Imaging the Rainbow: Multiplexing with Same-Species Antibodies for Immunofluorescence
Improve your IF multiplexing with these expert tips from our guest blogger, Derek Sung (IG: @Immunofluoresence)
Immunofluorescence (IF) is an incredible biomolecular technique that allows for detection of specific proteins using antibodies against that target raised in an animal, such as a rabbit, mouse, etc. This technique allows for analysis of protein expression at the cellular and sub-cellular level while preserving cellular and tissue architecture and can be performed on anything from cultured cells to tissue sections to entire organs.
One of the biggest inconveniences of performing IF is when you want to stain for two different targets, but your antibodies are from the same host species. This is a cardinal rule you first learn when doing immunofluorescence: never use two antibodies from the same host species! The reason is that a secondary antibody directed at a specific host species will react with both antibodies. As a result, they will both fluoresce the same color, rendering them indistinguishable when imaging. This blog post will describe how to perform IF using two antibodies of the same host species along with examples using Proteintech and ChromoTek products.
There are a few ways of using two antibodies from the same species, such as using specific antibody isotypes (IgG vs. IgM) or subclasses (IgG1b vs. IgG2b) and their corresponding secondaries. Proteintech has a wide range of both primary and secondary antibodies that allows for this versatility. Another way is to use conjugated antibodies (antibodies with a fluorophore already attached) to multiplex with the same host species, which will be the focus of this blog post. If at least one of your antibodies is conjugated, you can do this method!
Conjugated antibodies are great for many reasons:
-
They allow for quick, single-step labeling
-
You can use two antibodies from the same host species
-
Your staining often has less background since no secondary antibody is used
Some disadvantages of conjugated antibodies are:
-
Losing flexibility in choosing the fluorophore/color
-
Inability to use two antibodies with the same fluorophore
-
Less signal amplification
Proteintech’s large catalogue of CoraLite conjugated antibodies covers a wide range of targets, many of which have multiple fluorophore options. These CoraLite conjugated antibodies provide robust fluorescent signals for high-quality IF imaging.
Outlined below are two scenarios in which you can use conjugated antibodies for multiplexing with antibodies of the same species.
One conjugated + one unconjugated antibody
- Unconjugated E-cadherin was first applied alone to the sample
- The sample was washed in 1X PBS
- An anti-rabbit secondary and DAPI were applied for 30 minutes
- The secondary was washed in 1X PBS at least 3 times
- CoraLite 555-conjugated Cytokeratin 7 was applied alone to the sample
- The sample was washed in 1X PBS, mounted, and imaged
Tip: Make sure the secondary antibody is completely washed off (Step 4). Extra washes never hurt since any remaining secondary antibody can cross react with your conjugated antibody!
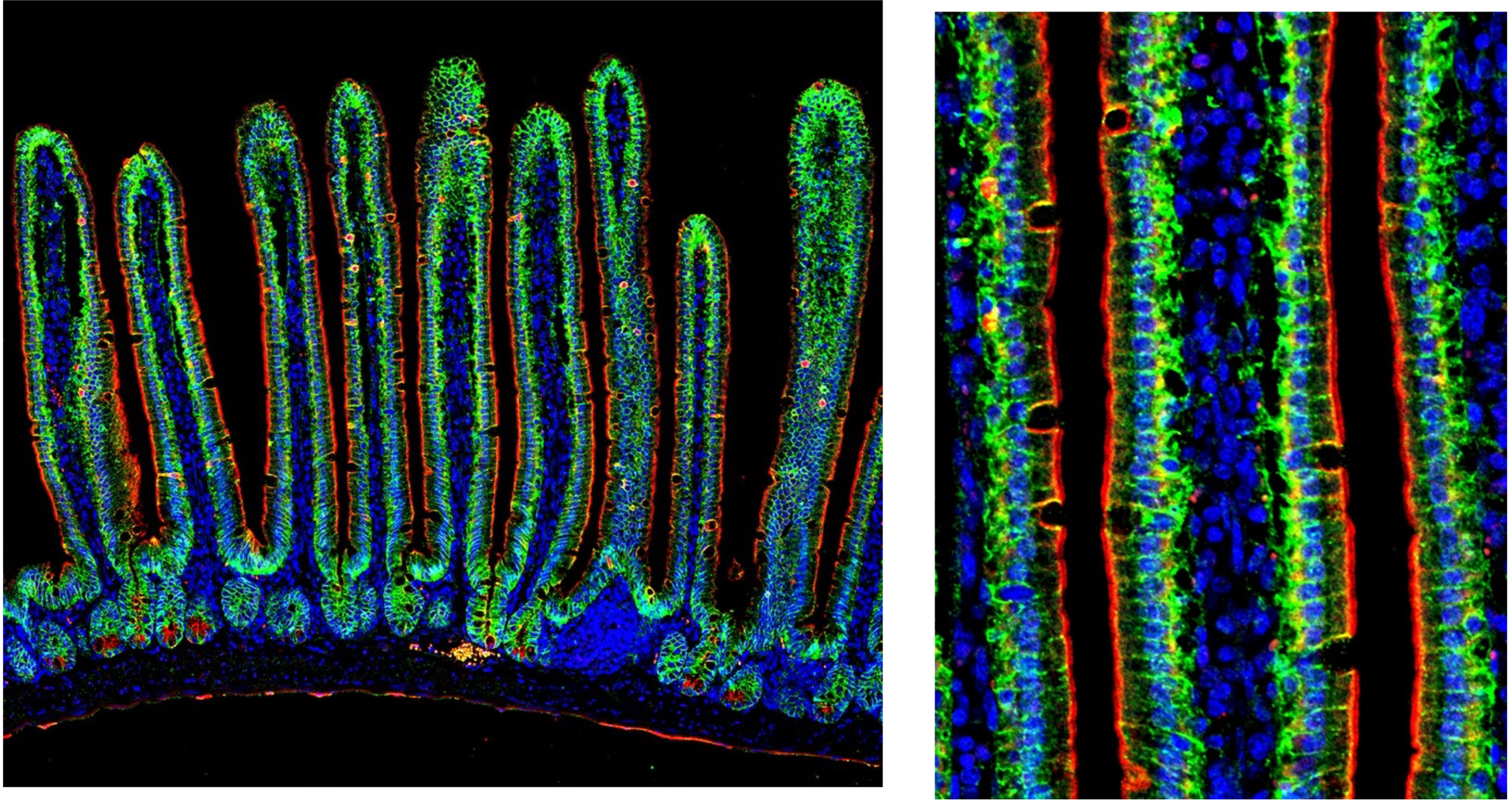
Image of adult small intestine using unconjugated E-cadherin antibody (green, 20874-1AP) and CoraLite 555-conjugated Cytokeratin 7 antibody (red, CL555-15539). Cytokeratin 7 labels the apical border of intestinal epithelial cells while E-cadherin labels cell-cell junctions in between epithelial cells.
Two (or more) conjugated antibodies
You’re in luck! If two (or more) of your antibodies are the same host species and conjugated, you can add them all directly to your sample. Since no secondary is involved, there will be no cross-reactivity. Just make sure all the conjugated antibodies contain different fluorophores.
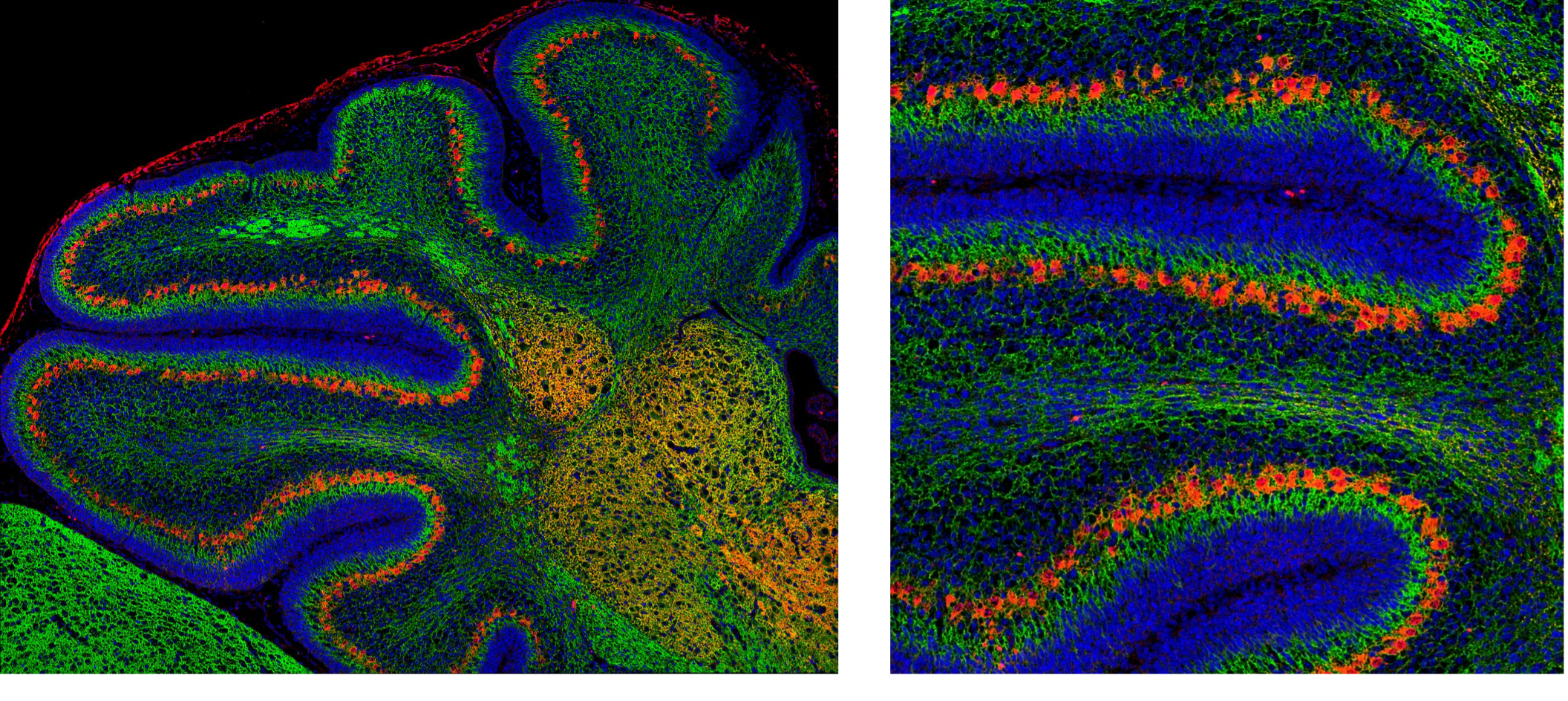
Image of neonatal cerebellum using CoraLite 488-conjugated Beta-III tubulin/TUBB3 antibody (green, CL488-66375) and CoraLite 594-conjugated Calbindin antibody (red, CL594-66394). Beta-III tubulin labels all neurons and calbindin labels Purkinje cells within the cerebellum.
If your antibodies from the same host species are not pre-conjugated from your supplier, you might need to label them yourself. Proteintech offers FlexAble Antibody Labeling Kits for fast and easy conjugations in just 10 minutes. FlexAble is a novel technology that uses a high affinity linker to conjugate fluorochromes, enzymes, and molecules in any buffer condition. Since the antibodies are labeled prior to incubating with your sample, FlexAble is ideal for same species multiplexing!
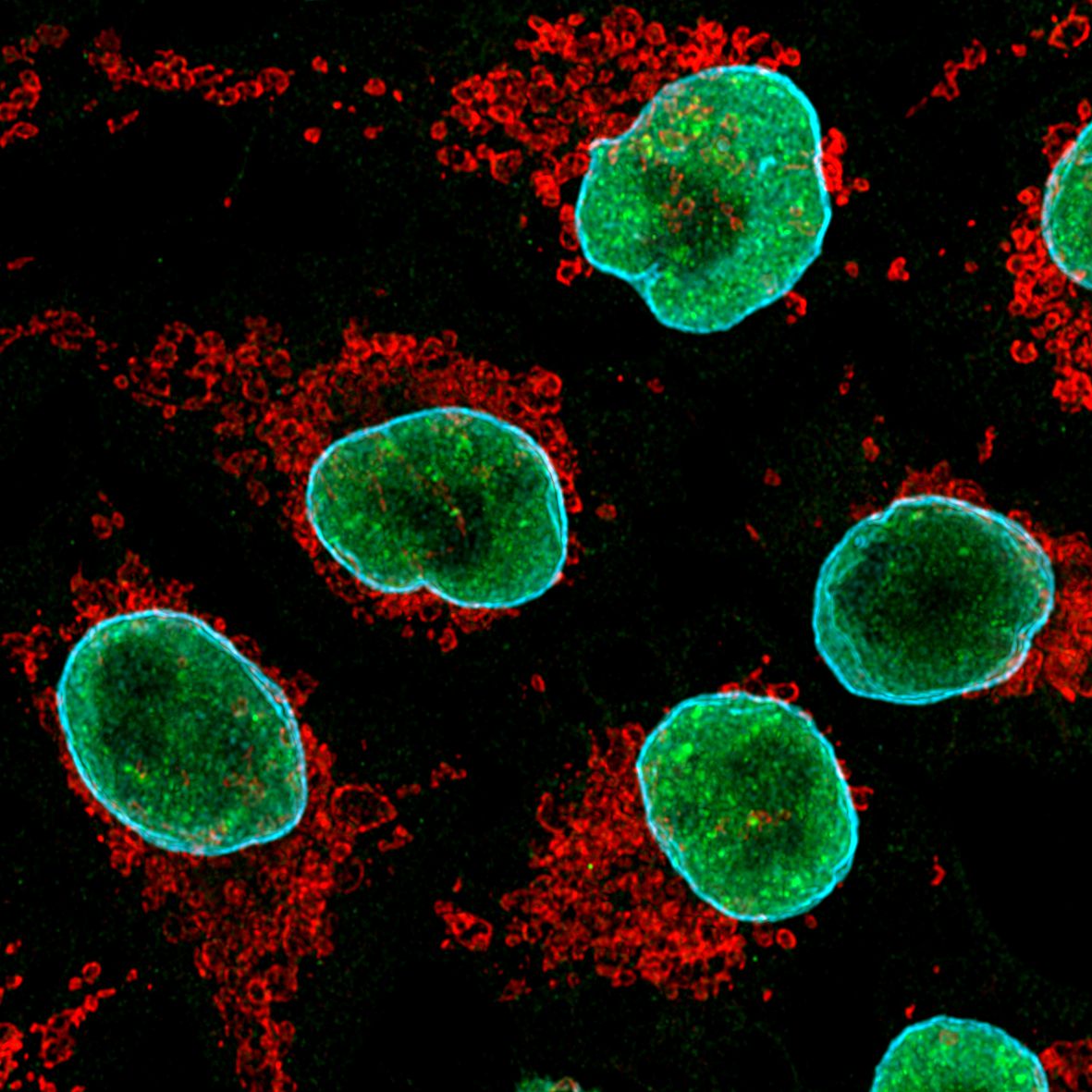 |
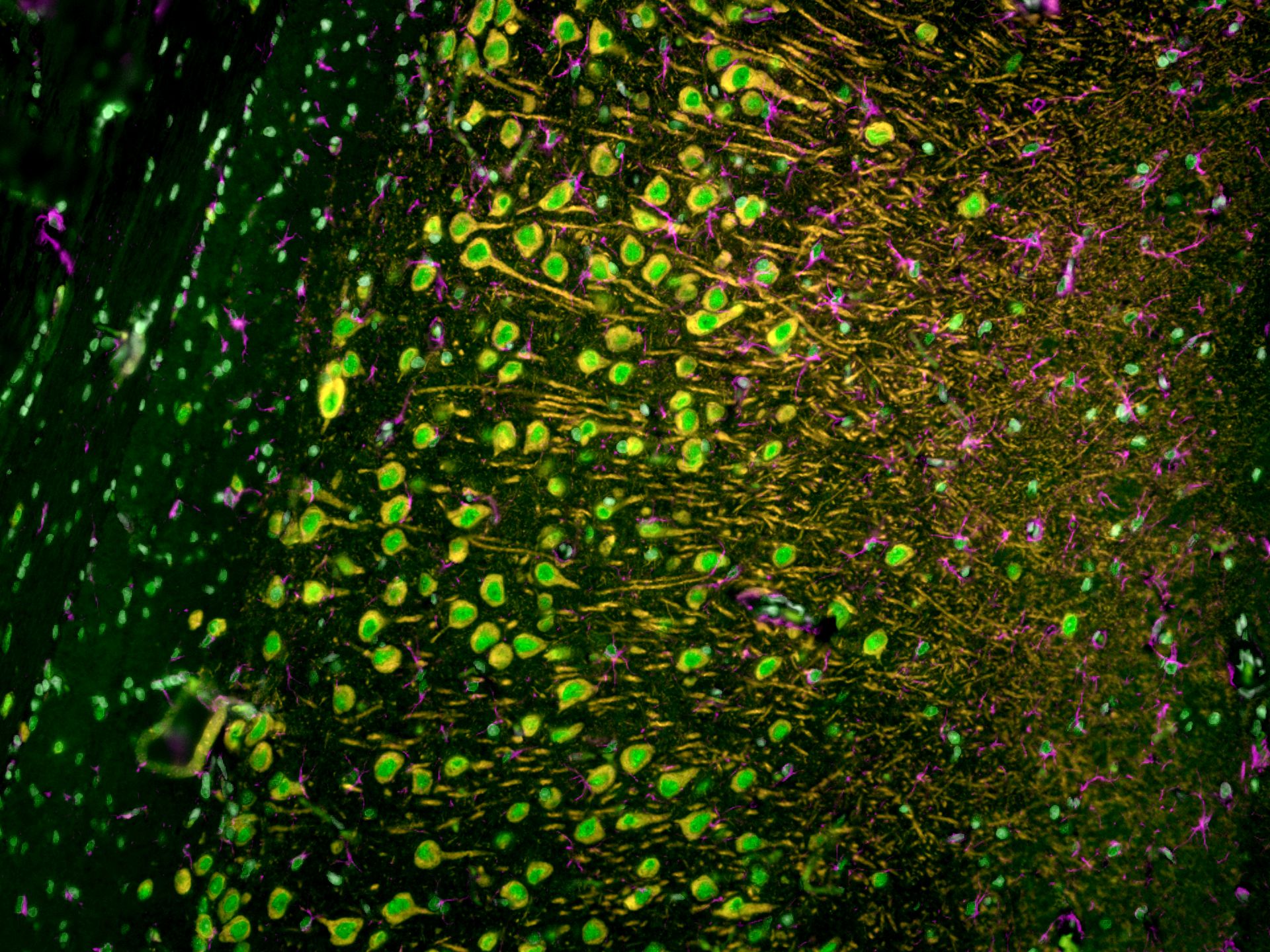 |
(Left) Image of cultured HeLa cells using anti-TDP-43 (10782-2-AP) labeled with FlexAble CoraLite® 488 Kit (KFA001, green), anti-Tom20 (11802-1-AP) labeled with FlexAble CoraLite® Plus 555 Kit (KFA002, red) and anti-Lamin B1 (12987-1-AP) labeled with FlexAble CoraLite® Plus 647 Kit (KFA003, cyan). (Right) Image of rat brain tissue using anti-MAP2 (17490-1-AP) labeled with FlexAble CoraLite® Plus 555 Kit (KFA002, orange), anti-GFAP (16825-1-AP) labeled with FlexAble CoraLite® Plus 647 Kit (KFA003, magenta) and CoraLite® 488-conjugated TDP-43 antibody (CL488-10782, green).
Related Content
CoraLite conjugated antibodies
Advantages of CoraLite direct staining for IF
Direct vs indirect immunofluorescence
How to reduce autofluorescence
Save time on your immunofluorescence experiments
9 Tips to optimize your IF experiments
Conjugation of fluorescent dyes to Nanobodies
GFP Nanobody for better images in immunofluorescence
GFP and RFP-Booster for better immunofluorescence imaging
Support
Newsletter Signup
Stay up-to-date with our latest news and events. New to Proteintech? Get 10% off your first order when you sign up.
