Tubulin Antibodies
Cracking the tubulin code: diversity in cells and microtubular organelles
The tubulin superfamily is ubiquitously expressed in eukaryotes and constitutes the major components of microtubules, cytoskeletal structures involved in cell division, cell motility, and intracellular transport (1).
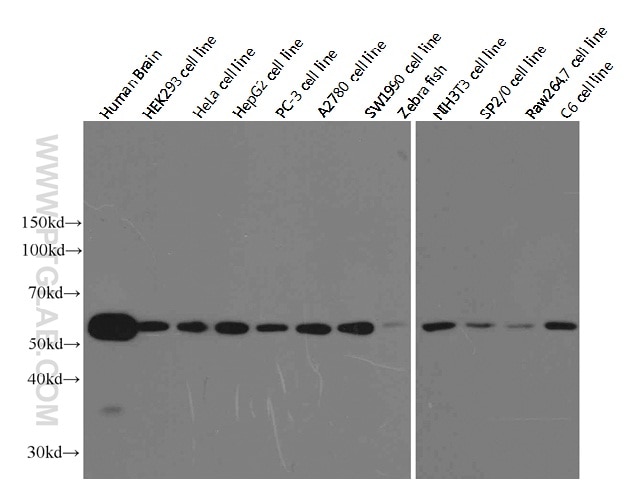
Besides these functions, the member α-tubulin is useful for scientists across fields as an internal control for Western blotting due to its high, ubiquitous expression pattern (Figure 1).
 |
|
Figure 1. Western blot analysis of alpha-tubulin in various tissues and cell lines using Proteintech antibody (66031-1-Ig) at a dilution of 1:10000. |
