Polyclonal vs. monoclonal antibodies
Both types have their unique advantages and disadvantages and can be used in a wide variety of applications.
Summary
- Polyclonal: Best for high sensitivity, detecting low-abundance proteins, and quick capture (IP/ChIP). Recognition of multiple epitopes makes them robust but prone to batch variability.
- Monoclonal: Best for high specificity, quantification, and therapeutic use. Offers high reproducibility (single epitope) but is more expensive and time-consuming to produce.
- Recombinant: High-precision monoclonal antibodies produced via in vitro cloning to ensure maximum lot-to-lot consistency and eliminate genetic drift.
- Which antibodies should you choose?
- Choose polyclonal for maximum sensitivity in general research, monoclonal for high-specificity quantification, or recombinant for long-term study reproducibility.
Introduction
Antibodies are large Y-shaped proteins called immunoglobulins which are produced by B cells as part of the adaptive immune response when encountering a foreign molecule. Due the strong affinity of an antibody to one particular sequence, an epitope (typically 4-6 amino acids in length), they are widely used in research to identify and detect target proteins of interest in a variety of different applications. Of the available antibody isotypes, IgG is most commonly used for research. To answer different research needs, there are two types of antibodies available to scientists: polyclonal and monoclonal. Polyclonal antibodies contain a heterologous mixture of IgGs against the whole antigen, whereas monoclonal antibodies are composed of a single IgG against one epitope (Figure 1.)
This blog aims to give a comprehensive overview of the advantages and disadvantages of these two types of antibodies to enable the user to best choose the type most suitable for their application.
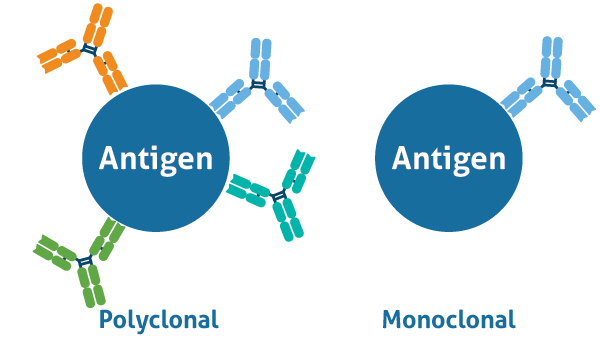 Figure 1. A) Polyclonal antibodies bind to the same antigen, but different epitopes; and B) monoclonal antibodies bind to the same epitope on a target antigen.
Figure 1. A) Polyclonal antibodies bind to the same antigen, but different epitopes; and B) monoclonal antibodies bind to the same epitope on a target antigen.
Polyclonal vs. monoclonal antibodies
This summary table highlights the five main differences between the two types of antibodies.
| Polyclonal antibodies | Monoclonal antibodies |
|---|---|
| Refer to a mixture of immunoglobulin molecules that are secreted against a particular antigen. | Refer to a homogenous population of antibodies that are produced by a single clone of plasma B cells. |
| Produced by different clones of plasma B cells. | Produced by the same clone of plasma B cells. |
| Production does not require hybridoma cell lines. | Production requires hybridoma cell lines. |
| A heterogeneous antibody population. | A homogenous antibody population. |
| Interact with different epitopes on the same antigen. | Interact with a particular epitope on the antigen. |
Polyclonal antibodies: Advantages and disadvantages
Advantages:
- Inexpensive and relatively quick to produce (+/- 3 months).
- Higher overall antibody affinity against the antigen due to the recognition of multiple epitopes.
- Have a high sensitivity for detecting low-quantity proteins.
- High ability to capture the target protein (recommended as the capture antibody in a sandwich ELISA).
- Antibody affinity results in quicker binding to the target antigen (recommended for assays that require quick capture of the protein; e.g., IP or ChIP).
- Superior for use in detecting a native protein.
- Easy to couple with antibody labels and rather unlikely to affect binding capability.
Disadvantages:
- Batch-to-batch variability as produced in different animals at different times.
- High chance of cross-reactivity due to a recognition of multiple epitopes (affinity purified antibodies display a minimum cross-reactivity).
Monoclonal antibodies: Advantages and disadvantages
Advantages:
- Batch-to-batch reproducibility (high homogeneity).
- Possibility to produce large quantities of identical antibody (an advantage for diagnostic manufacturing and therapeutic drug development).
- High specificity to a single epitope reflected in low cross-reactivity.
- More sensitive in assays requiring quantification of the protein levels.
- Low background noise.
Disadvantages:
- More expensive to produce. It is necessary to produce a pool of several monoclonal antibodies.
- Requires significantly more time to produce and develop the hybridized clone (+/- 6 months).
- More susceptible to binding changes when labeled (e.g. with a chromogen or a fluorophore).
