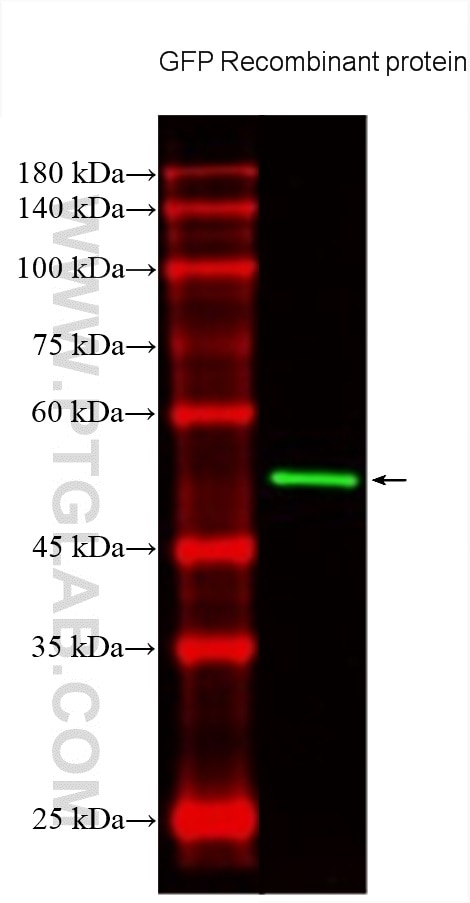
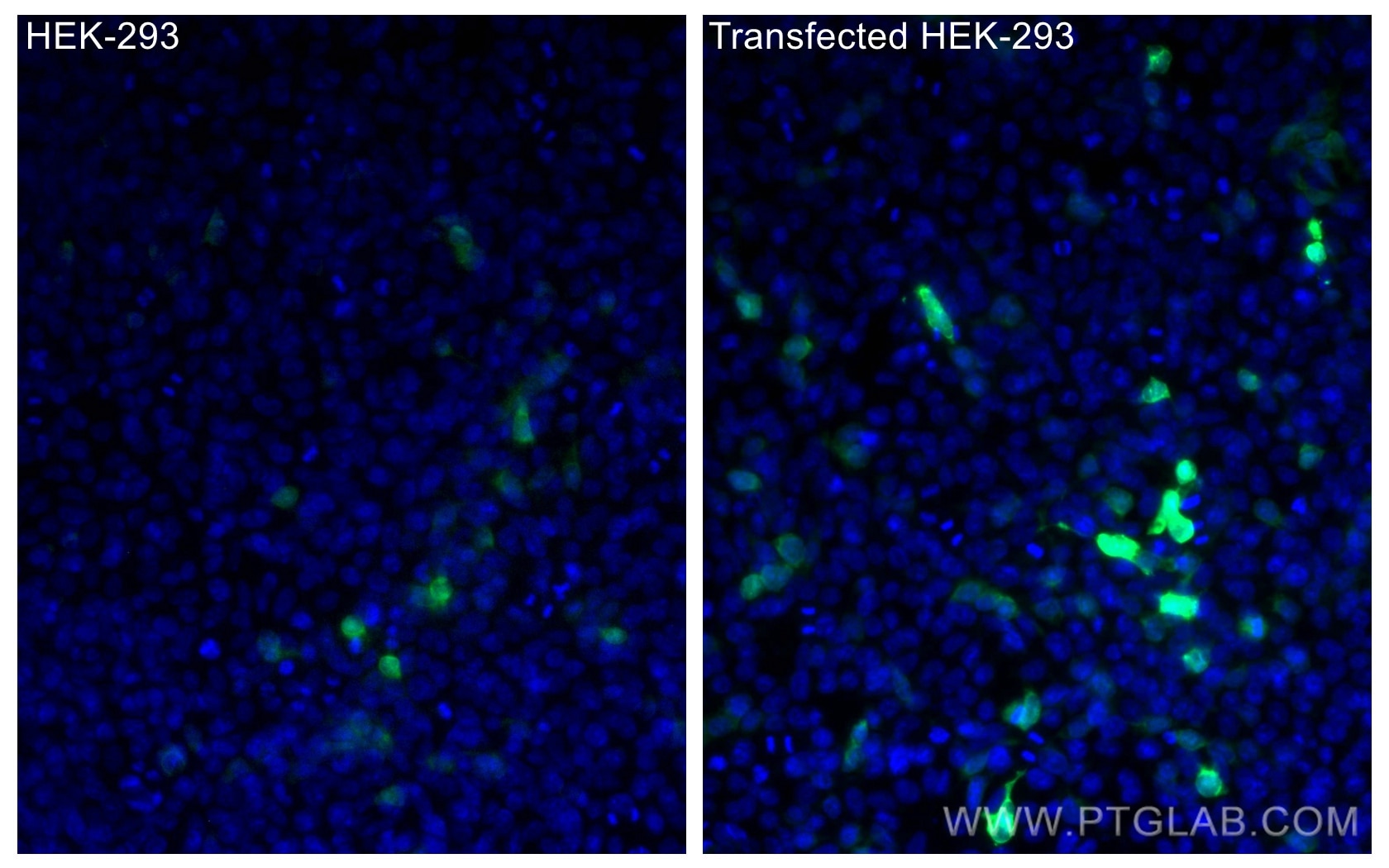
Tested Applications
| Positive WB detected in | Recombinant protein |
| Positive IF/ICC detected in | Transfected HEK-293 cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:2000-1:10000 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:50-1:500 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Product Information
CL488-66002 targets GFP tag in WB, IF/ICC applications and shows reactivity with recombinant protein samples.
| Tested Reactivity | recombinant protein |
| Host / Isotype | Mouse / IgG2a |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag2128 Product name: Recombinant aequorea victoria GFP tag protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 1-238 aa of M62653 Sequence: MSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFICTTGKLPVPWPTLVTTFSYGVQCFSRYPDHMKQHDFFKSAMPEGYVQERTIFFKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNYNSHNVYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGPVLLPDNHYLSTQSALSKDPNEKRDHMVLLEFVTAAGITHGMDELYK Predict reactive species |
| Full Name | GFP tag |
| Calculated Molecular Weight | 26 kDa |
| GenBank Accession Number | M62653 |
| Gene Symbol | |
| Gene ID (NCBI) | |
| RRID | AB_2919276 |
| Conjugate | CoraLite® Plus 488 Fluorescent Dye |
| Excitation/Emission Maxima Wavelengths | 493 nm / 522 nm |
| Excitation Laser | Blue laser (488 nm) |
| Form | Liquid |
| Purification Method | Protein A purification |
| UNIPROT ID | P42212 |
| Storage Buffer | PBS with 50% glycerol, 0.05% Proclin300, 0.5% BSA, pH 7.3. |
| Storage Conditions | Store at -20°C. Avoid exposure to light. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. |
Background Information
Green Fluorescent Proteins (GFPs) encompass a diverse range of proteins carrying a green chromophore, originating from various species and forming different protein lineages.
Wildtype GFP consists of 238 amino acid residues (26.9 kDa). GFP was first identified in the jellyfish Aequorea victoria. It emits green light with a peak wavelength of 509 nm upon excitation by blue light at 395 nm.
When fused with other proteins, GFP serves as a versatile reporter protein e.g. for quantifying expression levels or facilitates visualization of subcellular localization through fluorescence microscopy.
This antibody is a mouse (IgG2a) monoclonal antibody against GFP reactive to GFP, eGFP, eYFP, YFP and CFP. This antibody is labeled with CoraLite Plus 488.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for CL Plus 488 GFP tag antibody CL488-66002 | Download protocol |
| WB protocol for CL Plus 488 GFP tag antibody CL488-66002 | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |