Cell & Gene Therapy Solutions
Harness the power of our GMP-grade cytokines, cell isolation kits, flow cytometry antibodies, and more to advance your cell therapy workflow.
-
Scale up supportOptimize production processes and increase cell numbers to meet the growing demand.
-
On-time deliveryMaintain your production schedule and prevent delays in the preparation of lifesaving therapies.
-
Regulatory complianceEach product meets rigorous quality standards, ensuring the integrity and efficacy of the final therapy.
Featured cell and gene therapy products
GMP-grade antibodies
Extensive portfolio of recombinant GMP-grade antibodies designed to support cell separation, activation, and enrichment across cell and gene therapy workflows. Each antibody is produced in ISO-certified cleanrooms with animal-free components, following USP and WHO guidelines to ensure superior safety, consistency, and reproducibility.
GMP-grade cytokines
Proteintech GMP grade HumanKine growth factors and cytokines are produced in human cells, ensuring the right post-translational modifications required for the highest bioactivity.
CAR detection reagents
Proteintech provides a comprehensive portfolio of CAR detection reagents optimized for high sensitivity, batch-to-batch consistency, and compatibility with diverse workflows enabling accurate quantification and monitoring of CAR expression. Designed for verification, in-process analytics, and release testing of CAR engineered cell products.