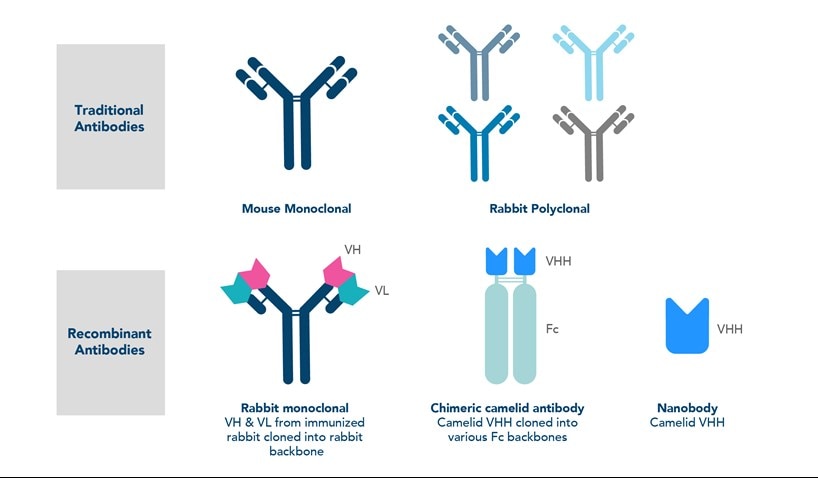
Recombinant antibodies
The next generation of monoclonal antibodies are recombinant antibodies, which are considered to be the future of antibody manufacturing. Recombinant antibodies are monoclonal antibodies that have been produced by in vitro cloning of the antibody heavy and light chain DNA sequences from the B cells or plasma cells of immunized animals. Unlike traditional monoclonal antibodies produced from hybridomas, the recombinant vectors are introduced into expression hosts (e.g. E. coli) to then produce the antibodies. This recombinant technology results in almost no lot-to-lot variability, removing the risk of genetic drift that can result in variations in of monoclonal antibodies.
Proteintech has a wide range of recombinant antibodies, and we are constantly adding new targets every month. Find out more about our recombinant antibodies and production process here.
Final remarks
Polyclonal antibodies are made using several different immune cells. They will have the affinity for the same antigen but different epitopes, while monoclonal antibodies are made using identical immune cells that are all clones of a specific parent cell.
For applications such as therapeutic drug development that require large volumes of identical antibody specific to a single epitope, monoclonal antibodies are a better solution. For general research applications, however, the advantages of polyclonal antibodies typically outweigh the few advantages that monoclonal antibodies provide. With affinity purification of serum against small antigen targets, the advantages of polyclonal antibodies are further extended.
FAQs
What is the difference between polyclonal and monoclonal antibodies?
Polyclonal antibodies contain a mixture of IgGs targeting multiple epitopes on a single antigen. This offers high sensitivity which is ideal for detecting low abundance targets, rare species, and mutated proteins. Monoclonal antibodies, by contrast, bind a single epitope making them advantageous for lower background, especially when analyzing high abundance targets. Recombinant antibodies are monoclonal antibodies that are produced by in vitro cloning of antibody heavy and light chains. This technology enables precise epitope recognition and consistent performance across lots, making them well-suited for reproducible results and long-term projects.
Is a monoclonal antibody better than a polyclonal antibody?
No, a monoclonal antibody is not necessarily better than a polyclonal antibody. The determining factor of which clonality to select will depend on your protein of interest. For example, low abundance proteins require maximum sensitivity, making polyclonal antibodies the smarter choice with a more robust signal. Monoclonal antibodies are best if you require high specificity and lot-to-lot consistency for reproducibility. However, signal might be harder to detect if your protein is not abundantly expressed.
Why do polyclonal antibodies vary between batches?
Polyclonal antibodies are produced by immunizing an animal (commonly a rabbit) with a specific antigen and collecting the resulting serum containing a heterogenous mixture of antibodies which recognize multiple epitopes. Lot-to-lot variability is an issue because each production cycle depends on a biological immune response that can differ over time, even within the same animal. Variability can be further amplified when different animals are used, as each will produce a unique immune response. As a result, polyclonal batches may differ in affinity, specificity, and sensitivity.
When should you choose a recombinant antibody over a monoclonal antibody?
Recombinant antibodies will be best for long-term projects where lot-to-lot consistency is critical or if you require large scale production. If a monoclonal antibody is not compatible with your experiment, recombinant technology also enables precise engineering of antibodies for optimal performance in a desired sample type or application.
When should you use a nanobody instead of a recombinant monoclonal antibody?
Nanobodies are a good choice if you are having difficulties with target accessibility. They are significantly smaller than rabbit monoclonal antibodies, allowing them to penetrate tissues and bind hard to reach epitopes with high affinity. They are ideal for certain applications, such as immunoprecipitation and super resolution microscopy. Recombinant monoclonal antibodies, on the other hand, have a full-length IgG structure which works better for multiple applications and compatibility with secondary antibodies.
Related Content
- How do I know if the antibody will cross-react?
- Protein or peptide antigen? Advantages and disadvantages
- Want to upgrade your immunofluorescence workflow? Go Direct!
- Secondary antibody selection
- Why are recombinant Nanobodies/ VHHs beneficial?
- How to write a good scientific abstract?
- Learn how to save precious hours on your IP, IF, and western blotting experiments
Support
Newsletter Signup
Stay up-to-date with our latest news and events. New to Proteintech? Get 10% off your first order when you sign up.

