Are Mesenchymal Stem Cells (MSCs) true stem cells?
A guide to MSC characteristics, differentiation pathways and supporting reagents for MSC culture.
Summary
- Mesenchymal stem cells (MSCs) are multipotent adult stem cells primarily found in bone marrow that can differentiate into chondrocytes, osteocytes, myocytes, and adipocytes.
- MSCs support tissue repair, bone regeneration, cartilage formation, and have immunomodulatory properties useful for treating inflammatory diseases.
- While called "stem cells," MSCs are more accurately pre-differentiated multipotent cells rather than true pluripotent stem cells like ESCs or iPSCs.
- Key growth factors for differentiation include FGF basic, TGF-β, BMPs, and PDGF, with specific combinations needed for each lineage (adipocytes, chondrocytes, etc.).
- Clinical applications are promising but face challenges like FDA approval, consistent protocols, and avoiding premature differentiation during expansion.
What are mesenchymal stem cells (MSCs)?
Mesenchymal stem cells (MSCs) are multipotent stem cells that are able to differentiate into a variety of different cells. First identified by Alexander Friedenstein in the 1960s–70s, they are primarily found in bone marrow at low concentrations (0.001–0.01%) and defined by plastic adherence in culture.
MSCs play a key role in skeletal tissue formation, maintenance, and repair, including bone and cartilage. Their primary differentiation pathways produce four main cell types: adipocytes (fat cells), myocytes (muscle cells), chondrocytes (cartilage cells), and osteocytes (bone cells), earning them the designation "multi-lineage stem cells."
Beyond bone marrow, MSCs can be isolated from diverse sources including adipose tissue, umbilical cord, amnion, synovial fluid, dermis, muscle, and dental pulp. They demonstrate three core therapeutic functions: tissue regeneration, angiogenesis promotion (particularly post-myocardial infarction), and immunomodulation that reduces inflammation and fibrosis.
These properties position MSCs as leading candidates for regenerative medicine and cell therapy, with clinical applications in graft-versus-host disease (GVHD), Crohn's disease, rheumatoid arthritis (RA), and multiple sclerosis (MS).
Due to low donor tissue yields, MSCs require controlled in vitro expansion using specialized media and growth factors. Proliferation capacity is finite, with senescence typically after 8–10 passages. Compared to other stem cells, MSCs exhibit superior self-renewal capacity and morphological characteristics of long, spindle-shaped, plastic-adherent cells.
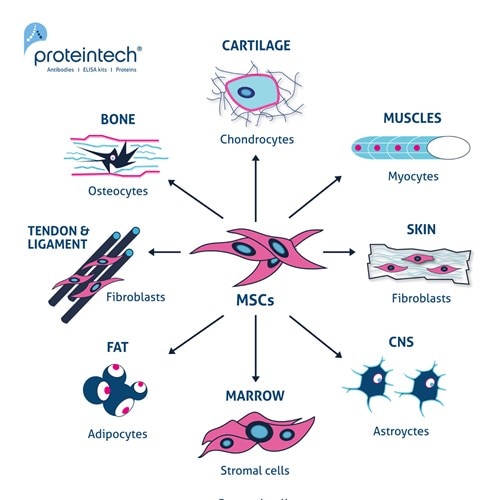
Figure 1. The multipotent potential of MSCs. Exposure to different inductive agents (e.g., cytokines and growth factors) can differentiate MSCs into chondrocytes, myocytes, fibroblasts, astrocytes, stromal cells, adipocytes, and osteocytes.
Mesenchymal Stem Cells: is it time to change the name?
Commonly, MSCs are thought to be true stem cells as they can undergo self-renewal and differentiation into a plethora of tissues of the mesenchymal lineage (6). However, MSCs are a particular type of pre-differentiated stem cells. They remain undifferentiated, but they are not true stem cells. Figure 2 contains further detail on the differentiation pathways of embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). ESCs and iPCs differentiate into Ectoderm, Mesoderm, and Endoderm lineages.
