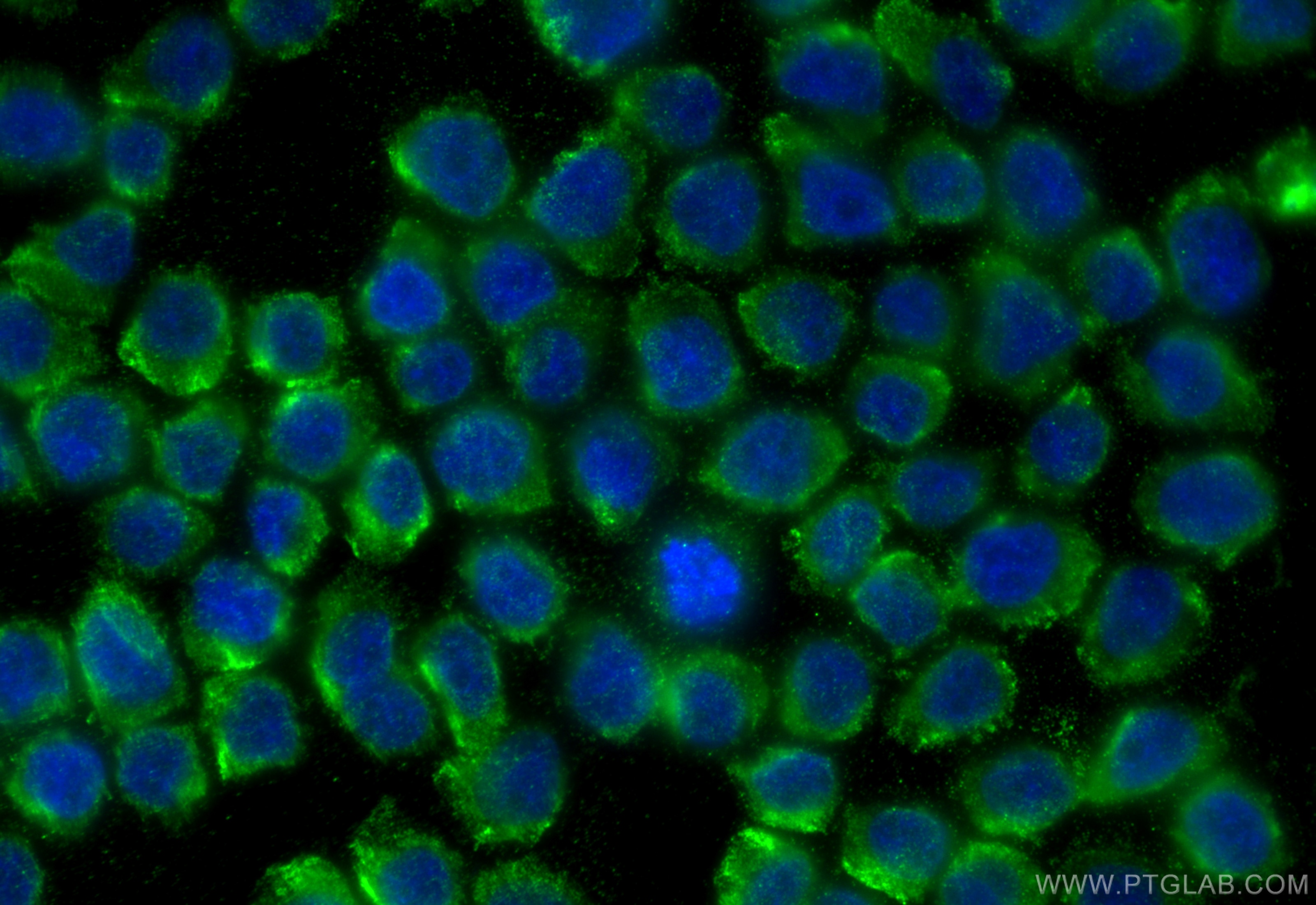
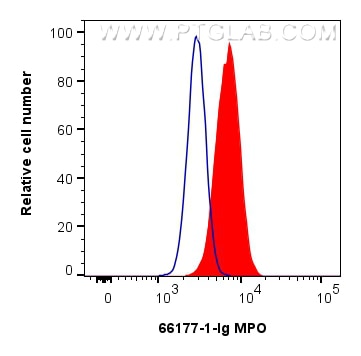
Tested Applications
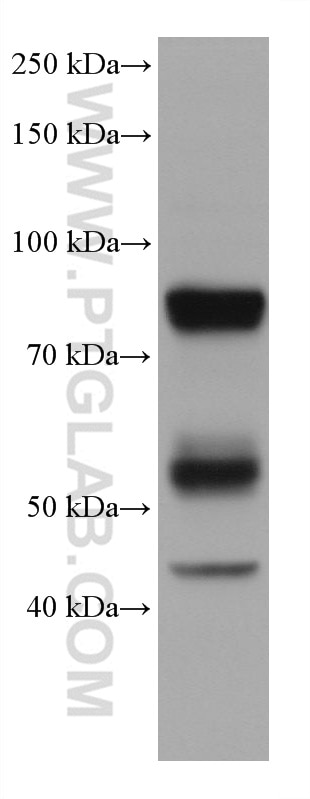