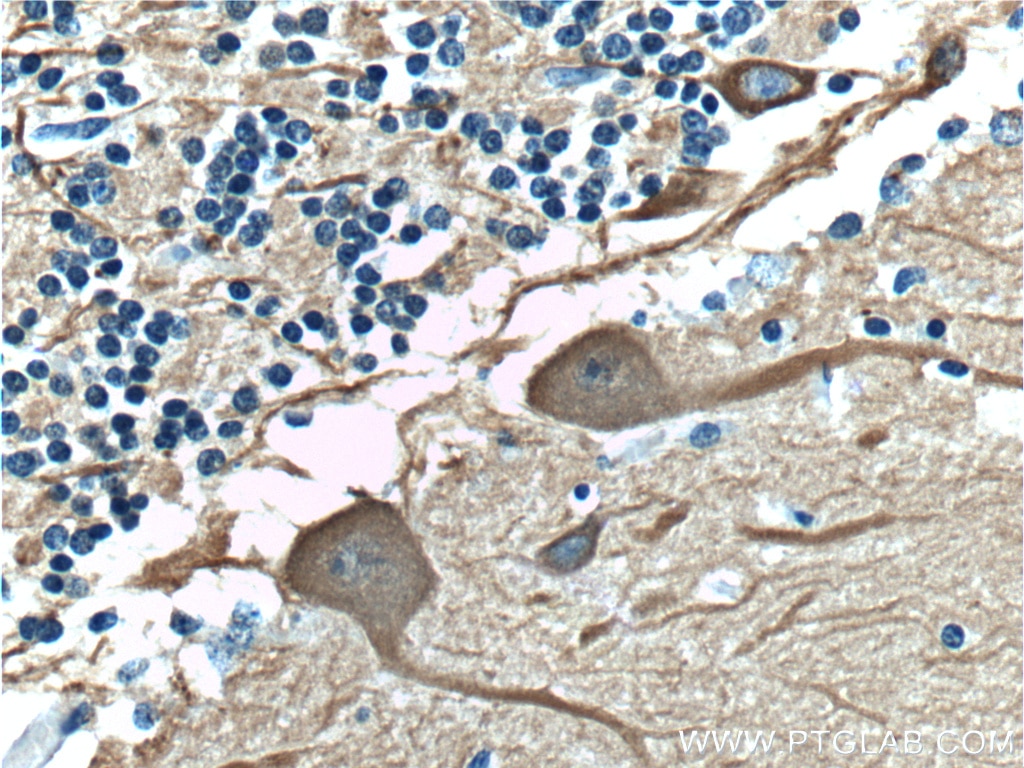
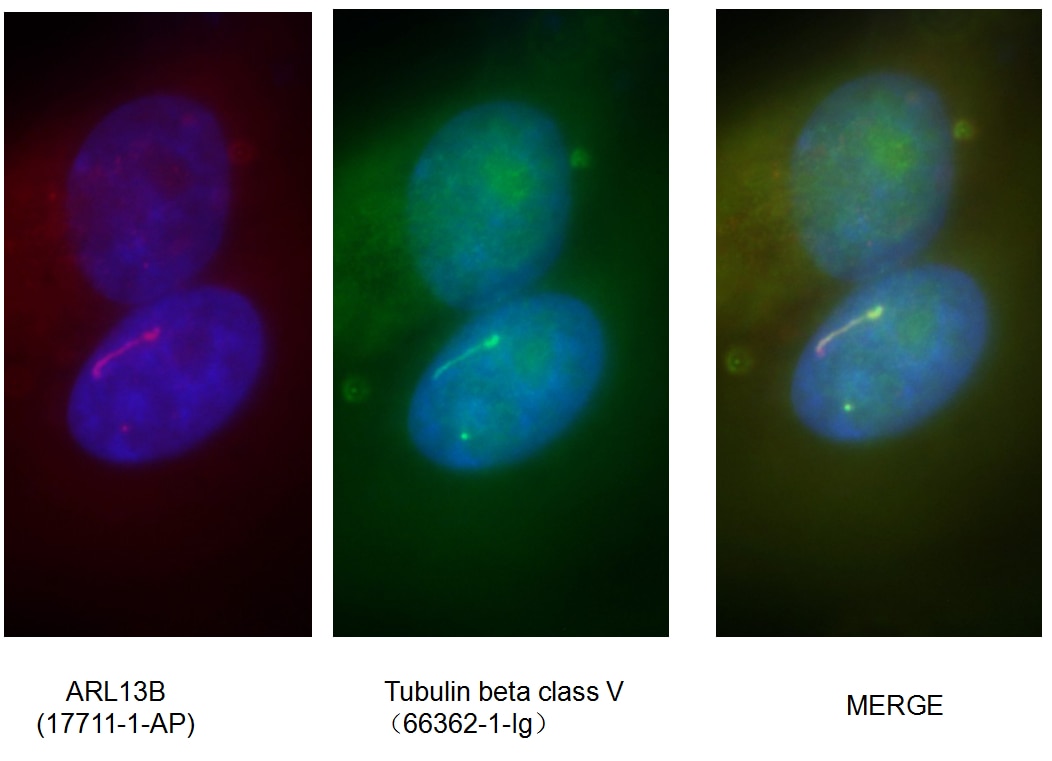
Mammalian β-tubulin comprises 7 isotypes: βI (TUBB), βII (TUBB2A, TUBB2B), βIII (TUBB3), βIVa (TUBB4), βIVb (TUBB2C), βV (TUBB6), and βVI (TUBB1). βIII and βV tubulin display a strong pattern of cellular expression and distribution. βIII tubulin is expressed in neurons of the central and peripheral nervous system at all developmental stages, making it a useful neuron marker (Figure 2) (2). Lastly, βV tubulin is mostly expressed in ciliary structures, and alterations in that expression pattern (e.g., diffuse expression) can be a marker of cancer (Figure 3) (3).
