Cancer stem cells as a key to cure cancer
A guide to cancer stem cells and the markers that can be used to isolate cell populations.
Although recent advances in immunotherapy and CAR-T cells have rapidly improved cancer therapy and remission rates, there is still no “silver bullet” for cancer. One possibility for this is that we are not targeting the right cells. Within tumors, there exists a subpopulation called the “cancer stem cells” (CSCs). These cells are cancerous yet have properties of stem cells: self-renewal and the ability to differentiate into multiple cell types that occurs in tumors.
Nearly 150 years ago, Julius Cohnheim first proposed the CSC hypothesis (also called the “embryonal-rest theory”) that suggested the presence of embryonic-like cancerous cell remnants in adult tissues that can develop into cancer in a non-spontaneous way (1). There was scant experimental evidence for this hypothesis until 1994, when John Dick and colleagues demonstrated that leukemia-initiating stem cells (LSCs) present in the blood of leukemia patients may induce acute myelogenous leukemia (AML) when transplanted into severe combined immunodeficient mice (2). Further research uncovered a broad spectrum of cell surface stem cell markers (e.g., CD133, CD44, and CD24) that allow the identification of CSCs in human solid tumors, including brain, breast, prostate, pancreas, liver, ovary, skin, colon cancers, and melanoma (3-6) (Figure 1 based on 7).
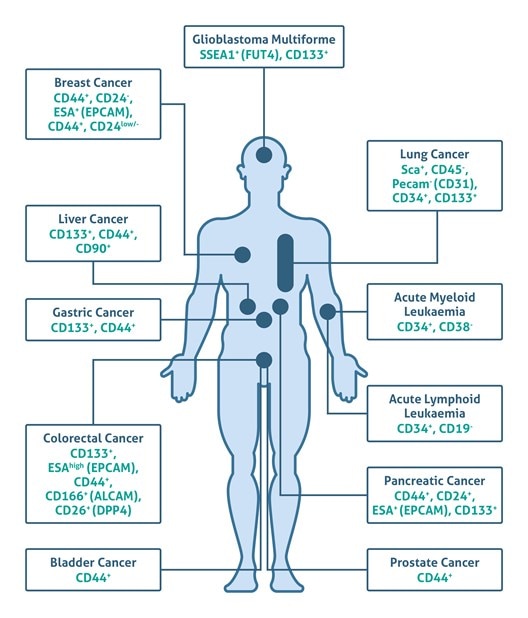 |
|
Figure 1 Click here to view an interactive version of this diagram.Table of Circulating Tumor Cell (CTCs) markers for flow cytometry |
Besides their stem cell properties, CSCs have become a focus of pharmacological studies due to their resistance to many cancer therapies. Although further investigation regarding CSCs is still needed, there is evidence that these cells play an important role in the prognosis of cancer, progression, and therapeutic strategy. In other words, long-term patient survival may depend on the elimination of CSCs.
The ability to isolate pure CSC populations using cellular markers has led to advances in our understanding of the heterogeneity and plasticity of the CSC phenotype. These markers include a broad spectrum of surface molecules such as CD44 (15675-1-AP), CD24 (18330-1-AP), CD133 (18470-1-AP, Figure 2), or LC3B (18725-1-AP, Figure 3).