| Positive WB detected in | HL-60 cells |
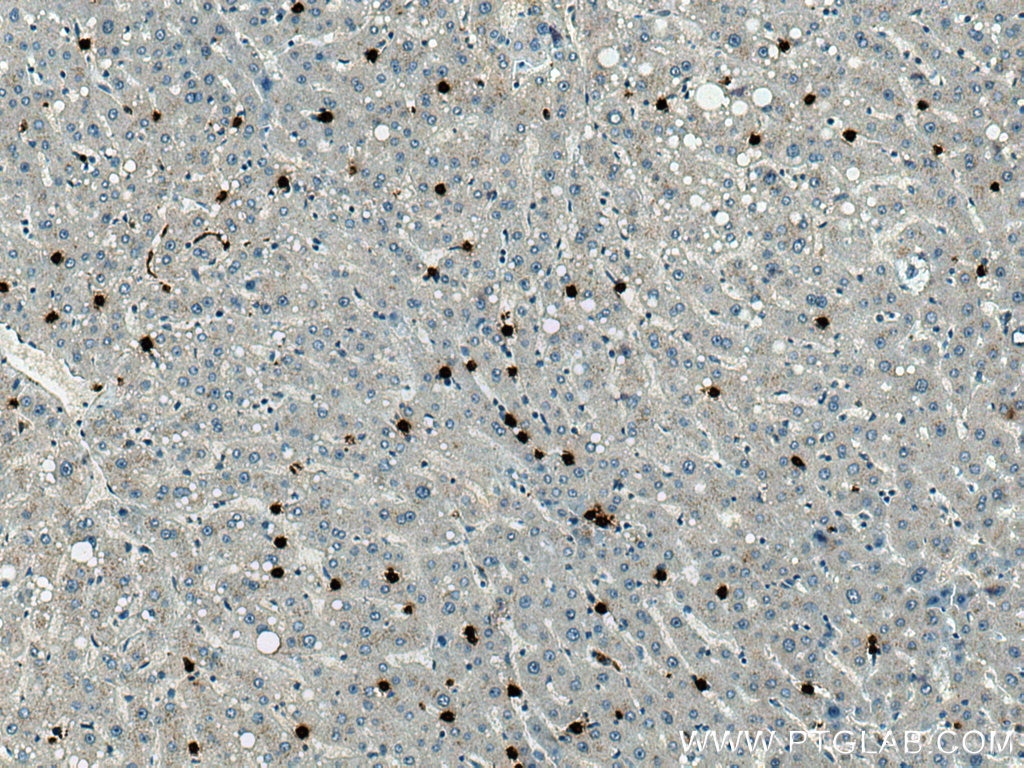
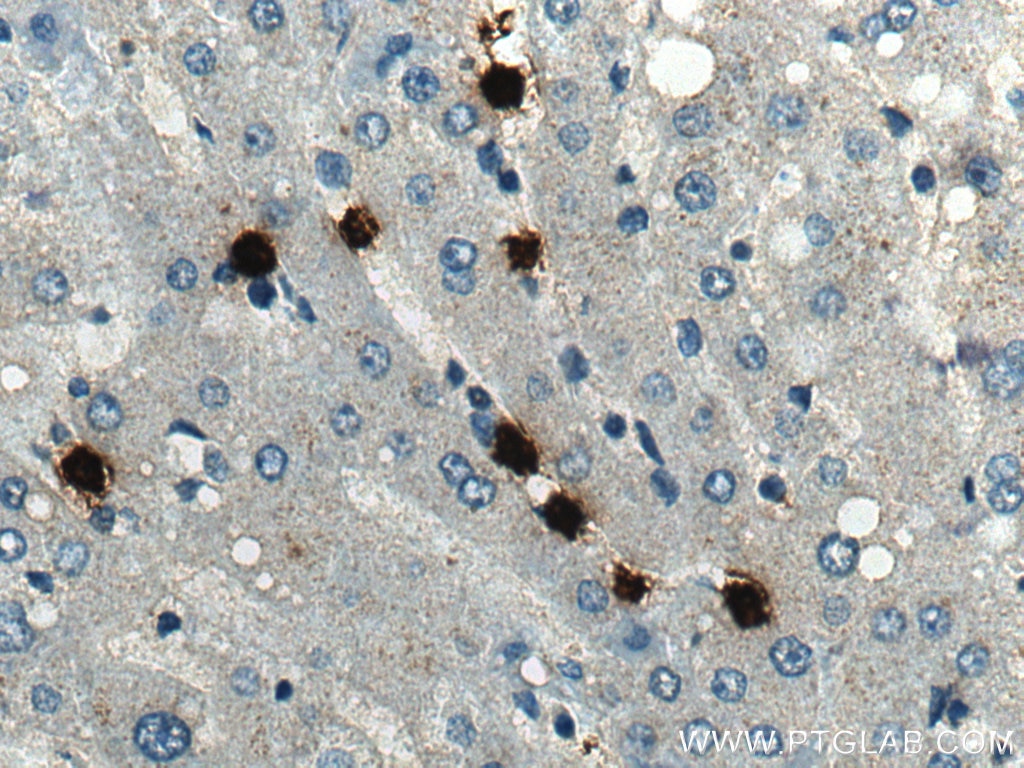