 |
|
Figure 2. Immunohistochemistry of paraffin-embedded human cerebellum tissue slide using TUBB3-specific Antibody (66375-1-Ig) at a dilution of 1:400 (under 40x lens). |
 |
|
Figure 3. Immunofluorescent analysis of fixed MDCK cells using ARL13B cilia marker (17711-1-AP) and Tubulin beta class V (66362-1-Ig) antibodies. |
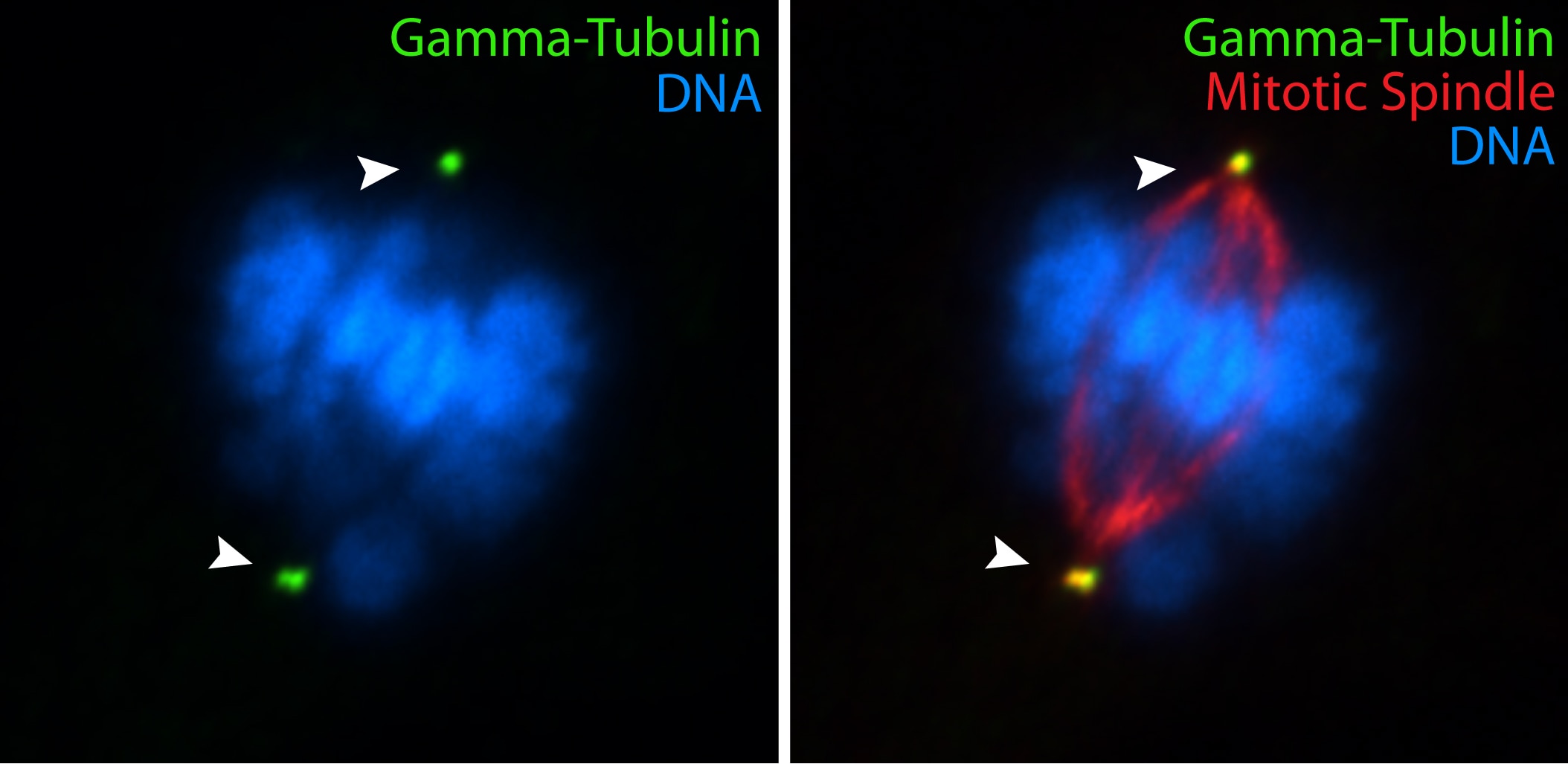
The highly conserved protein γ-tubulin (Figure 4) plays a major role in the nucleation of microtubule assembly and is required for microtubule nucleation in vivo (4).
