Molecular markers for liver cancer
Discover molecular markers for hepatocellular carcinoma.
Hepatocellular carcinoma (HCC) and cholangiocarcinoma (CCA), primary liver cancers, are the fifth most common cancers and the third most common cause of cancer mortality (Parkin DM, 2000). Gold standards for cancer diagnostics include classification and subtyping of cancer-based on imaging, serum markers, tissue analysis, and molecular analysis of markers for liver cancer.
Currently, biomarkers for large liver tumors and late-stage disease have been found and clinical trials with tivantinib, c-met inhibitor, are ongoing (Santro et al., 2013). Sorafenib, an inhibitor of tyrosine protein kinases, is used to treat advanced HCC (Llovet et al., 2008).
From 1973 to 2010, a significant improvement in HCC therapy outcome was noted. It is suggested that earlier detection of HCC at a curative stage and an increase in the number of patients, which undergo liver transplantation, contribute to this improvement (Njei et al., 2015).
Torbenson and Schirmacher (2015) stress that HCC has heterogeneous etiologic and molecular profiles and, therefore, varied curative responses. The high recurrence rate and restricted survival in HCC are associated with its high resistance to therapy. There is an urgent need for early diagnostics molecular markers and personalized treatment of HCC. Currently, 60-70% HCC are diagnosed based on liver imaging without histology of biopsied liver.
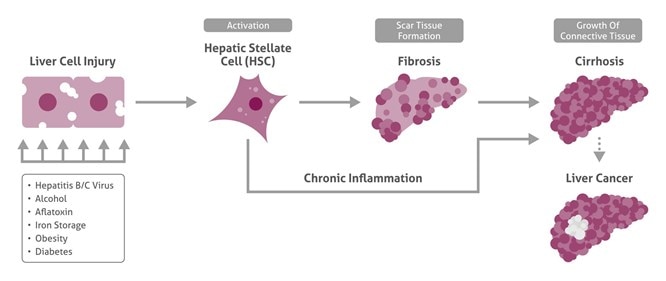
After liver damage and inflammation-related activation of hepatic stellate cells (HSC), fibrogenesis starts, leading further to cirrhotic liver and liver cancer.
Major molecular markers of late HCC have been identified
Critical molecular biomarkers that are associated with HCC have been recently found. These include deregulation of cell cycle and apoptosis checkpoints with activation of MDM2 and gankyrin and inactivation of ARF, Rb/p16, p27, p53 and IGF2R. In addition, it involves developmental and oncogenic pathways with activation of Wnt/b catenin, Hedgehog, MET, telomerase, myc, PI3K/Akt, bII-spectrin, and with inactivation of prickle 1 and Pten (reviewed by El-Serag and Rudolph, 2007; Kitisin et al., 2007). Further investigations of markers, as well as studies on HCC classification are ongoing.
Risk factors for HCC onset are clear
Important to note, that HCC is developed in the damaged liver (not healthy). In about 80% cases it follows cirrhosis. Risk factors of HCC development include chronic infection with hepatitis viruses (B, C, and D), having hemochromatosis, obesity, insulin resistance and diabetes mellitus, consumption of oral contraceptives, alcohol and as well as intoxication by aflatoxin, vinyl chloride and other hepatotoxins (El-Serag and Rudolph, 2007).
New molecular markers found to be downregulated or inactivated in HCC
In 2015, an investigation of HCC revealed new possible molecular targets for diagnostics and therapy. Therapy in HCC with downregulated molecular markers could be directed to restore respective protein levels.
Degirolamo et al. found that the downregulation of the farnesoid X receptor (FXR) is associated with HCC. This receptor is the transcriptional regulator of bile-acid homeostasis and is expressed in the liver and the intestine. Authors investigate FXR knockout and overexpression mouse models. When overexpressed in intestine, FXR restored bile-acid homeostasis through the FGF15 axis and prevented HCC development even in the absence of hepatic FXR. These results highlight the role of FXR in crosstalk between the gut and the liver. The authors suggest to use intestinal-selective FXR modulators in individuals with somatic FXR mutation to prevent HCC.
Koral et al., found that a loss of leukocyte-specific protein 1 (LSP1) leads to HCC. LSP1 as a target was found in the copy number variation analysis in human HCC. In the liver, LSP1 is normally expressed in macrophages and endothelial cells. In the hepatocytes, it is expressed only during proliferation until its cessation. The authors show that enhanced expression of LSP1 leads to decreased proliferation of rat hepatocytes in vitro and in vivo. They suggest testing LSP1 as possible target to early HCC diagnostics and HCC curation.
Zhi et al., suggest that bII-spectrin downregulation promotes HCC development. bII-spectrin is an adapter protein for Smad3/Smad4 in TGF-b signaling that regulates cell proliferation and apoptosis. The authors found that HCC cells with decreased bII-spectrin levels have activated Wnt signaling and acquisition of stem cell-like features. In addition to this, these cells possess very low levels of the known Wnt signaling inhibitor, kallistatin, and display dysregulation of TGF-b and Wnt signaling. Zhi et al., show that decreased bII-spectrin and kallistatin expression is associated with decreased relapse-free survival.