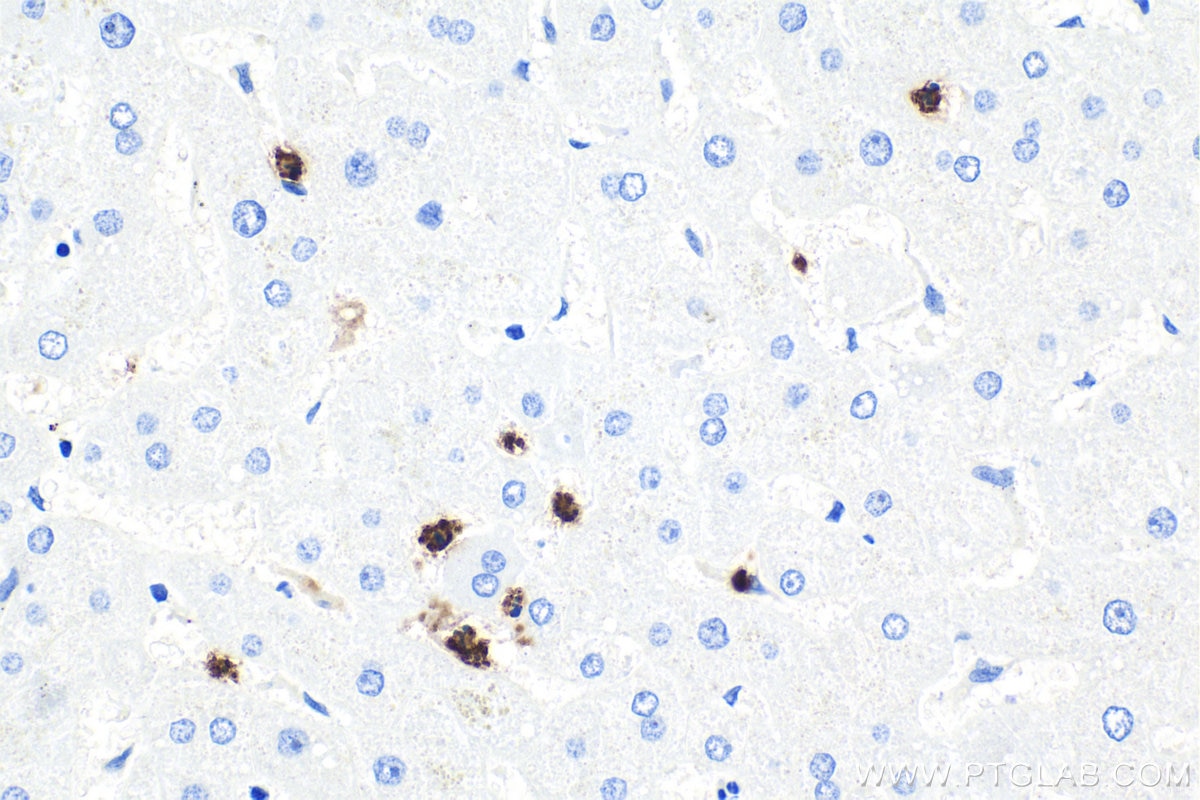
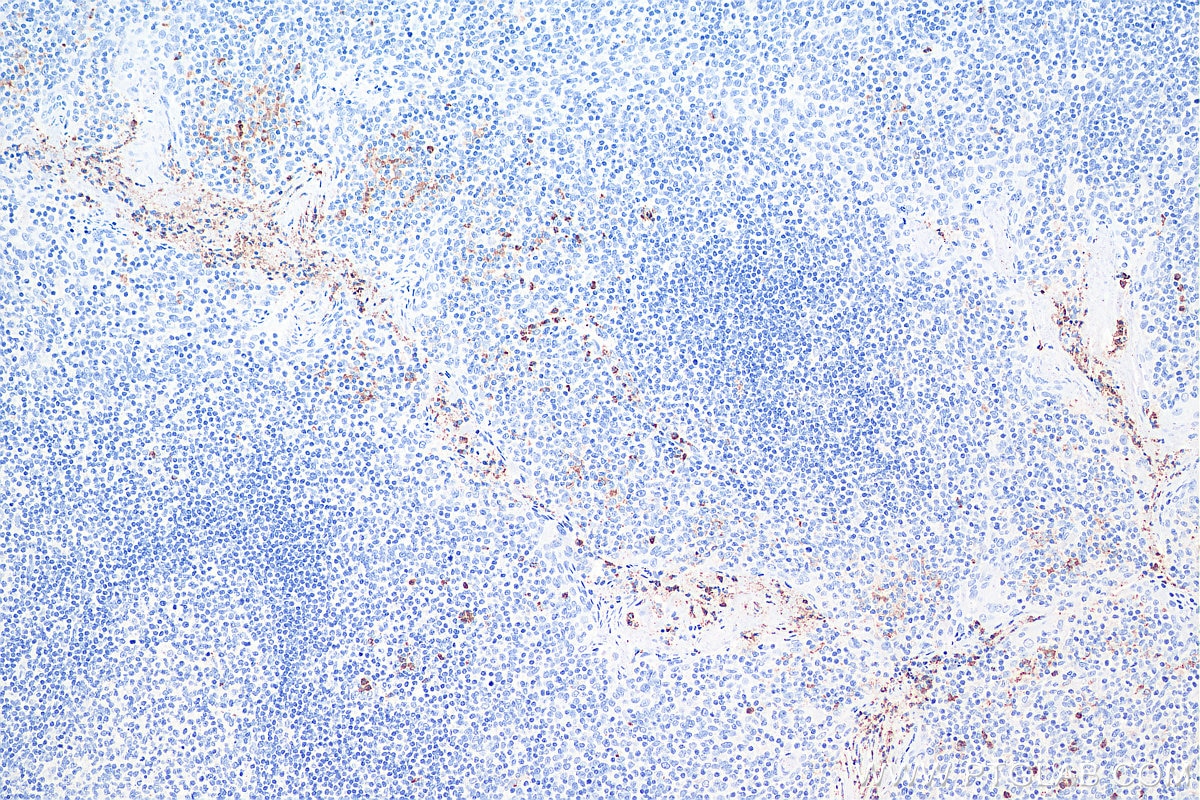
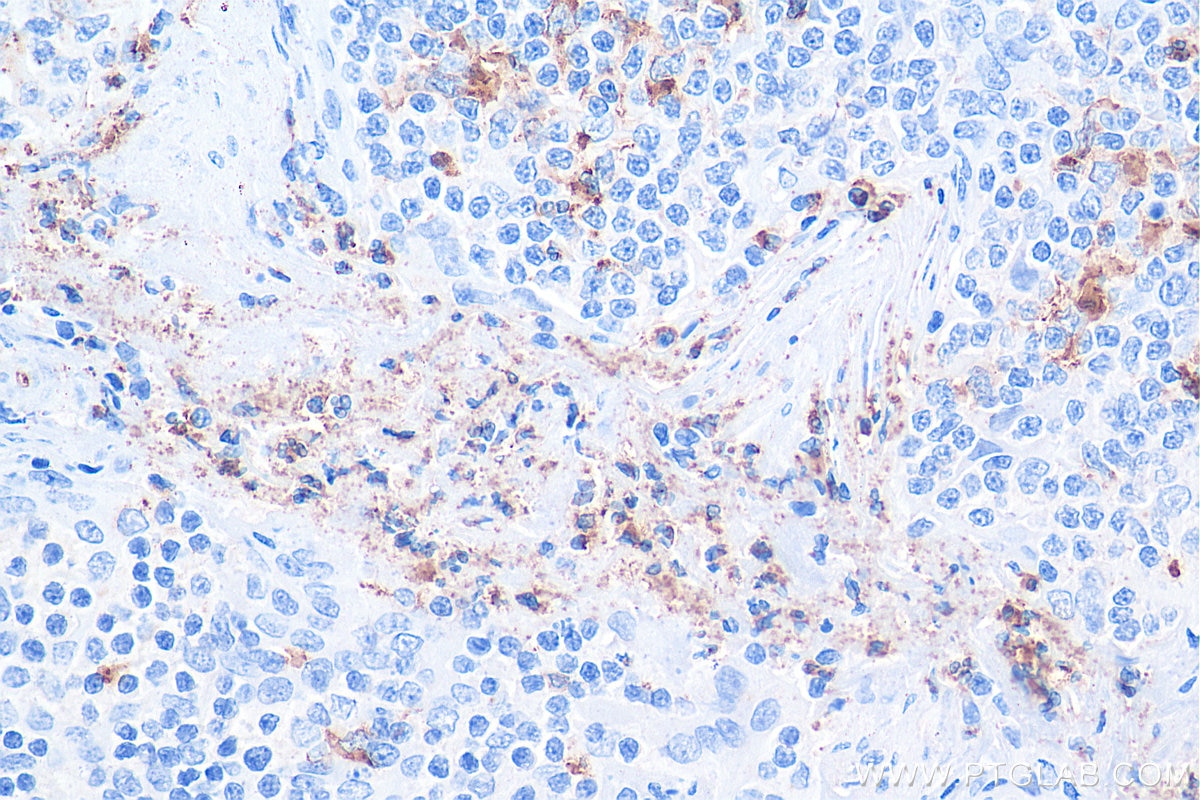