Flow cytometry antibodies
Enhance your research accuracy and efficiency with our comprehensive range of high-quality antibodies conjugated to highly bright CoraLite Plus dyes.
ELISA kits
Ultra sensitive ELISA kits for detection and precise quantification of secretomes of T cells, B cells, NK cells and many more.
Magnetic cell separation kits
Simply incubate your heterogeneous cell suspension with Proteintech’s magnetic cell separation particles to discover quick, easy, column-free cell depletions and isolations, resulting in a high purity homogeneous cell population.
Product data
HumanKine IL-2 demonstrating greater bioactivity and better T cell expansion
Interleukin-2 (IL-2) is a cytokine used for culturing a wide range of leukocytes; inducing proliferation in CD4+ and CD8+ T cells, maintaining normal T regulatory cells function, and is essential in immune homeostasis and self-tolerance.
Proteintech’s HumanKine human cell expressed recombinant IL-2 is up to 40x more active than our leading competitor IL-2 when benchmarked side-by-side in the competitor’s optimized assay protocol. The use of human expression systems in the production of IL-2 ensures that the protein is structurally and functionally optimized for compatibility with the human immune system. This results in a more efficient interaction between Humankine IL-2 and human T cells, leading to heightened bioactivity and increased proliferation rates.
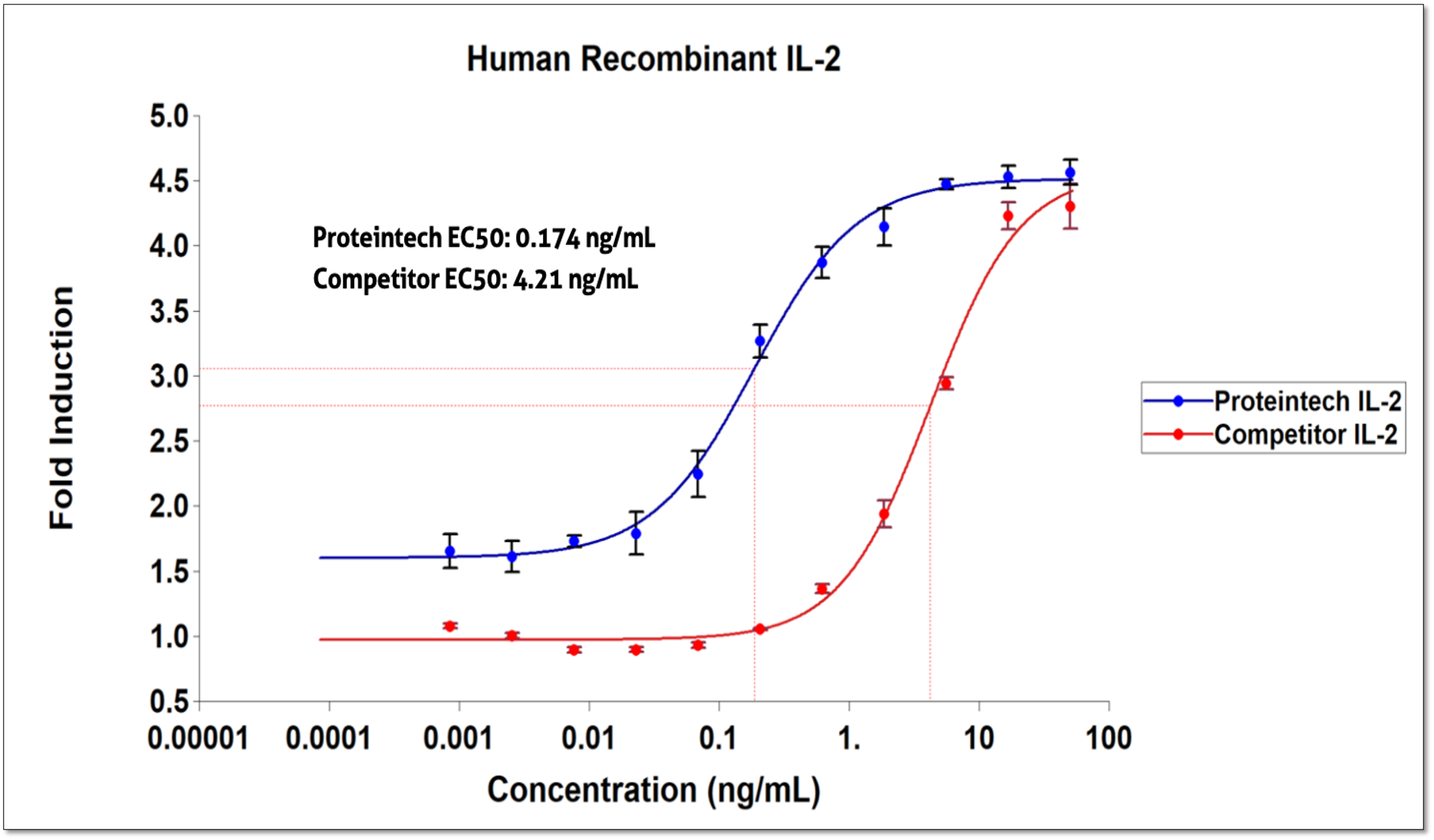
IL-2 bioactivity Comparison to Leading Competitor
Assay Method: IL-2 induction of CTLL-2 cells (mouse cytotoxic T cell)
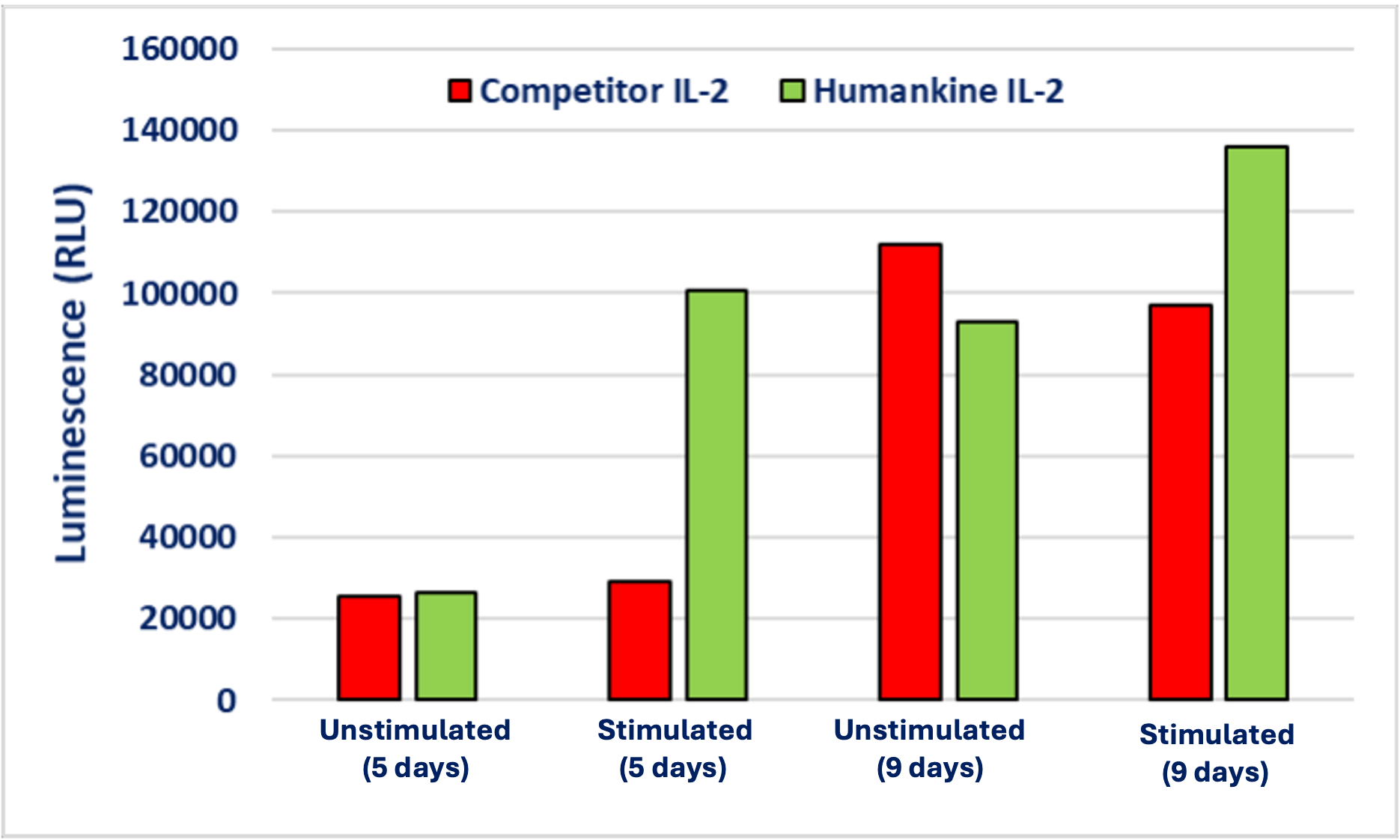
Comparison of Humankine IL-2 vs competitor product demonstrating greater levels of γδ T cell expansion and proliferation using HumanKine Il-2
The GMP CD3 (OKT3) antibody delivers performance comparable to the market standard
Proteintech’s GMP-grade CD3(OKT3) antibody demonstrates T cell activation performance equivalent to leading market alternatives. In comparative studies, it induced comparable levels of proliferation and activation marker expression (CD25, CD69), validating its potency and reliability. These findings confirm that GMP CD3 antibody delivers consistent, high-quality performance for T cell activation in cell therapy manufacturing workflows.
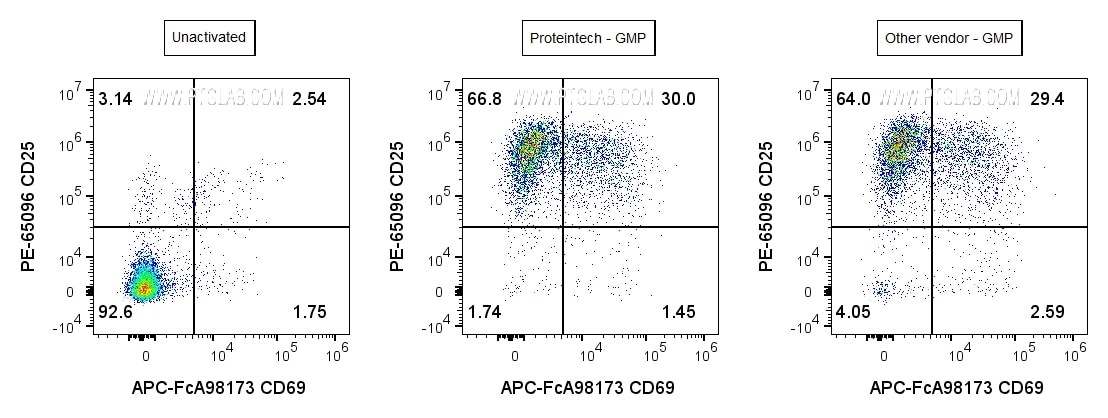
Human PBMCs were incubated for 3 days with soluble CD3 (OKT3) Recombinant Monoclonal Antibody (CT-1000-GMP) at 50ng/mL (center), or GMP-grade Anti-Human CD3 (OKT3) Antibody from another vendor at 50ng/mL (right), or cultured in media alone (left). Cells were then stained with Anti-Human CD3 (AB-65151), Anti-Human CD25 (PE-65096), Anti-Human CD69 (APC-FcA98173). Cells were gated on singlets (FSC-H vs FSC-A), live (Phantom Dye-), and CD3+ lymphocytes.