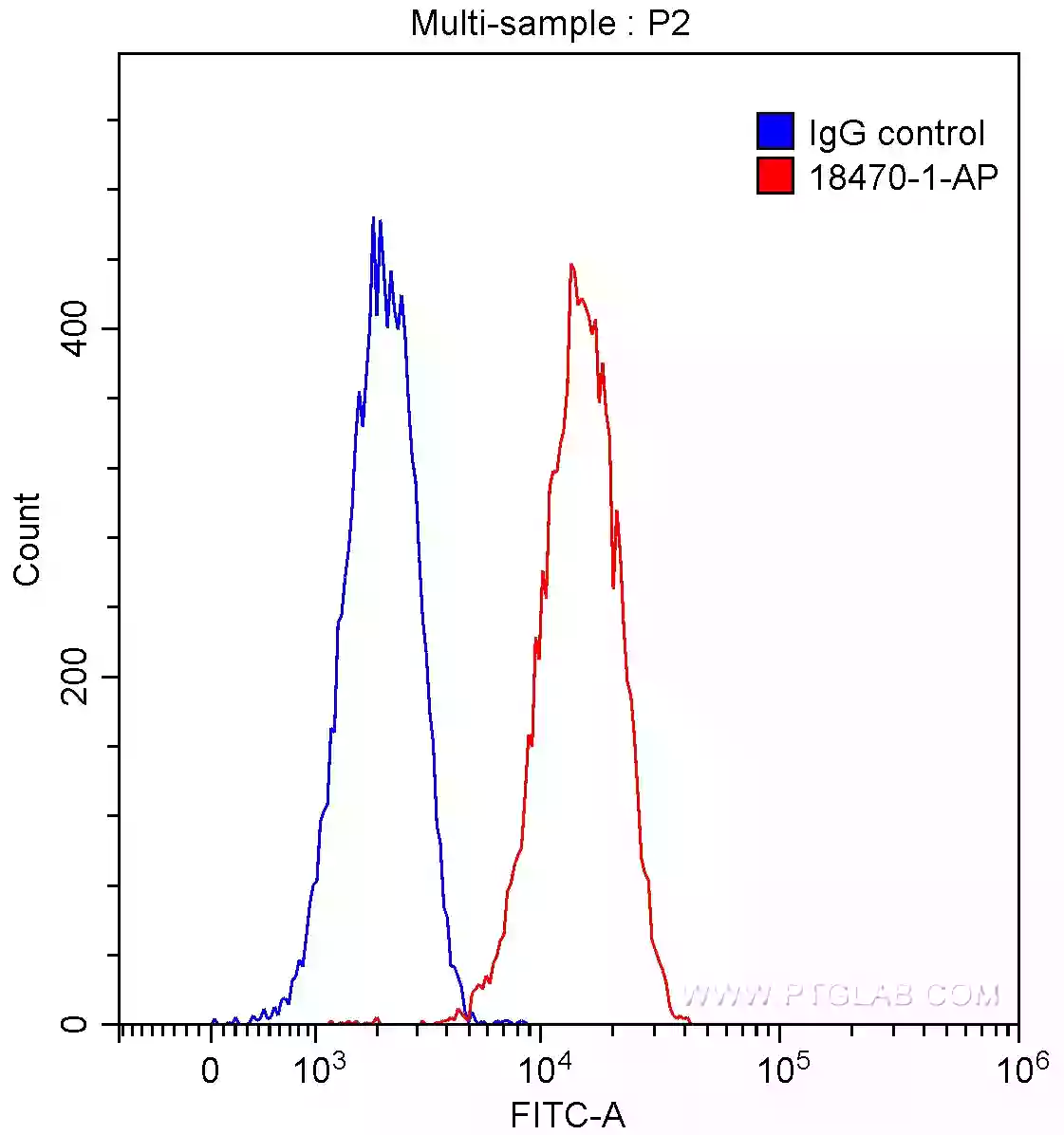 |
|
Figure 2. 1X10^6 HeLa cells were stained with 0.2ug CD133 antibody (18470-1-AP, red) and control rabbit IgG antibody (blue). Fixed with 4% PFA and blocked with 3% BSA (30 min). Alexa Fluor 488-conjugated AffiniPure Goat Anti-Rabbit IgG(H+L) was applied at a dilution of 1:1500. |
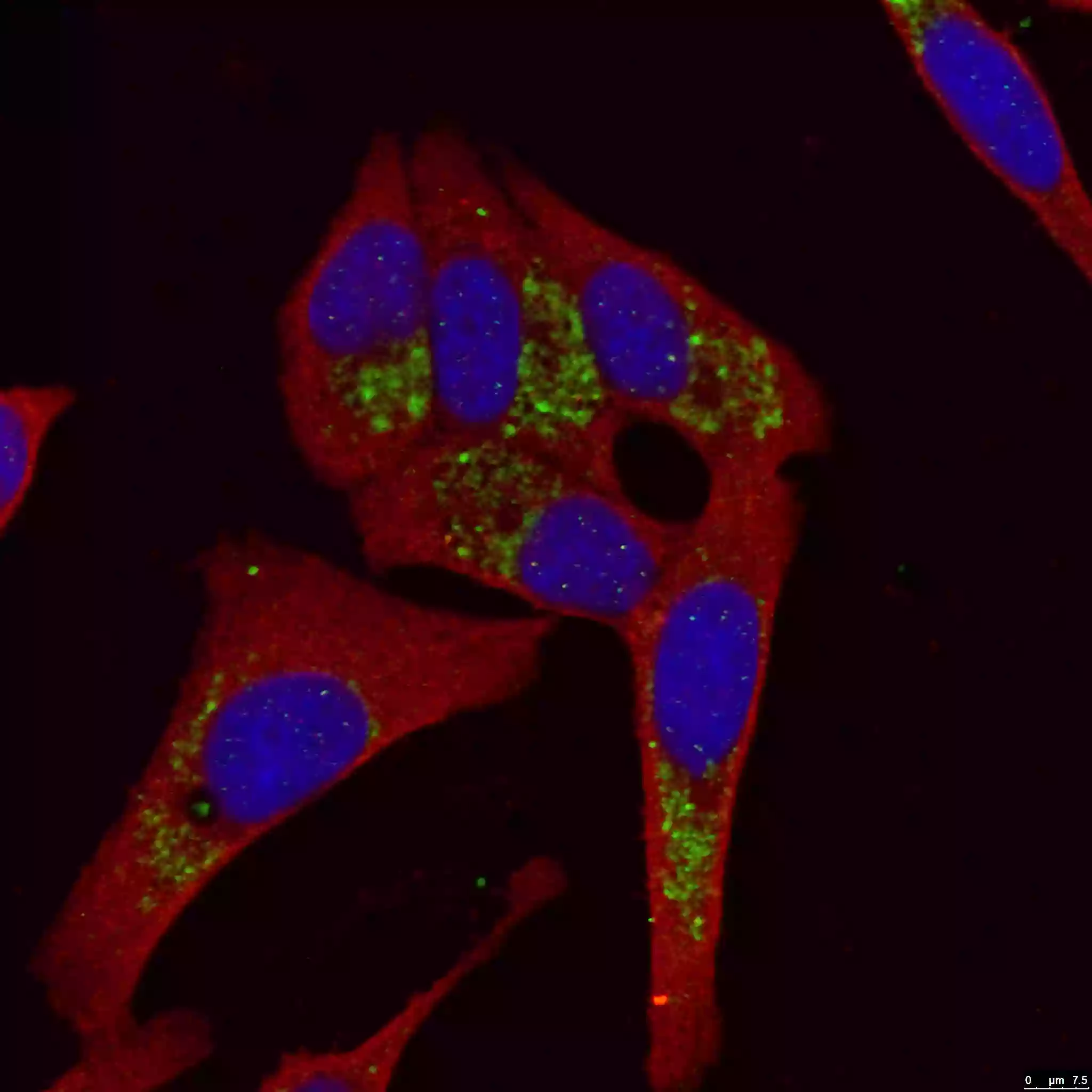 |
|
Figure 3. LC3B-Specific Antibody staining (18725-1-AP; 1:50; green) with HepG2 cells. Cells were fixed with 4% PFA, permeabilized with 0.2% Triton X-100, and co-stained with Tubulin (66031-1-Ig; 1:100; red), under 40x. |
Evidence supporting the CSC hypothesis and the potential of CSCs as a pharmacological target continues to accumulate. Since stem cells are also durable against cellular insults, one promising avenue for inducing the therapeutic sensitivity of CSCs is the blocking of either the properties or related pathways (Notch, Hedgehog, or Wnt, among others) of normal stem cells. Moreover, just as the stem cell niche plays a crucial role in the maintenance of stemness, many intra-microenvironmental connections and relationships between the microenvironment and the tumor play a crucial role in enriching the CSC subpopulation; as such, disrupting these relationships may be key to curbing their resistance to therapy. Additionally, CSCs could provide clues about the novel stem cell pathways activated during cancer progression, which, in turn, could guide further therapeutic steps and clinical trials design.
Written by Karolina Szczesna; PhD in Biomedicine
Product Manager, Proteintech Ltd.
References:
- Cells of origin in cancer.
- A cell initiating human acute myeloid leukaemia after transplantation into SCID mice.
- Identification and targeting of cancer stem cells.
- Identification of human brain tumour initiating cells.
- The role of CD133 in the identification and characterisation of tumour-initiating cells in non-small-cell lung cancer
- ALDH1-bright epithelial ovarian cancer cells are associated with CD44 expression, drug resistance, and poor clinical outcome.
- In vitro models of cancer stem cells and clinical applications.
Related Content
Molecular markers for liver cancer

Support
Newsletter Signup
Stay up-to-date with our latest news and events. New to Proteintech? Get 10% off your first order when you sign up.
