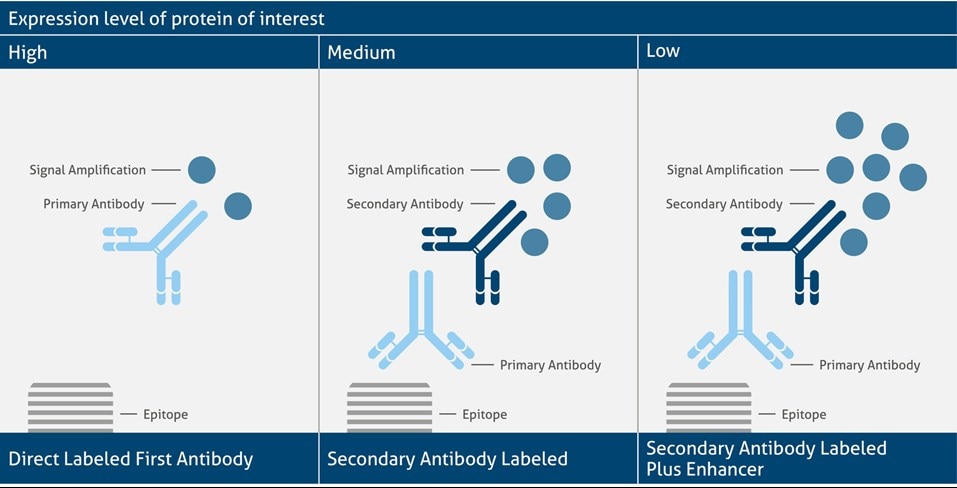
In direct detection, the primary antibody contains the label. On the other hand, in indirect detection, the secondary is labeled. The choice of detection method depends on the expression level of the antigen. As primary labeled antibodies show a comparable low signal generation, they are suitable for highly expressed antigens. Direct detection is also advantageous for multi-labeling experiments. Medium expressed antigens instead show the best signal when analyzed via a secondary labeled antibody that helps to amplify the signal intensity. Indirect labeling, however, always shows a higher background noise. For very low-expressed proteins the indirect detection plus an enhancer (e.g., avidin, streptavidin) helps to further amplify the signal. This type of detection often needs some additional optimization steps to get the best signal-to-noise ratio.
Different Detection Systems and Signal Amplification
Chromogenic Vs Fluorescent Signal Generation
A signal is generated by fluorophores or enzymes. The detection method and available microscope determine the choice of label.
If working with fluorescent labels, the fluorophore can be conjugated either to the primary or secondary antibody. Especially for multi-color staining experiments, fluorescent labels are beneficial for detecting different cellular compartments at the same time. A disadvantage of fluorophores is their short lifetime. Thus, enzyme labels are often the preferred choice for visualization of tissue stainings.
If working with enzymatic labels, the enzyme is attached to the primary or secondary antibody and then forms an insoluble colored product when an organic substrate is added. The most commonly used enzymatic labels are horseradish peroxidase and alkaline phosphatase.