Validation Data Gallery
Technical Specifications
| GeneID | 1950 |
| Species | Human |
| Expression | HEK293 |
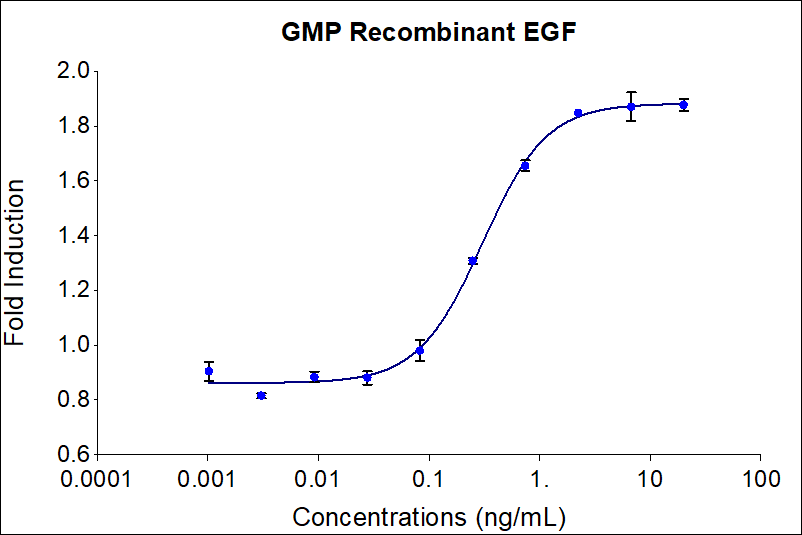
| EC50 | 0.1-0.6 ng/mL |
| Specific Activity | minimally 1.00x106 IU/mg, typically 5.49x106 IU/mg |
| Purity | >95% |
| Endotoxin | <0.1 EU/μg |
| Accession Number | P01133 |
| Molecular Mass | 6 kDa reduced, 10 kDa non-reduced, monomer, glycosylated |
| Formulation | 1x PBS, See Certificate of Analysis for details |
| Species Reactivity | human,monkey |
Stability and Reconstitution
| Stability and Storage | Product Form | Temperature Conditions | Storage Time (From Date of Receipt) |
|---|---|---|---|
| Lyophilized | -20°C to -80°C | Until Expiry Date | |
| Lyophilized | Room Temperature | 2 weeks | |
| Reconstituted as per CofA | -20°C to -80°C | 6 months | |
| Reconstituted as per CofA | 4°C | 1 week | |
| Avoid repeated freeze-thaw cycles. | |||
| Reconstitution | Briefly centrifuge the vial before opening. It is recommended to reconstitute the protein to 0.2 mg/mL in sterile 1x PBS pH 7.4 containing 0.1% endotoxin-free recombinant human serum albumin (HSA). Gently swirl or tap vial to mix. |
GMP Quality Policies
HumanKine® GMP (Good Manufacturing Practice) recombinant proteins are manufactured and validated in accordance with ISO 13485 quality management system and is compliant with GMP.
Our GMP recombinant proteins are animal component free (ACF), xeno free (XF) and tag free (TF). Read more about these policies here.
Background
EGF (epidermal growth factor) is a 6 kDA, single chain protein that serves a mitogenic factor for numerous cell types, particularly epithelial cells and fibroblasts. It is predominantly produced and secreted by the kidneys in humans. EGF activates cells through binding with its corresponding receptor (EGFR). Receptor binding activates several downstream pathways leading to cell proliferation, differentiation, and apoptosis. EGF plays important roles in embryonic development, tissue regeneration, transport through ion channels, and wound healing. Dysregulated EGF signaling has been linked with renal disease progression, and EGF levels could potentially be used as biomarker to monitor disease-related cell damage (PMID: 15110798, 24513230, 27868030).
Synonyms
Urogastrone