New molecular markers found to be upregulated or activated in HCC
These markers identified in 2015 could be selected for the HCC curation through blocking them.
Su et al., found that expression of known oncomarker of HCC, gankyrin, is promoted by IL-1b/IRAK-1 signaling through JNK and NF-Y/p300/CBP complex. Upregulation of gankirin can be blocked by respective IRAK-1 and JNK inhibitors. This work shows link between gankyrin, hepatic inflammation and HCC development. The authors suggest that inhibition of gankirin is a potential target to treat HCC.
Huntzicker et al., show that overexpression of Jag1 and Notch2 is associated with HCC development. The researchers used antibody to block Notch1, Notch2, Notch3, or jagged1 (Jag1) in a mouse model with HCC and CCA. They found that only inhibition of Jag1 and Notch2 reduces cancer progression, and these targets might be a therapeutic option for the treatment.
Lu et al., show that upregulation of C/EBPa is associated with HCCs. The authors examined 191 patients by using HCC tissue microarrays and found lower survival in C/EBP positive probes. C/EBPa is a transcription factor belonging to the CCAAT/enhancer-binding protein family involved in cellular differentiation and energy metabolism.
Ohrnberger et al., found that serum response factor (SRF) is overexpressed in HCC. SRF is a ubiquitously expressed transcription factor. It is controlled by both Ras/MAPK and Rho/actin signaling. The authors generated transgenic mouse with conditional expression of constitutively active SRF in hepatocytes. The mice livers develop HCC with activating Ctnnb1 mutations and hypomethylation of Igf2/H19 oncofetal genes. Ohrnberger et al., suggest simultaneous inhibition of Ras/MAPK and Rho/actin signaling for a subset of HCC with upregulated SRF.
Reichl et al., show that the upregulation and activation of the receptor tyrosine kinase Axl is involved in HCC development. The authors used mesenchymal hepatoma cell line and patient samples. Reichl et al., found that knockdown of Axl decreases migration of hepatoma cells and leads to resistance of cells to TGF-b-mediated apoptosis. Axl signaling causes phosphorylation of Smad3 linker region, consequent expression of PAI1, MMP9, and Snail. Moreover, Axl expression is associated with lower survival. These data show the link between Axl and TGF-b signaling. Furthermore, Axl is suggested to be a promising target for HCC therapy.
Srivastava et al., investigate roles of astrocyte elevated gene-1 (AEG-1) and c-Myc that are overexpressed in HCC. To understand the molecular mechanism of cooperation between these two oncogenes, authors generated transgenic mice that express either AEG, or c-Myc or both and investigated in detail which mice and under which conditions (spontaneous or with hepatotoxins) develop HCC and metastases. They also performed RNA-sequencing analysis and identified gene signature in livers of these mouse models. Srivastava et al., suggest to use these models for further investigation of mechanisms of aggressive HCC and as a test model for therapeutic targets.
Conclusion
HCC is characterized by molecular heterogeneity and is to 80% developed in damaged livers after 10-20 years of chronic damage. Strategies of personalized medicine, expression profiling of biopsied HCC tissues, biomarker validation and classification of HCC will improve early HCC diagnostics, targeted therapy and survival.
Related Links
Nature: recent liver related publications
References
- El-Serag HB and Rudolph KL. Gastroenterology 2007;132:2557-2576
- Degirolamo et al., Hepatology 2015;61:161-170
- Huntzicker et al., Hepatology 2015;61:942-952
- Njei et al., Hepatology 2015; 61: 191-199
- Ohrnberger et al., Hepatology 2015;61:979-989
- Kitisin et al., Oncogene 2007;26:7103-7110.
- Koral et al., Hepatology 2015;61:537-547
- Llovet et al., N Engl J Med 2008; 359:378-390
- Lu et al., Hepatology 2015;61:965-978
- Parkin DM. Lancet Oncol 2001;2:533-543
- Reichl et al., Hepatology 2015;61:930-941
- Santoro et al., Canet Oncol 2013Jan;14(1):55-63
- Srivastava et al., Hepatology 2015;61:915-929
- Su et al., Hepatology 2015;61:585-597
- Zhi et al., Hepatology 2015;61:598-61
About the Author
 |
Iryna Ilkavets studied biochemistry in Belarussian State University and got her PhD in molecular biology at University of Munich. She worked in the Institute of Photobiology of NASB (Minsk, Belarus), and then in Germany at Munich University, at Heidelberg University, and at Heidelberg Institute of Theoretical Studies. Since 2007, she has been focusing on liver pathology research.
|
Related Content
Cancer stem cells as a key to cure cancer

Support
Newsletter Signup
Stay up-to-date with our latest news and events. New to Proteintech? Get 10% off your first order when you sign up.