 |
|
Figure 4. IF results with hTERT-RPE cells using gamma-tubulin (15176-1-AP) antibody (by Moshe Kim, laboratory of Dr. William S Trimble, University of Toronto). |
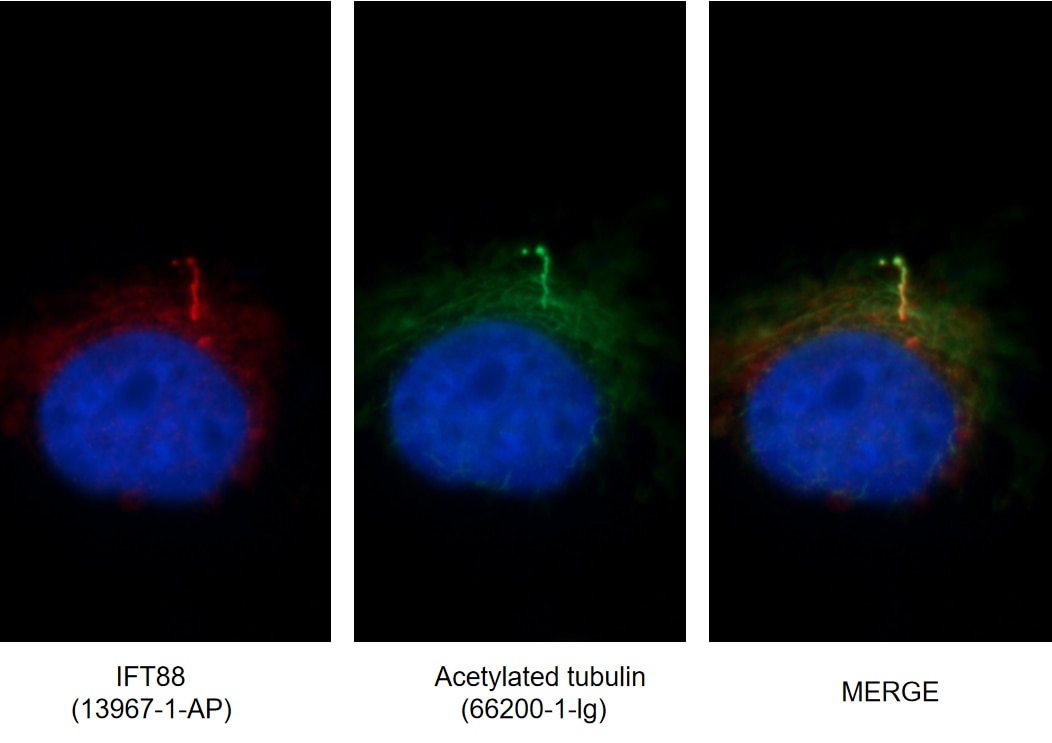
Tubulin and microtubules can be post-translationally modified in a variety of ways. Understanding the roles these modifications play in determining the functions and properties of microtubules has presented a major challenge that is only now being met (5). Acetylation occurs on lysine-40 in the N-terminal domain of α-tubulin and is implicated in regulating microtubule stability, cell motility, and axon regeneration. Recent studies present that acetylated α-tubulin is enriched in cilia and is widely used as a marker of primary cilia, a hair-like organelle on the surface of almost all cell types in vertebrates (Figure 5).
 |
|
Figure 5. Immunofluorescent images of MDCK cells stained for IFT88 rabbit pAb (13967-1-AP) and acetylated tubulin mouse mAb (66200-1-Ig) at dilution of 1:50, further stained with Alexa Fluor 594-conjugated AffiniPure Goat Anti-Rabbit IgG (H+L) for 13967-1-AP, and Alexa Fluor 488-conjugated AffiniPure Goat anti-Mouse IgG (H+L) for 66200-1-Ig. |
Determining how each modification affects cellular function remains a major challenge and has implications for health. In Alzheimer’s disease (AD), hyperphosphorylation of microtubule-associated protein tau results in microtubule destabilization and cytoskeletal abnormalities. Recent studies have uncovered that tubulin has increased levels of post-translational modifications in AD, suggesting the possibility that modifications may be a method of mitigating the effects of tau on the stability and function of microtubules (6). Further investigation may discover novel therapeutic and diagnostic approaches for AD.