| Positive IHC detected in | human liver tissue, human tonsillitis tissue Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
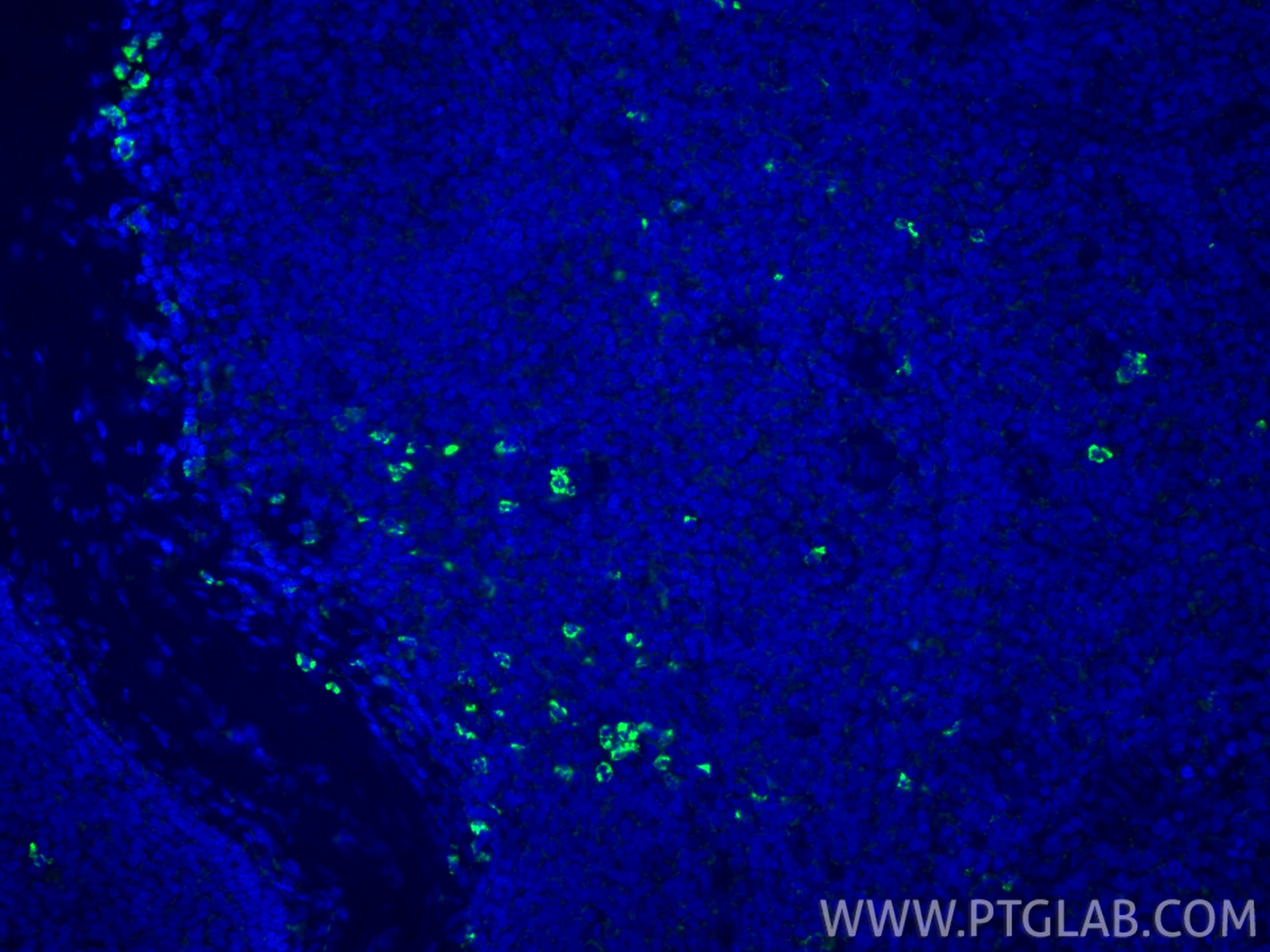
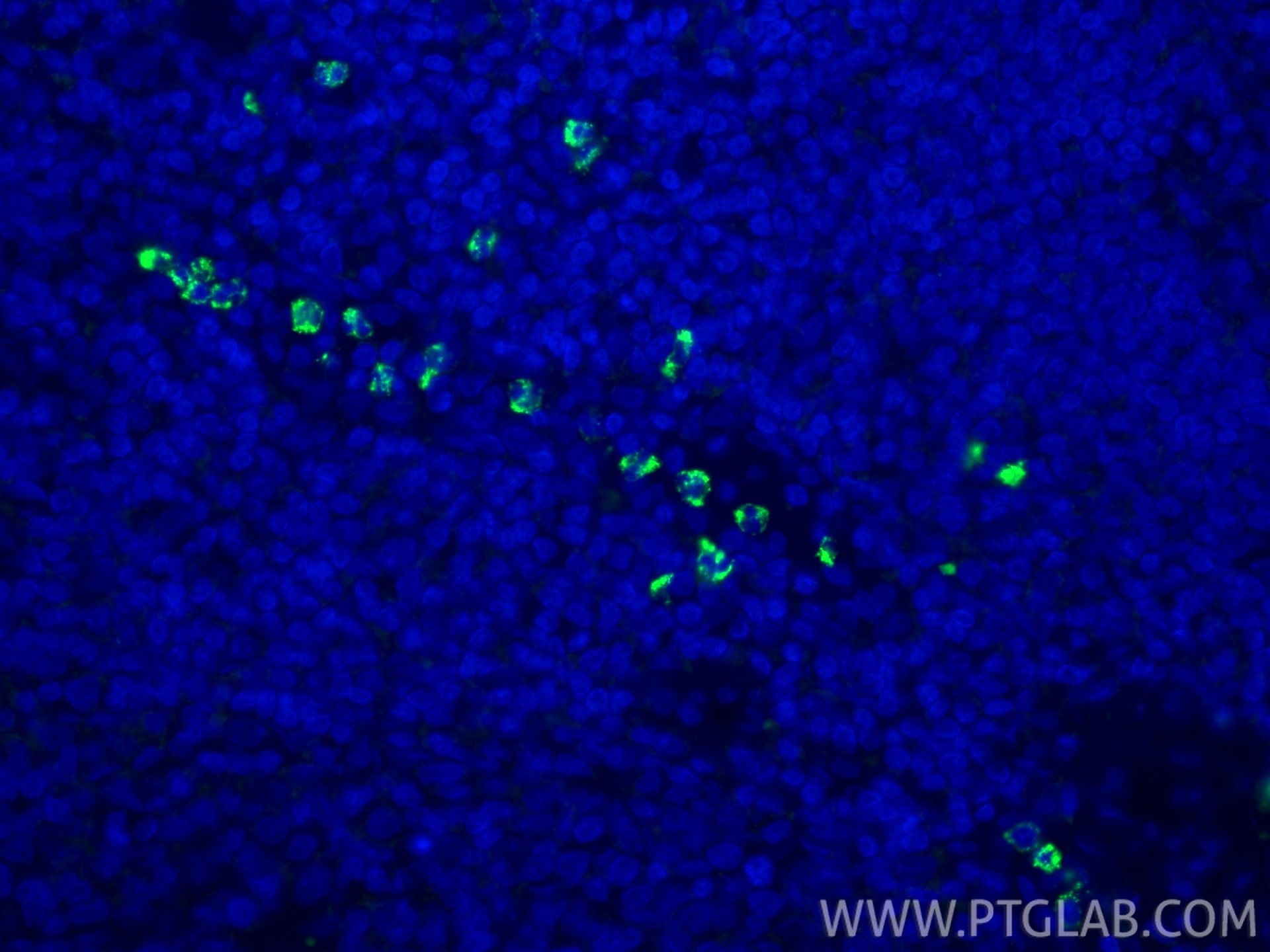
| Positive IF-P detected in | human tonsillitis tissue |
| Positive IF/ICC detected in | HL-60 cells |
| Positive FC (Intra) detected in | HL-60 cells |
Not recommend for IHC about rat sample. Mouse monoclonal antibodies of IgA isotype can be detected with "anti-mouse IgG (H+L)" secondary antibodies.