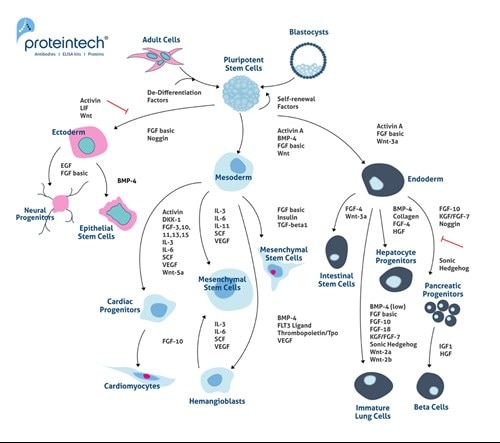
Figure 2. Differentiation pathways for embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). ESCs and iPCs differentiate into ectoderm, mesoderm, and endoderm lineages.
Which growth factors and cytokines are necessary for mesenchymal stem cells (MSCs) differentiation?
Depending on the environment MSCs grow in, they can give rise to a variety of lineage-specific cell types. The four principal cell types they differentiate into are myocytes, chondrocytes, osteocytes, and adipocytes. Table 1 contains a summary of the key growth factors needed for the differentiation process.
| Cell type | Growth Factor Used for Differentiation |
|---|---|
| Myocytes |
|
| Chondrocytes |
|
| Osteocytes |
|
| Adipocytes |
|
Table 1. Growth factors and cytokines needed for MSCs differentiation into a specific cell type.
How do mesenchymal stem cells (MSCs) support tissue regeneration?
Bone
The potential of MSCs for tissue repair and wound healing is well known. It has been shown extensively in the literature that MSCs and their progenitors are able to differentiate into chondrocytes under certain conditions in vivo and in vitro (2). Furthermore, these cells are nonimmunogenic; thus, allogeneic transplant of MSCs does not require host immunosuppression (7).
Cartilage
MSCs are able to repair cartilage. Indeed, depending on the culture environment, MSCs can differentiate into chondrocytes and stimulate chondrogenesis (3). It has been identified that a constant supply of insulin-like growth factor-1 (IGF-1) and transforming growth factor-β1 (TGF-β1) regulate the proliferation and differentiation of periosteal mesenchymal cells during chondrogenesis (3).
Heart tissue
MSCs affect post-acute myocardial infarction remodeling, stimulate regeneration of injured cardiac tissue, and induce coronary artery angiogenesis (10). Furthermore, MSCs can also differentiate into cardiac cell types, although complete differentiation into functional cells has not yet been achieved.
What are the future prospects for mesenchymal stem cells (MSCs)?
MSCs still require approval by the Food and Drug Administration (FDA). Currently, this process is delayed by inconsistent immunosuppression results, variability in cell quality, inconsistent protocols, varying dosages, and differing transfusion patterns (12).
One of the current challenges is avoiding the differentiation of MSCs into lineage-specific mature cells (12). In order to better understand the mechanism behind differentiation events, well-known growth factors and their interactions with MSCs need to be investigated further.
References
- Mesenchymal stem cells: clinical applications and biological characterization.
- Multilineage mesenchymal differentiation potential of human trabecular bone‐derived cells.
- Combined effects of insulin-like growth factor-1 and transforming growth factor-β1 on periosteal mesenchymal cells during chondrogenesis in vitro.
- Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Angiogenesis.
- Mesenchymal stem cells for the management of inflammation in osteoarthritis.
- Generation of functional mesenchymal stem cells from different induced pluripotent stem cell lines.
- Mesenchymal Stem Cells for Bone Repair and Metabolic Bone Diseases.
- Bone regeneration by implantation of culture-expanded human mesenchymal stem cells.
- Percutaneous autologous bone-marrow grafting for nonunions.
- Mesenchymal Stem Cells (Textbook Excerpt).
- Human Mesenchymal Stem Cells Differentiate to a Cardiomyocyte Phenotype.
- Mesenchymal stem cells and immunomodulation.
- Mesenchymal stem cells: Immunomodulatory capability and clinical potential.