Human Memory/Naïve T Cell Panel for flow cytometry analysis
Characterizing T cell subtypes is critical to optimize methods for T cell expansion during cell therapy developmet. This panel is designed for the separation of T lymphocytes (CD3+) into Helper T Lymphocyte, (CD4+), Cytotoxic T Lymphocyte (CD8+), Naïve T Lymphocytes (CD45RA expressing) and Memory T Lymphocytes (CD45RO expressing). Naïve T cells have not previously encountered an antigen while memory T cells have and can therefore, more quickly respond to subsequent encounters of that antigen.
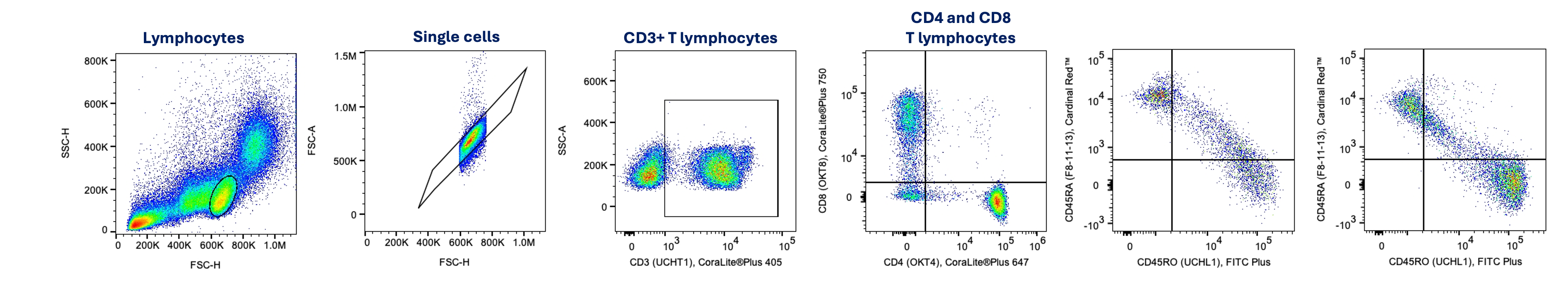
CD8+ Cytotoxic T lymphocytes (left) and CD4+ Helper T lymphocytes (right) were gated and evaluated for CD45RA and CD45RO expression.
Related Content
Key considerations for raw material selection in cell therapy manufacturing
Learn more about regulatory requirements and best practices in sourcing raw materials for cell based therapy manufacturing.
Non visible textTips and tricks for iPSC culture
Top tips for successful induced pluripotent stem cell and embryonic stem cell culture.
Non visible textCoraLite conjugates for Immunofluorescence
Largest portfolio of fluorophore conjugated primary antibodies on the market.
Non visible textIntroduction to t-SNE for Flow Cytometry
Understand the pros and cons of t-SNE, an algorithm for visualizing high-dimensional data.
Non visible textKey considerations for raw material selection in cell therapy manufacturing
Learn more about regulatory requirements and best practices in sourcing raw materials for cell based therapy manufacturing.
Non visible textTips and tricks for iPSC culture
Top tips for successful induced pluripotent stem cell and embryonic stem cell culture.
Non visible textCoraLite conjugates for Immunofluorescence
Largest portfolio of fluorophore conjugated primary antibodies on the market.
Non visible textIntroduction to t-SNE for Flow Cytometry
Understand the pros and cons of t-SNE, an algorithm for visualizing high-dimensional data.
Non visible text