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:1000-1:8000 |
| Immunohistochemistry (IHC) | IHC : 1:400-1:1600 |
| Immunofluorescence (IF)-P | IF-P : 1:200-1:800 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:200-1:800 |
| Flow Cytometry (FC) (INTRA) | FC (INTRA) : 0.25 ug per 10^6 cells in a 100 µl suspension |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 2 publications below |
| WB | See 22 publications below |
| IHC | See 20 publications below |
| IF | See 53 publications below |
| IP | See 1 publications below |
| ELISA | See 1 publications below |
Product Information
66177-1-Ig targets MPO in WB, IHC, IF/ICC, IF-P, FC (Intra), IP, ELISA applications and shows reactivity with human, rat samples.
| Tested Reactivity | human, rat |
| Cited Reactivity | human, rat, bovine, cow |
| Host / Isotype | Mouse / IgA |
| Class | Monoclonal |
| Type | Antibody |
| Immunogen |
CatNo: Ag17564 Product name: Recombinant human MPO protein Source: e coli.-derived, PGEX-4T Tag: GST Domain: 606-657 aa of BC130476 Sequence: CGLPQPETVGQLGTVLRNLKLARKLMEQYGTPNNIDIWMGGVSEPLKRKGRV Predict reactive species |
| Full Name | myeloperoxidase |
| Calculated Molecular Weight | 745 aa, 84 kDa |
| Observed Molecular Weight | 90 kDa |
| GenBank Accession Number | BC130476 |
| Gene Symbol | MPO |
| Gene ID (NCBI) | 4353 |
| RRID | AB_2881572 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Caprylic acid/ammonium sulfate precipitation |
| UNIPROT ID | P05164 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
The MPO gene encodes myeloperoxidase, a lysosomal hemoprotein located in the azurophilic granules of polymorphonuclear (PMN) leukocytes and monocytes. In response to stimulation, MPO is activated into a transient intermediate with potent antimicrobial oxidizing abilities(PMID:17650507). The mRNA is translated into a single protein of 90 kDa, which displays enzymatic activity and undergoes proteolytic maturation into a heavy chain of 59 kDa and a light chain of 13.5 kDa; these subunits then dimerize into the mature tetramer and the mature MPO is a heterotetramer composed of two identical heavy chains and two identical light chains(PMID:12773517). Fragments with molecular masses of 43-47 kDa were formed by autocatalysis during warming in sample buffer (PMID:12960244). The 24-kDa material had a map identical to that of 13.5 kDa subunit and represents a dimer of the 13.5 kDa subunit (PMID:3008892). Defects in MPO are the cause of myeloperoxidase deficiency (MPOD). It has 3 isoforms produced by alternative splicing.
Protocols
| Product Specific Protocols | |
|---|---|
| FC protocol for MPO antibody 66177-1-Ig | Download protocol |
| IF protocol for MPO antibody 66177-1-Ig | Download protocol |
| IHC protocol for MPO antibody 66177-1-Ig | Download protocol |
| WB protocol for MPO antibody 66177-1-Ig | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Nat Commun Overcoming therapeutic resistance in oncolytic herpes virotherapy by targeting IGF2BP3-induced NETosis in malignant glioma | ||
Cell Death Differ NLRX1 mediated impaired microglial phagocytosis of NETs in cerebral ischemia and reperfusion injury | ||
Adv Sci (Weinh) Cancer Cell-Derived Large Extracellular Vesicles Promote Venous Thromboembolism by Activating NETosis Through Delivering CYBA | ||
PLoS Pathog Prion protein signaling induces M2 macrophage polarization and protects from lethal influenza infection in mice. | ||
Int Immunopharmacol Sappanone A enhances hepatocyte proliferation in lipopolysaccharide-induced acute liver injury in mice by promoting injured hepatocyte apoptosis and regulating macrophage polarization | ||
FEBS J A combined model of serum neutrophil extracellular traps, CD8+ T cells, and tumor proportion score provides better prediction of PD-1 inhibitor efficacy in patients with NSCLC |