Related Products
| Antigen Name | Catalog No | Application |
| alpha Tubulin | 66031-1-Ig | ELISA, IHC, FC, IP, WB, IF |
| 11224-1-AP | ELISA, WB, IP, IHC, FC, IF | |
| HRP-66031 | ELISA, WB | |
| acetylated Tubulin (Lys40) | 66200-1-Ig | ELISA, WB, IHC, IF |
| beta Tubulin | 10094-1-AP | ELISA, WB, IF, IP, IHC, FC |
| 10068-1-AP | ELISA, WB, IHC, IF, IP | |
| 66240-1-Ig | ELISA, WB, IHC, IF, IP | |
| TUBB3-Specific | 66375-1-Ig | ELISA, IHC, WB, IF |
| Tubulin beta class V | 66362-1-Ig | ELISA, WB, IF, IHC |
| gamma Tubulin | 15176-1-AP | ELISA, IF, WB, IP, IHC |
| 26195-1-AP | ELISA, WB, IF | |
| 66320-1-Ig | ELISA, WB, IF |
Loading control antibodies
| GAPDH Antibody | 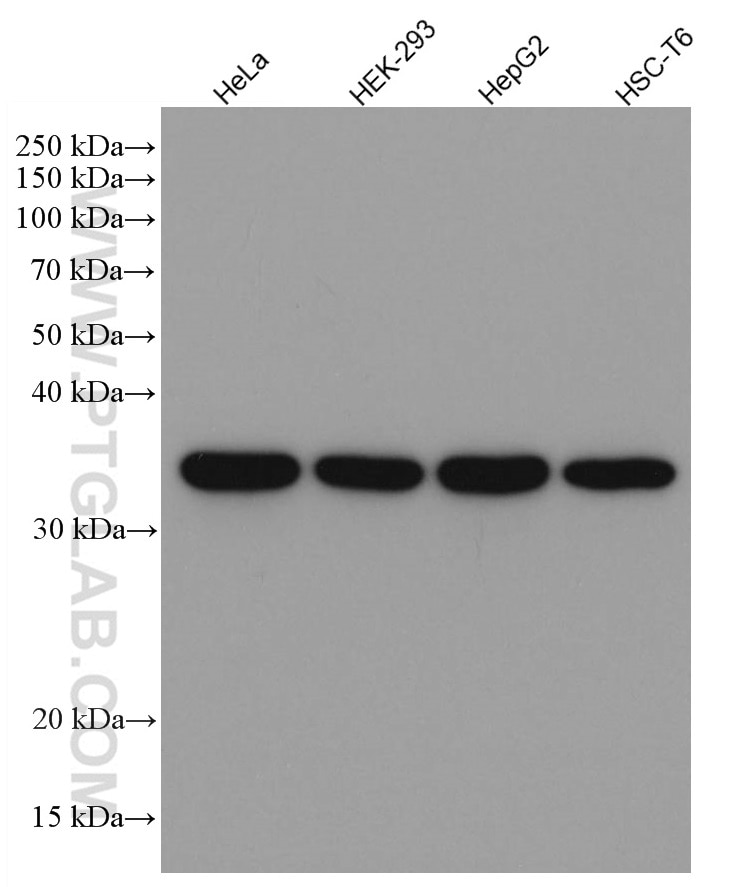 |
| Catalog no.: 60004-1-Ig | |
|
GAPDH is commonly used as a protein loading control in western blot due to its consistently high expression in most cell types. This enzyme participates in several cellular events such as glycolysis, DNA repair, and apoptosis. Proteintech monoclonal GAPDH antibodies are raised against a whole-protein antigen of human origin and have over 4,960 citations. |
| Beta Actin Antibody (KD/KO validated) | 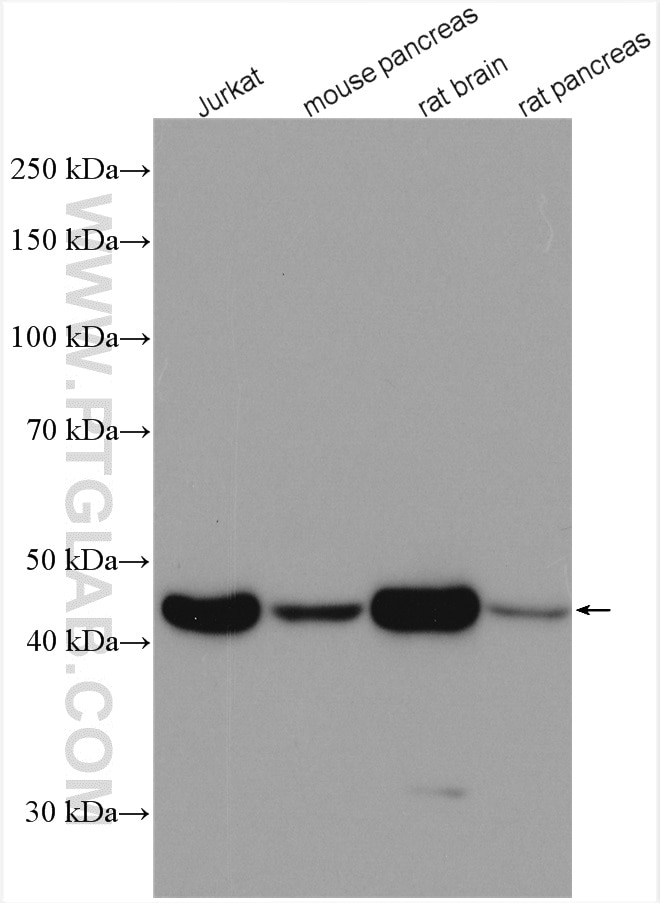 |
| Catalog no.: 66009-1-Ig | |
|
Beta-actin is usually used as a loading control due to its broad and consistent expression across all eukaryotic cell types and the fact that expression levels of this protein are not affected by most experimental treatments. 66009-1-Ig has been cited in over 2,460 publications and has wide species reactivity. |
